Abstract
Therapeutic goals for HIV-infected patients receiving antiretroviral therapy include minimizing risk of future physical disability. Presarcopenia and sarcopenia precede age-associated physical disability. We investigated their prevalence and the predictive value of patient mid-upper arm circumference (MUAC) for them. Eighty community-dwelling patients ≥45 years old demonstrating durable viral suppression were evaluated. Sarcopenia was defined as low skeletal muscle index (SMI, skeletal muscle kg/height m2) and either low strength or poor performance by handgrip dynamometry and gait speed, respectively. Presarcopenia was defined as low SMI only. MUAC was interpreted according to National Health Statistics percentile. Prevalence of sarcopenia and presarcopenia was 5.0% and 20.0%, respectively. Male gender (odds ratio [OR] 10.72; P < .026), recreational psychoactive substance use (OR 5.13; P < .037), and intravenous drug use transmission category (OR 6.94; P <.0327) were associated with presarcopenia. Higher body mass index (OR 0.80; P < .0007), MUAC (OR 0.83; P < .024), and large skeletal frame (OR 0.09; P < .003) were negatively associated with presarcopenia. Finding that a participant did not have a MUAC <25th percentile on physical examination had a 90.4% negative predictive value for presarcopenia. Although sarcopenia was uncommon, presarcopenia was highly prevalent in midlife and older HIV-infected males. Determination of MUAC percentile may identify those least likely to demonstrate skeletal muscle deficit and improve patient selection for mass and function testing.
Introduction
Life expectancy for people with HIV infection receiving combination antiretroviral therapy (cART) has increased markedly and is expected to approach that of the general population. 1,2 In New York State alone, over 75% of people living with HIV infection are older than 40 years of age, and 40% are 50 years of age or older. 3 Long-term survival in some HIV-infected persons may be accompanied by chronic immune activation that may potentiate age-related low-grade inflammation, cellular senescence, and physiologic decline. 4 Maximizing disability-free life expectancy has now become a focus of attention in HIV medicine and research.
Sarcopenia, central to the etiology of frailty, precedes age-associated physical disability and is defined as age-related loss of skeletal muscle mass and function. 5 In contrast, presarcopenia is defined as age-related loss of skeletal muscle mass only. 5 Investigators have documented age-related decline in skeletal muscle mass initiating in the third or fourth decade and becoming prominent after 50 years of age in the general population. 6 As a result, older persons may have difficulty executing instrumental activities of daily living and experience social isolation. 7,8 With more severe skeletal muscle deficit, they may experience a predisposition to falls and fragility fractures with ensuing physical disability, leading to dependence. 7,9 In a predominantly midlife European HIV cohort, one-half of subjects performed poorly in tests of lower limb muscle performance and balance. 10
Skeletal muscle volume is the predominant determinant of mid-upper arm circumference (MUAC) in nonobese persons. Determination of MUAC is recommended by the United Nations Subcommittee on Nutrition in the assessment of adult undernutrition. 11 Limb skeletal muscle and MUAC have been independently associated with all-cause mortality in community–dwelling, HIV-infected midlife adults and midlife and older adults without known HIV infection. 12,13 In addition, MUAC has been validated as a screening tool for undernutrition in a number of settings (ie, antenatal care, geriatrics, etc). 14,15
The goals of the present study were to determine the prevalence of sarcopenia and presarcopenia in community-dwelling HIV-infected persons aged 45 years or older stably receiving cART. Additionally, since limb skeletal muscle constitutes more than 75% of total body skeletal muscle and is independently associated with femoral neck bone mineral density, we evaluated the predictive value of MUAC for sarcopenia and presarcopenia. 16,17
Methods
This was a cross-sectional, period-prevalence study conducted in an academic infectious disease practice within an urban New York State AIDS Designated Center. Community-dwelling, HIV-infected male and female patients aged 45 years or older, receiving cART, and presenting for routine care were asked by their physician to participate. Eligible patients demonstrated undetectable (<20 copies/mL) HIV-1 RNA by quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) for a minimum of 18 months with no more than 2 isolated intercurrent assays (“blips”) with detectable HIV-1 RNA <400 copies/mL. Patients with persistent viremia and those with malignancy were excluded to prevent confounding by skeletal muscle loss unrelated to aging (ie, AIDS wasting, untreated HIV infection, cancer cachexia, etc). Other exclusion criteria included acute medical illness, presence of skeletal prostheses or orthoses, paralysis, Parkinson's disease, and Asian ethnicity (to date, the skeletal muscle predictive equation has not been validated in persons of Asian ethnicity). 18 All participants met with a single investigator for the study procedures. The New York Hospital Queens institutional review board approved the study, and informed consent was obtained from all participants.
