Abstract
Seven rapid diagnosis tests (RDTs) of HIV were evaluated by a panel group who collected serum samples from patients in Abidjan (HIV-1 = 203, HIV-2 = 25, HIV-dual = 25, HIV = 305). Kit performances were recorded after the reference techniques (enzyme-linked immunosorbent assay). The following RDTs showed a sensitivity of 100% and a specificity higher than 99%: Determine, Oraquick, SD Bioline, BCP, and Stat-Pak. These kits were used to establish infection screening strategies. The combination with 2 or 3 of these tests in series or parallel algorithms showed that series combinations with 2 tests (Oraquick and Bioline) and 3 tests (Determine, BCP, and Stat-Pak) gave the best performances (sensitivity, specificity, positive predictive value, and negative predictive value of 100%). However, the combination with 2 tests appeared to be more onerous than the combination with 3 tests. The combination with Determine, BCP, and Stat-Pak tests serving as a tiebreaker could be an alternative to the HIV/AIDS serological screening in Abidjan.
Keywords
Introdution
Infection by HIV is a worldwide public health issue, particularly in the high prevalence areas. According to the World Health Organization (WHO), 33.3 million people were infected by HIV by the end of 2009. In the same year, around 2.6 million new infections were recorded, and 1.8 million deaths were related to HIV/AIDS. Of the global population living with HIV, 68% of the infected persons are in sub-Saharan Africa; and among them 10.6 million people need treatment with antiretroviral (ARV) medications. 1 With a prevalence of 3.4%, Ivory Coast is the country most affected with HIV in the West African serological block. 2
The strategy to fight against HIV/AIDS is based on a number of actions, among which the biological screening of the infection is of great importance. Indeed, biological diagnosis is the main starting point of prevention efforts and, more than anything else, in the initiation of care and treatment efforts. In practice, diagnosis of HIV infection in adults is based on the presence of the specific antibodies. The “conventional strategy” generally includes screening of specific antibodies using an immunoenzymatic technique, such as enzyme-linked immunosorbent asssay (ELISA), followed by the confirmation of a positive result by using a specific test like Western blot. The conventional strategy cannot always be carried out in developing countries because it requires adapted laboratory equipment, and a qualified staff and it is costly. The development of very sensitive and specific commercial reagents for screening and confirmation helped promote new strategies for rapid diagnosis tests (RDTs) for detection of HIV infection. 3 Therefore, in Africa, where HIV-1 and/or HIV-2 are endemic, these “alternative strategies” are simpler and less costly than the conventional ones and are recommended by the WHO. 4
In Africa, extensive genetic diversity and the existence of abnormal variants were observed. 5 In these conditions, the performances of RDTs differ from those observed in a reference laboratory. 6 –9 Thus, in order to make these analysis strategies efficient, the tests used should be very sensitive and specific to the patients of each country. Commercial RDTs must always be evaluated in a given geographic area, with locally collected serum samples, in order to determine the sensitivity and specificity values in the field. For these reasons, the evaluation of an alternative screening strategy must always be conducted in order to take into account the multiple factors (variability of virus strains, local seroprevalence, local operational factors, laboratory qualification) that are not always predictable and that are likely to affect the results. 10 –14
The aim of this study is to evaluate the performance of commercial RDTs that are available in the Ivory Coast. The study was conducted in particular to find an alternative strategy in order to ensure individual diagnoses of HIV infection as well as result notifications to patients with the best possible reliability at a lower cost.
Materials and Methods
The evaluation concerned a sample group of serum collected from the serum bank of RETROCI project and kept at 80°C. The sample group of serum from Ivorian patients was made up of 508 samples of which 203 contained antibodies against HIV and 305 did not contain antibodies. The HIV-positive samples were categorized as 154 (HIV-1), 25 (HIV-2), and 25 (HIV Dual).
During this study, 7 RDTs were evaluated: SD Bioline HIV 1/23.0 (Standard Diagnostic, Korea), Precise HIV (Ranbaxy, India), BCP HIV-1/-2 (Bio-Tech, Ben CosPham, Korea), Determine (Abbott, Abbott Park, IL), Oraquick (Orasure, Bethlehem, PA), Visiteck (Omega Diagnostic, United Kingdom), HIV 1/2 Stat-Pak (Chembio, Medford, NY). All these RDTs used the immunochromatographic principle. The technical characteristics of RDT as described by the manufacturers are reported in Table 1.
