Abstract
Patients with newly diagnosed multiple myeloma (MM) may present with profound functional impairment due to high disease burden, and an Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 4 is often viewed as a barrier to intensive therapy despite potential reversibility. We report six newly diagnosed, bedbound MM patients (ECOG 4) treated with individualized multi-agent regimens incorporating proteasome inhibitors, immunomodulatory agents, and anti-CD38 monoclonal antibodies when feasible. Advanced features included extensive skeletal disease, renal dysfunction, hyperviscosity, and high-risk cytogenetics. All patients demonstrated rapid biochemical response, with a median M-protein reduction of approximately 75%; five of six achieved a very good partial response or better, including one complete response. Functional recovery paralleled depth of response, with a median time to meaningful improvement of 10 weeks. All patients improved to ECOG 2 or better, regaining ambulation and independence in activities of daily living. One patient proceeded to autologous stem cell transplantation and another completed stem cell mobilization. No irreversible treatment-limiting toxicities were observed. These findings suggest that severe functional impairment at presentation often reflects reversible disease burden and that aggressive, individualized therapy can result in substantial clinical and functional recovery in patients initially considered too debilitated for intensive treatment.
Introduction
MM is a plasma cell malignancy characterized by clonal proliferation in the bone marrow and overproduction of monoclonal immunoglobulins. 1 The disease commonly leads to osteolytic bone lesions, pathological fractures, anemia, renal dysfunction, and other organ damage. 2 Bone involvement is present in the majority of patients and is a major cause of pain, immobility, and diminished quality of life in MM.3,4 Historically, patients with advanced myeloma and significant skeletal disease often became bedridden or wheelchair-bound. However, the advent of highly effective combination therapies, including proteasome inhibitors, immunomodulatory drugs, and anti-CD38 monoclonal antibodies, have markedly improved response rates and survival in MM.5-7 Deep responses such as complete response (CR) or stringent CR (sCR) are now achievable in the majority of newly diagnosed patients with aggressive combination regimens, and attaining these responses correlates with prolonged progression-free survival. 8 Early initiation of high-efficacy therapy is particularly critical in symptomatic myeloma patients; if treatment is delayed, complications like spinal cord compression can cause permanent neurological damages. 9 Conversely, prompt cytoreduction can relieve tumor-related pain and neurological impairment, offering a chance to reverse functional disability even in frail or immobile patients. 10
Importantly, tolerating intensive therapy is feasible for many debilitated or older patients, and can lead not only to disease control but also to improved quality of life and physical function. 11 Recent evidence shows that frail myeloma patients experience significant gains in physical functioning, pain reduction, and overall health status during active treatment. 11 In fact, effective therapy can transform patients’ daily capabilities; as patients achieve deeper remission and longer survival, they often wish to resume normal activities and even exercise regimens. 12 Here we present a retrospective case series of 6 MM patients who were severely disabled (bedbound or non-ambulatory) at presentation and who, through early and aggressive intervention, achieved remarkable recovery of mobility and function. This series underscores how modern multi-agent and combination therapy, initiated early and tailored as needed, can reverse profound disability in MM, allowing patients to regain independence and reintegrate into society. We highlight both the clinical and social outcomes of these cases and discuss the implications for managing frail myeloma patients.
Methods
We retrospectively identified six patients treated at our academic institution who presented with MM and ECOG performance status 4 due to myeloma-related bone lesions and pathological vertebral fractures. Baseline demographic and disease characteristics (including age, sex, International Staging System stage (ISS), cytogenetic risk, and myeloma subtype) were collected from the medical record. Each patient underwent frontline therapy with contemporary intensive regimens initiated promptly after diagnosis. Treatments were selected based on disease risk status and patient fitness, following standard myeloma protocols and clinical judgment. All patients received combination regimens incorporating a corticosteroid such as dexamethasone plus at least two novel agents, namely a proteasome inhibitor, immunomodulator, and/or monoclonal antibody, with adjustments as needed for response or tolerability. Supportive measures such as analgesia, bone protective therapy (bisphosphonate), physical therapy, and assistive devices were provided in parallel to maximize rehabilitation.