Transmission category (mode of HIV acquisition), comorbidities, concurrent medications, recreational psychoactive substance use, educational attainment, years of known HIV infection, and cART were determined by physician documentation and patient report. Medications for comorbidities were recorded and grouped for statistical analysis according to the Anatomical Therapeutic Classification System. Participants’ current cART was recorded and categorized for statistical analysis as follows: nucleoside reverse transcriptase inhibitors only (NRTI), non-nucleoside reverse transcriptase inhibitor (NNRTI)–based, integrase inhibitor (InI)–based, or protease inhibitor (PI)–based. Hepatitis B virus and hepatitis C virus (HCV) serology was performed routinely at patient entry into care. Lymphocyte subset panels and HIV-1 qRT-PCR were performed at Lab-Corp (Raritan, New Jersey) or Quest Diagnostics (Teterboro, New Jersey).
We used the European Work Group on Sarcopenia in Older People (EWGSOP) height-normalized diagnostic criteria and recommended clinical testing for the evaluation of patient skeletal muscle mass, strength, and function. 5 Stature is a correlate of gait speed (GS), hand grip strength, and skeletal muscle mass. 19–21 Normalization of these parameters for height allows interindividual and intergroup comparisons. Skeletal muscle mass (kg) was determined by tetrapolar bioelectrical impedance analyzer resistance in ohms (Quantum 101Q; RJL Systems, Clinton Township, Michigan) using the equation of Janssen et al. 22 Low skeletal muscle index (SMI, skeletal muscle kg/height m2) was defined as ≤10.75 for males and ≤6.75 for females. 5,22 Sarcopenia was defined operationally as low SMI and either low muscle strength or performance. 5 Skeletal muscle strength was evaluated by handgrip dynamometry and performance by observed GS. Presarcopenia was defined operationally as low SMI only. 5
Handgrip strength measurement employed American Society of Hand Therapists standardized procedure and isometric dynamometer (Jamar; Sammons-Preston, Bolingbrook, Illinois). 23 Tabulated handgrip strength (kg) was determined by the mean of 3 dominant hand measurements. Low handgrip strength was defined for males as follows: body mass index (BMI) ≤24, ≤29 kg; BMI 24.1 to 28, ≤30 kg; and BMI > 28 ≤ 32 kg. It was defined for females as follows: BMI ≤ 23, ≤17 kg; BMI 23.1 to 26, ≤17.3 kg; BMI 26.1 to 29 ≤18 kg; and BMI > 29, ≤21 kg. 5
GS was evaluated by timed 4.572 m walk at the patients’ usual pace from a standing start. It was determined in meters per second (m/s) and tabulated as the mean of 2 walks. Low performance cut points were determined for males as ≤1.73 m height, GS < 0.65 m/s; >1.73 m height, GS < 0.76 m/s. Cut points were determined for females as ≤1.59 m height, GS < 0.65m/s; >1.59 m height, < 0.76 m/s. 5
BMI was calculated as total body weight in kg/height in m2. Skeletal frame size was evaluated by height-to-wrist circumference ratio. 24 MUAC was measured at the midpoint between the acromial and olecranon processes of the scapula and ulna, respectively. National Health and Nutrition Examination Survey reference data were used to ascertain MUAC percentile. 25
Statistical analysis of the data was performed using SAS version 9.2 (SAS Institute, Cary, North Carolina). The relationships between skeletal muscle and handgrip strength, GS, height, age, and weight in all study participants, for each gender, were determined using the Spearman correlation coefficient.