Operational Characteristics of HIV Infection Serological Screening Test according to Data from the Manufacturer.a
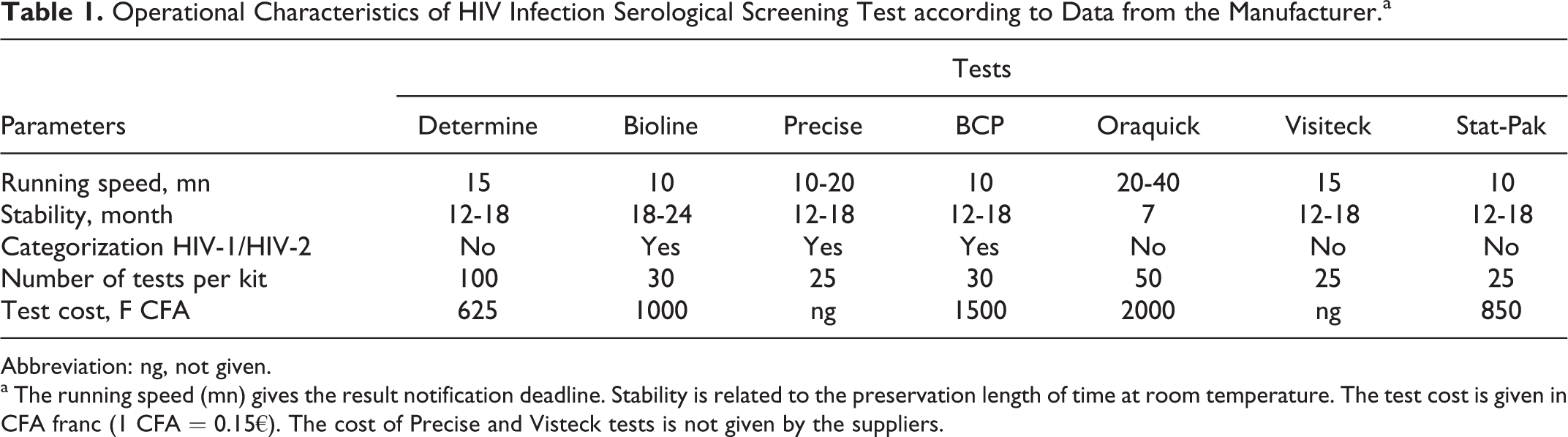
Abbreviation: ng, not given
a The running speed (mn) gives the result notification deadline. Stability is related to the preservation length of time at room temperature. The test cost is given in CFA franc (1 CFA = 0.15€). The cost of Precise and Visteck tests is not given by the suppliers.
From the thawed serum samples, the assays were carried out according to the recommendations of the manufacturer. Every single rapid test was independently observed by visual reading by 3 people. The final result was the one associating at least 2 identical responses of 3 (positive, negative, or uncertain in the case of lack of positive signal sharpness).
In a first approach, the results obtained with the 7 RDTs were compared to the results obtained according to the reference algorithm in order to calculate the sensitivity and the specificity of each kit. The more efficient tests were reported on the basis of sensitivity (100%) and specificity (>99%)
In a second approach, alternative strategies were evaluated using the chosen kits. The performance of different series or parallel combinations of the chosen kits were tested. The different possible associations of diagnosis were studied on the basis of 4 various strategies. Strategy A combines 2 screening tests in a series without confirmation of discordant serum and corresponds to WHO strategy II. 4 Strategy B combines 2 screening tests in a series with confirmation of discordant serum by a third screening test, which corresponds to WHO strategy III. 4 Strategy C combines 2 screening tests in parallel without confirmation of serum samples that are not doubly negative or positive. Finally, strategy D combines 2 screening tests in parallel with confirmation of serum samples that are not doubly negative or positive by a third screening test.
The sensitivity (true positive/true positive + false negative), and the specificity (true negative/true negative + false positive), the positive predictive values (PPVs; true positive/true positive + false positive), and the negative predictive values (NPVs; true negative/true negative + false negative) were calculated for each screening test and for each association from the contingency table with ELISA test of the RETROCI project algorithm as a reference basis. As regards the calculations, an undermined result was considered a true positive when the serum was positive with reference technique; an undermined result was considered a false positive if the serum was negative with reference techniques. The average cost of a screening test was calculated on the basis of the unit cost per test and for each association. Epi Info program version 6.7 was used for analysis.
Results
Of the 203 samples detected positive by the reference method, Determine, Oraquick, Bioline, BCP, and Stat-Pak tests highlighted the presence of anti-HIV antibodies in 203 samples; demonstrating a sensitivity of 100%. Precise and Visiteck tests detected 202 of the 203 positive samples, which equates to a sensitivity of 99% (Table 2).
Performances of the HIV Rapid Diagnostic Assays.a

Abbreviations: CI, confidence interval, enzyme-linked immunosorbent assay; FN, false negative; FP, false positive; NPV, negative predictive value; PPV, positive predictive value; TN, true negative; TP, true positive, Se, sensitivity, Sp, specificity.
a From the contingency table with ELISA test of the RETRO-CI project algorithm as reference basis, serum samples were categorized under TP, FP, TN, and FN. The Se, the Sp, the PPV, and the NPV were calculated for each screening test from the contingency table, values in parenthesis are CI 95%.
Oraquick and Bioline tests highlighted the absence of anti-HIV antibodies on all the 305 samples without anti-HIV antibodies detected by reference technique, that is, a specificity of 100%. The reported specificity rate for the other tests were, respectively, 99.67% (BCP, Stat-Pak), 99.37% (Precise), and 99.34% (Determine; Table 2).
The criteria for inclusion of an individual rapid HIV assay into the National algorithm were initial sensitivity of 100% and initial specificity of ≥99%. Determine, Oraquick, Bioline, BCP, and Stat-Pak kits demonstrated a sensitivity of 100% and a specificity of 99% and were kept for further study. The results of the evaluation of 4 diagnosis strategies with different configurations of the 5 previously considered are reported in Table 3. Associated in series, Oraquick-Bioline and Oraquick-BCP, gave sensitivities and specificities of 100%. On the other hand, in parallel combination, only the association Oraquick-BCP gave levels of 100% performance. The combinations of the 3 tests, whatever the type of strategy, gave sensitivities and specificities of 100%.
Performances of Different Strategies for HIV Infection Serological Screening.a

Abbreviations: ELISA, enzyme-linked immunosorbent assay; FN, false negative; FP, false positive; NPV, negative predictive value; PPV, positive predictive value; TN, true negative; TP, true positive, Se, sensitivity, Sp, specificity.
a From the contingency table with ELISA test of the RETRO-CI project algorithm as reference basis, serum samples were categorized under TP, FP, TN, and FP. The Se, the Sp, the PPV, and the NPV were calculated for each screening test from the contingency table.
Discussion
This study was conducted on serum from black individuals living in Abidjan in order to take into account the variability of HIV strains in a given territory, especially in Africa. It aimed at evaluating the performance of each commercial kit in order to put forward an algorithm for HIV-infection diagnosis in Abidjan.
According to the WHO, a test evaluation for HIV diagnosis must contain a minimum of 200 positive samples and 200 negative samples so as to provide a confidence interval of 95% ± 2% for the estimated sensitivity and specificity. This study used 203 positive samples and 305 negative samples, which helps to validate all the reported results. 5 Determine, Oraquick, Bioline, BCP, and Stat-Pak tests showed a sensitivity of 100%. Identical rates were cited by several authors. 7,8,13,14,15,16 On the other hand, other studies presented sensitivity levels under those reported in the study. 6,9,17
As far as specificity is concerned, only Oraquick and Bioline tests obtained 100%. The reported values are under those cited in the literature. 6,16,18 The Determine test used in the algorithm of HIV screening in Ivory Coast showed a specificity of 99.34%. Variations in performance of rapid HIV assays in different settings, especially with different levels of antigenic challenge, are well known. In addition, differences in the performance may relate in part to their inability to detect low antibody titers in people taking antiretroviral therapy with low viral loads. However, in the present study, no specific information was solicited regarding the use of ARV drugs, making it difficult to explain the lowest performance of some of the rapid HIV assays evaluated. The lack of specificity of serological tests in relation to serum from African patients was described using several kits available in the market. 6,10,12,13 It appears at first sight that using only 1 screening test (WHO Strategy I) is not conducive to the individual diagnosis. The divergences between the results reported in the study and those cited by various authors make it necessary to validate the available tests in a given context before routine use for HIV infection screening.