The prevalence rates and exact 95% binomial confidence intervals (CIs) were determined for sarcopenia and presarcopenia. Persons with sarcopenia were excluded from subsequent analyses to identify characteristics that place people at risk for presarcopenia. Associations with presarcopenia were analyzed in age-adjusted linear regression models of the function of demographic and clinical characteristics. Statistical significance was defined as P < .05.
MUAC was also dichotomized into <25th percentile and ≥25th percentile for the determination of measures of validity (sensitivity, specificity, positive, and negative predictive values). MUAC <25th percentile was defined as a positive screen and MUAC ≥25th percentile a negative screen for presarcopenia.
Results
Patients were recruited for the study over a 7-month period (January to July 2011). Of the 205 patients screened, 82 met the entry criteria and consented to participation in the study. Two patients were excluded, one for carpal tunnel syndrome flare-up, which prevented handgrip testing, and one for incomplete HIV qRT-PCR data. Demographic and clinical characteristics of the 80 participants are presented in Table 1. Median age for all study participants was 54 years, but 66 years for the sarcopenic subgroup. Although one-half of the participants were male, they were responsible for the majority in the presarcopenic and sarcopenic subgroups (93.8% and 75%, respectively). The study included similar proportions of white/non-Hispanic, black, and Hispanic participants. HIV risk factors were predominantly sexual transmission (either heterosexual or men who have sex with men [MSM]) rather than intravenous drug use (IVDU), and median educational level attained in all the participant subgroups was greater than 1 year of postsecondary education. The median duration of known HIV infection was over 15 years and current CD4 count over 600 cells/mm3. Nadir CD4 interquartile range was 43 to 300 cells/mm3, and one-half of the participants had a history of opportunistic infection (OI) or HIV-associated disease. Stable receipt of cART was documented in participants, and NNRTI- or PI-based regimens were predominant. Participants receiving antihypertensive, antihyperglycemic, and lipid-modifying agents were 33.8%, 32.5%, and 12.5%, respectively.
Participants Demographic and Clinical Characteristics.a

Abbreviations: OI, opportunistic infection; SMI, skeletal muscle index; MSM, men who have sex with men; IVDU, intravenous drug use; HCV, hepatitis C virus; cART, combination antiretroviral therapy; NRTI, nucleoside reverse transcriptase inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; InI, integrase inhibitor; PI, protease inhibitor.
a Continuous measures are summarized as median (interquartile range).
b Nine persons deny behavioral risk of HIV.
c Antihypertensives, lipid-modifying agents, antihyperglycemic agents, chronic obstructive airway disease, antidepressents, antivirals other than those for HIV, thyroid hormone, and bisphosphonates.
d Marijuana and/or cocaine; use unknown for one participant.
e Defined as meeting Centers for Disease Control and Prevention recommendations for exercise.
The majority of participants had normal skeletal muscle mass (normal SMI). Presarcopenia and sarcopenia were seen in 20.0% (95% CI: 12.5%-31.9%) and 5% (95% CI: 1.4%-12.3%) of the participants, respectively (Table 1). The few participants with sarcopenia were predominantly male, older, had shorter duration of known HIV infection, and had lower nadir and current CD4 cells. None of the participants with sarcopenia were coinfected with HCV. Participants with presarcopenia were also predominantly male, but age, duration of known HIV infection, and nadir and current CD4 cells were similar to participants with normal SMI. One-quarter of the participants who demonstrated presarcopenia had HCV coinfection.
We analyzed the data set for relationships between skeletal muscle mass and age, height, weight, handgrip strength, or GS in the 80 participants (Figure 1A-D and Table 2). In both male and female participants, skeletal muscle mass was lower with increasing age (male ρ = −.273, P < .0478; female ρ = −.414, P < .0317). Skeletal muscle mass was greater with increasing height or total body weight (height, male ρ = .401, P < .0029, female ρ = .595, P < .0011; weight, male ρ = .493, P < .0002, female ρ = .629, P < .0004). In males, greater handgrip strength was observed in those with correspondently greater skeletal muscle mass (ρ = .632, P < .0001; Table 2). In females, although greater handgrip strength was observed in those with greater skeletal muscle mass, it only trended toward statistical significance (ρ = .370, P = .0575). There was no statistically significant correlation between skeletal muscle mass and GS in either male or female participants (male, ρ = .065, P = .642; female ρ = −.0327, P = .871).

Correlation between skeletal muscle and age, height, body weight, and handgrip strength. In females, there was a statistically significant negative correlation between skeletal muscle and age (a) and statistically significant positive correlations between skeletal muscle and height (b) and weight (c). In males, there was a statistically significant negative correlation between skeletal muscle and age (a) and statistically significant positive correlations between skeletal muscle and height (b), body weight (c), and handgrip strength (d).
Participant’s Anthropomorphic, Strength, and Performance Characteristics.a
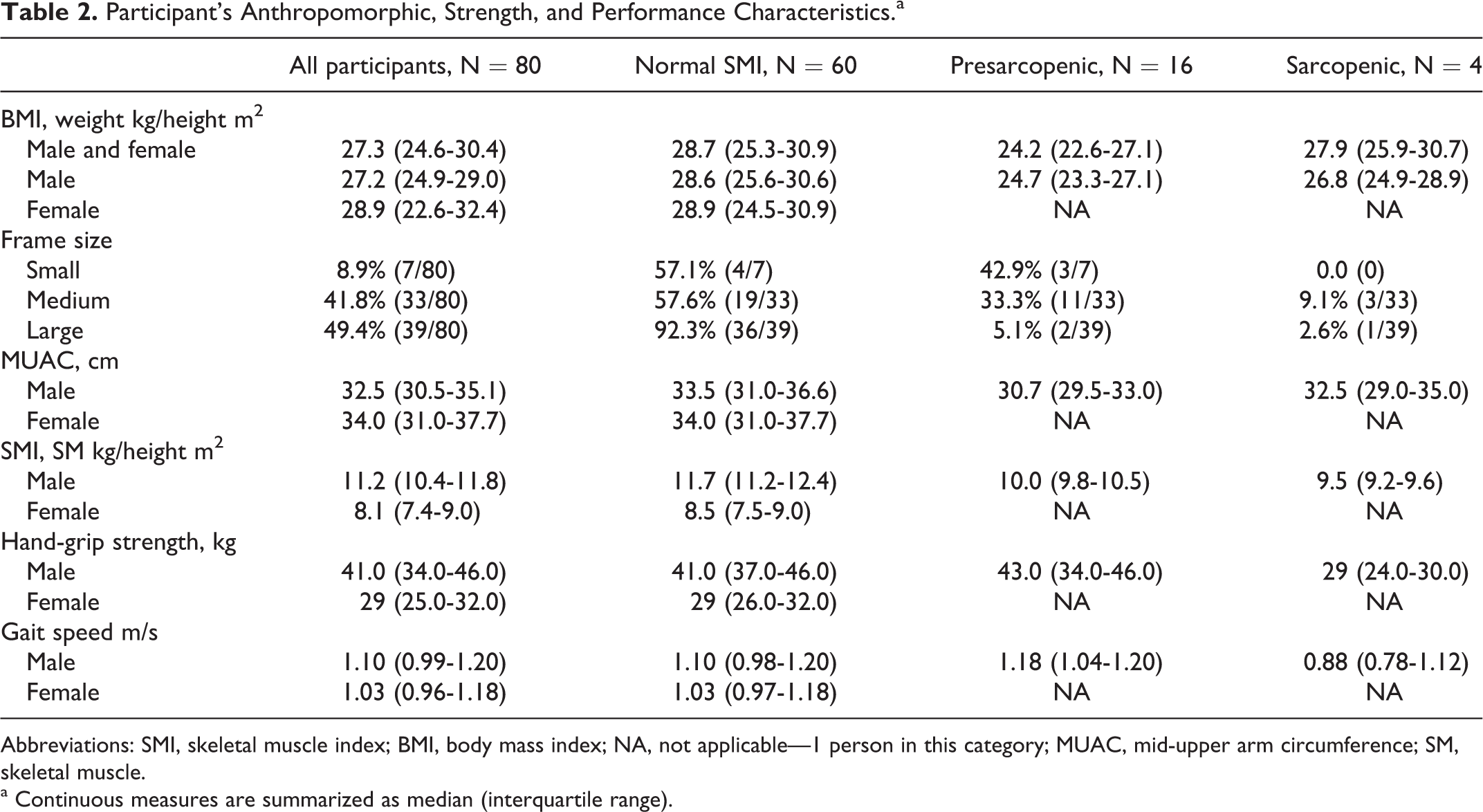
Abbreviations: SMI, skeletal muscle index; BMI, body mass index; NA, not applicable—1 person in this category; MUAC, mid-upper arm circumference; SM, skeletal muscle.
a Continuous measures are summarized as median (interquartile range).
Participants anthropometric, strength, and performance characteristics are presented in Table 2. Participants with sarcopenia were older (median age 66 years) and had lower median CD4 counts (437 cells/mm3) compared to the other groups (Table 1). Their BMI was similar to participants with normal SMI. Of the 4 participants, 3 had a history of OI and/or HIV-associated disease. All had nadir CD4 count <200 cells/mm3. Handgrip strength in all 4 participants with sarcopenia was low and markedly below that of participants with normal SMI and presarcopenia. Interestingly, handgrip strength in participants with presarcopenia was similar to that of participants with normal SMI and to community-dwelling adults without known HIV infection documented in published reports. 