The promotion of strategies said to be alternatives for serological diagnosis of HIV infection aims to reduce resorting to a confirmation test (Western Blot), while working so that these strategies remain reliable. Four alternative strategies were explored, 2 tests in series/parallel and 3 in series/parallel. Altogether 20 combinations were tested. Whatever the type of strategy, sensitivity and NPV calculated with aid of the Ivorian panel were 100%. However, strategies that associated tests in parallel often lack specificity. They were always onerous, for they were based on tests that were simultaneously conducted and not in a sequential way, which increases the number of the tests to be carried out.
Among the associations of WHO strategy II (association of 2 tests in series), Oraquick-Bioline and Oraquick-BCP combinations showed better performances (specificity, sensitivity, PPV, and NPV of 100%). Moreover, they were rapid, inexpensive, easy to perform, and have results that are easy to interpret. In addition to these characteristics, the test should be able to be stored at room temperature with a long shelf life and should require no additional equipment or ancillary supplies to be performed (Table 1) according to WHO recommendations of ideal tests. 19 These 2 combinations helped to diagnose a case of HIV infection in the Ivorian context, with the minimum certainty. The other combinations lacked specificity. These results reveal the necessity of systematically using a strategy that helps to confirm the positivity of a first screening test. With a cost of 3000 French African Community Francs (FCFA), the combination Oraquick and Bioline had the advantage of being the least onerous. As regards WHO strategy III, Determine combination as first test, BCP as second test for confirmation, and Stat-Pak as tiebreaker was the best configuration, since it associated an excellent reliability (sensitivity, specificity, PPV, and NPV of 100%) and was less expensive. Altogether, Oraquick-Bioline combination (WHO strategy II) and Determine-BCP-Stat-Pak (WHO strategy III) showed the best performances. However, with a cost of 3000 FCFA, the combination associating 2 tests (Oraquick, Bioline) appeared to be more onerous than the combination of 3 tests (Determine, BCP, Stat-Pak) for a cost of 2475 FCFA.
The validation of kits in relation to a sample group of serum from Ivorian patients helped to keep 5 screening kits of the 7 tested ones and search out 2 configurations that favored a reliable diagnosis of HIV infection in an Ivorian context. If the laboratory equipment is sufficient (central laboratory, a specialized institute laboratory) to the use of 3 kits, Determine (screening), BCP (confirmation), and Stat-Pak (categorization) is the best kit configuration in terms of reliability and cost. If the laboratory equipment is less sufficient (laboratories of general hospitals, health services), the use of 2 rapid tests, that is, to say Oraquick (screening) and Bioline (confirmation), ensures a reliable screening as well but has major drawbacks for being more expensive than the strategy combining the 3 tests.
Conclusion
HIV infection screening tests, thanks to rapid techniques, are proving to be reliable enough for individual notification of results in cases of voluntary HIV infection screening among asymptomatic individuals in the Ivory Coast. This involves resorting to at least 2 kits: the first for screening and the second for confirmation. Series combination of 3 tests, Determine, BCP, and Stat-Pak, proves to be the most efficient in terms of both the technique and cost.
Footnotes
Acknowledgments
We thank the Reto-CI Project of Abidjan and the staff in all departments of the National Laboratory of Public Health (NLPH, Abidjan), particularly Dr Adje (Retro-CI). The views expressed are those of the authors and should not be construed to represent the positions of the Retro-CI Project and the LNPH.
Authors’ Note
Loukou and Zinzendorf designed the study. Cabran, Lathro, Adouko, and Agbessi were responsible for HIV testing on serum. Zinzendorf and Adouko analyzed the data. Zinzendorf and Cabran wrote the draft paper. All other authors have been involved in critical revision of the article. All authors have given final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Laboratory of Public Health.