26,27 Only 1 of the 4 participants demonstrated low GS of 0.54 m/s and MUAC <25th percentile (data not shown). The median number of medications for comorbidities in participants with sarcopenia was higher than that of participants with normal SMI and those demonstrating presarcopenia, but the difference was not statistically significant.
Participant parameters associated with presarcopenia after age-adjusted linear regression analysis are shown in Table 3. A greater percentage of male compared to female participants demonstrated presarcopenia (28.3 versus 3.7%; odds ratio [OR] 10.7, 95% CI 1.33-86.55, P < .026). Greater BMI or MUAC was negatively associated with presarcopenia. With each unit increase in BMI, the odds of presarcopenia decreased by 20% (OR 0.80, 95% CI 0.67-0.94, P < .007). Similarly, the odds of presarcopenia decreased by 17% with each centimeter increase in MUAC (OR 0.83, 95% CI 0.70-0.98, P < .01). Large skeletal frame compared to small and medium skeletal frame was also negatively associated with presarcopenia (OR 0.09, 95% CI 0.02-0.42, P< .003).
Age-Adjusted Linear Regression Analysis Odds Ratios for Presarcopenia.

Abbreviations: MSM, men who have sex with men; IVDU, intravenous drug use; OI, opportunistic infection; NNRTI, non-nucleoside reverse transcriptase inhibitor; BMI, body mass index; MUAC, mid-upper arm circumference; OR, odds ratio; CI, confidence interval.
The odds of identifying presarcopenia were greater in participants whose mode of HIV acquisition was IVDU than in participants who reported acquisition via heterosexual sex (OR 6.9, 95% CI 1.17-41.0, P < .033). Greater odds for the identification of presarcopenia in participants in MSM transmission category trended toward statistical significance (OR 4.2, 95% CI 0.98-17.85, P < .053). No participant reported current or recent IVDU, and duration of known HIV infection in presarcopenic participants was not significantly different from that of participants with normal SMI (P = .379).
The odds of identifying presarcopenia were greater in participants who reported current recreational psychoactive substance use other than tobacco or alcohol (ie, marijuana and/or cocaine) compared to participants who denied use (OR 5.1, 95% CI: 1.11-23.78, P < .037). History of OI, current receipt of a NNRTI, or lipid-modifying agent trended toward association with presarcopenia (history OI/HIV associated disease OR 2.9, 95% CI: 0.89-9.42, P < .077; NNRTI, OR 2.75, 95% CI 0.86-8.78, P < .088; and lipid-modifying agent, OR 2.93, 95% CI 0.85-10.03, P < .088).
We evaluated the predictive value of participant MUAC percentile for presarcopenia or sarcopenia. Finding that participants did not have a MUAC below the 25th percentile on examination had a 90.4% negative predictive value for presarcopenia. Demonstration of a MUAC below the 25th percentile by participants had a positive predictive value for presarcopenia of 44.0%, sensitivity 68.8%, and specificity 78.3%.
Although, one-third of the participants were receiving an antihypertensive agent and 12.5% a glucose-lowering agent, neither pharmaceutical category was associated with presarcopenia in our analysis. No participant reported decreased overall food or protein intake, anorexia, or change in their sense of taste or smell or food insecurity.
Discussion
In this preliminary study, we document a high prevalence of presarcopenia in community-dwelling participants with durably suppressed HIV infection and CD4 cell reconstitution. The gender-specific cutoff points for low skeletal muscle mass were based on levels associated with physical disability risk and not merely the central range of a young reference group. 22,28,29 The odds of identifying presarcopenia were 10.7-fold greater in males compared to females in spite of the absence of clinically significant differences between male and female duration of known HIV infection.
Our finding of an association with male gender is consistent with a cohort study demonstrating lower skeletal muscle mass in midlife HIV-infected males, but not females, compared to HIV sero-negative controls. 30 Investigators had previously proposed that the greater adipose tissue depot inherent in female gender may attenuate skeletal muscle mass loss in AIDS wasting disease, and this may be so for age-related loss as well. 31–33 In the general population, females demonstrate skeletal muscle mass stability until later age than males. 6 Our data on skeletal muscle mass in predominantly midlife participants is consistent with predisposition in males for earlier manifestation of presarcopenia. This suggests that an opportunity may exist in this population for secondary intervention. However, this requires further confirmation in longitudinal studies.
The prevalence of presarcopenia documented in this predominantly midlife group was similar to that found in community-dwelling older persons (<70 years) without known HIV infection. 34,35 However, a longitudinal investigation of skeletal muscle mass loss in a community-dwelling cohort of HIV-infected participants and sero-negative controls did not find greater velocity of skeletal muscle mass loss in persons of either gender with HIV infection. 30
None of the participants reported current or recent IVDU or was receiving methadone maintenance therapy. However, the association of presarcopenia risk with IVDU transmission category may be a durable effect of IVDU on skeletal muscle mass maintenance and/or accrual. The increased odds for presarcopenia documented in participants reporting current recreational psychoactive substance use further suggests a negative effect of substance use (regardless of pharmacologic class or route of administration) on skeletal muscle. This is an area for further investigation.
Higher BMI, MUAC, and large skeletal frame were negatively associated with presarcopenia in our data set. This was consistent with the parallel increase in skeletal muscle mass with height and body weight seen in all 80 participants as a group. We evaluated for skeletal frame size (height-to-wrist circumference ratio), which is rarely appreciated in clinical care or research and may be more informative than height alone in risk assessment. Greater weight and larger bones requiring more force production for movement may attenuate age-related skeletal muscle loss. Use of SMI to normalize skeletal muscle for height thereby eliminating bias for greater stature is a strong point in the EWGSOP criteria. Accordingly, our prevalence rates were compared to those of investigations in which skeletal muscle was also normalized for height. 34,35
Increased odds for presarcopenia in association with MSM transmission category and current receipt of lipid-lowering medication trended toward statistical significance. This is consistent with the literature documenting lower likelihood of overweight in MSM but higher likelihood of metabolic risk factors for chronic disease. 36 A greater percentage of patients receiving NNRTI- compared to PI-based ART demonstrated presarcopenia and the increased odds trended toward statistical significance. This finding is not consistent with a cohort study in HIV-infected men that documented an association of efavirenz with skeletal muscle gain (or attenuation of loss). 30 Sample size limited our investigation of this trend, and this will likely be an area of future research.
Low skeletal muscle mass is central to the definition of sarcopenia. Our finding of a 90.4% negative predictive value for participants demonstrating MUAC within or above the 25th percentile suggests use of this parameter effectively identifies those patients least likely to demonstrate skeletal muscle deficit. These data (MUAC) may decrease unnecessary testing. However, the poor positive predictive value, with limited specificity and sensitivity, indicates additional need for evaluation by bioelectrical impedance analysis or dual energy X-ray absorptiometry for the diagnosis of presarcopenia or sarcopenia. In addition, low limb muscle mass has been associated with low bone mineral density in HIV-infected males, and males in the general population. 17,37 Patients with a MUAC below the 25th percentile under evaluation for presarcopenia/sarcopenia may also be candidates for evaluation of bone mineral density.
Measurement of MUAC can be performed by most clinicians, requiring minimal patient disrobing, no special equipment (apart from an inelastic tape measure), or complex mathematical operations for use of the result. It is less susceptible to distortion by fluid retention than lower extremity circumferences and is largely independent of height. 38 However, it may not be an appropriate parameter in those with lymphedema, anasarca, or extreme obesity.
The 4 participants with sarcopenia demonstrated low handgrip strength in addition to low skeletal muscle mass and were older than participants with presarcopenia or normal SMI. In longitudinal studies, lower midlife handgrip strength and greater rate of strength decline over time were predictive of future physical disability. 29,39 We believe this underscores the need for clinical evaluation of muscle strength, in addition to muscle mass, in the care of midlife and older HIV-infected patients.
Our study was limited by sample size and its cross-sectional design. We identified associations but could not infer temporal relationships or causation. In addition, our study was not powered to detect associations with specific cART regimens. Denervation has been linked to age-related skeletal muscle change and investigation of whether a history of exposure to potentially neurotoxic antiretrovirals (ie, the thymidine analogs or didanosine) is associated with presarcopenia in patients with HIV infection is an area for future work. 40 There was only 1 female in the presarcopenia group and this prevented use of gender as a covariate in logistic regression models. Participants were privately insured or receiving Medicare with supplemental private insurance and had a median 2 years postsecondary education. This suggests that they may have achieved better quality of life with fewer health damaging exposures, such as food insecurity and/or unstable housing, than many HIV-infected patients in the United States, where HIV infection is strongly associated with urban poverty. 41 Evaluation for skeletal muscle deficit in persons with access to antiretroviral medications whose ability to act in their own interest is nevertheless constrained is an area for future investigation.
In conclusion, in the present study of HIV-infected persons 45 years or older with durably suppressed HIV infection, we found highly prevalent presarcopenia in midlife in association with male gender. Male gender, IVDU transmission category, or current recreational psychoactive substance use markedly increased odds for the identification of presarcopenia in participants. Our data set suggests that MUAC percentile may improve patient selection for evaluation of skeletal muscle mass and function in clinical care. Longitudinal investigations are needed to determine the impact of low midlife skeletal muscle mass on disability rates in aging HIV-infected persons in care and appropriate preventive interventions.
Footnotes
Acknowledgments
The authors thank Joanna Kopacz for assistance with data management and all participants without whom this work would not have been possible.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