Disease responses were assessed according to International Myeloma Working Group (IMWG) criteria, 13 including serum/urine monoclonal protein (M-protein) measurements, serum free light chain levels, and bone marrow evaluations as appropriate. Key response milestones, such as partial response, VGPR (very good partial response), CR, and sCR were defined per IMWG standard definitions. 13 Functional improvement was defined clinically as the ability to ambulate (with or without assistance) or perform independent transfers (e.g., from bed to chair) after having previously been unable to do so. The time from therapy initiation to documented functional improvement was recorded for each case. Descriptive statistics, including median and range, were used to summarize the time to improvement and extent of M-protein reduction across the cohort.
Cases
Patient 1
A 55-year-old woman with standard-risk light chain MM international staging system (ISS stage II) presented with severe back and pelvic pain and a CT of the abdomen and pelvis revealing a large sacral alar mass compressing pelvic nerves (Figure 1). She was essentially bedbound, unable to sit upright or lay on her back, at diagnosis and required a gurney for transportation. Baseline labs showed elevated free lambda light chains with minimal intact M-protein, anemia (hemoglobin 8 g/dL), and normal renal function. Cytogenetic testing showed no high-risk abnormalities. She promptly began induction therapy with daratumumab, bortezomib, lenalidomide, and dexamethasone (Dara-VRD). After two cycles, a partial response was attained, but a persistent sacral tumor prompted intensification of therapy to daratumumab, carfilzomib, lenalidomide, and dexamethasone (Dara-KRD). Contrast-enhanced computed tomography (CT) of the abdomen and pelvis at initial diagnosis demonstrating a large destructive soft tissue mass centered in the left sacral ala
This regimen achieved a deep response. By the end of induction, she reached a stringent complete response (sCR) with negative immunofixation and normalized free light chain ratio. The patient’s functional gains paralleled her tumor response. Within approximately 8 weeks of treatment, her pain had markedly decreased and she progressed from being bedbound to standing and taking steps with assistance. She proceeded to ASCT after achieving sCR. Post-transplant, she continued to improve, regaining the ability to walk with a cane and perform basic activities of daily living. At last follow-up, she remained in sCR and was ambulatory with minimal assistance.
Patient 2
A 69-year-old woman with standard-risk light chain MM (ISS stage II) initially presented with multiple vertebral compression fractures of the thoracic and lumbar spine and severe kyphosis, rendering her bedbound and required a gurney for transportation (Figure 2). Thoracolumbar spine imaging at diagnosis demonstrating multiple vertebral compression fractures involving the mid-thoracic and lumbar vertebral bodies, with marked anterior vertebral height loss and exaggerated thoracic kyphosis
She also had anemia (hemoglobin 9 g/dL) and moderate renal impairment (creatinine ∼2.0 mg/dL) at diagnosis; cytogenetics showed standard-risk features. Induction therapy with cyclophosphamide, bortezomib, and dexamethasone (CyBorD) was chosen given her age and renal function. This achieved a partial response with reduction in light chain levels and improvement of renal function. She was transitioned to lenalidomide plus dexamethasone (Rd) in order to simplify outpatient management. Over the course of therapy, her back pain and mobility gradually improved; by about 3 months in, she could transfer independently and walk short distances with a brace. However, due to persistent light chain elevation, and a plateaued response, her therapeutic regimen was switched to selinexor, pomalidomide, and dexamethasone (Sel-Pd). This regimen led to further tumor reduction and ultimately a sCR. Remarkably, alongside achieving deep remission, the patient overcame substantial socioeconomic and physical challenges. With improved health and mobility, she was able to resume educational pursuits that had been interrupted by illness as she completed her community college degree and was admitted to a University of California program. At last follow-up, she remained in sCR on maintenance therapy, fully independent in ambulation and daily activities.
Patient 3
A 32-year-old man with standard-risk IgG kappa MM (ISS stage II) presented with extensive spinal involvement, including lytic lesions in multiple vertebrae (Figure 3). He had severe back pain, lower extremity weakness, bedbound and required a gurney for transportation. Initial cytogenetics indicated standard-risk. He was started on daratumumab, bortezomib, lenalidomide, and dexamethasone (Dara-VRD). Despite some early clinical improvement, his course was complicated by non-adherence to the treatment regimen, leading to disease progression after a few cycles. He developed worsening pain and new lesions, which prompted a change in therapy. Spinal imaging demonstrating osteolytic involvement of the thoracolumbar spine at diagnosis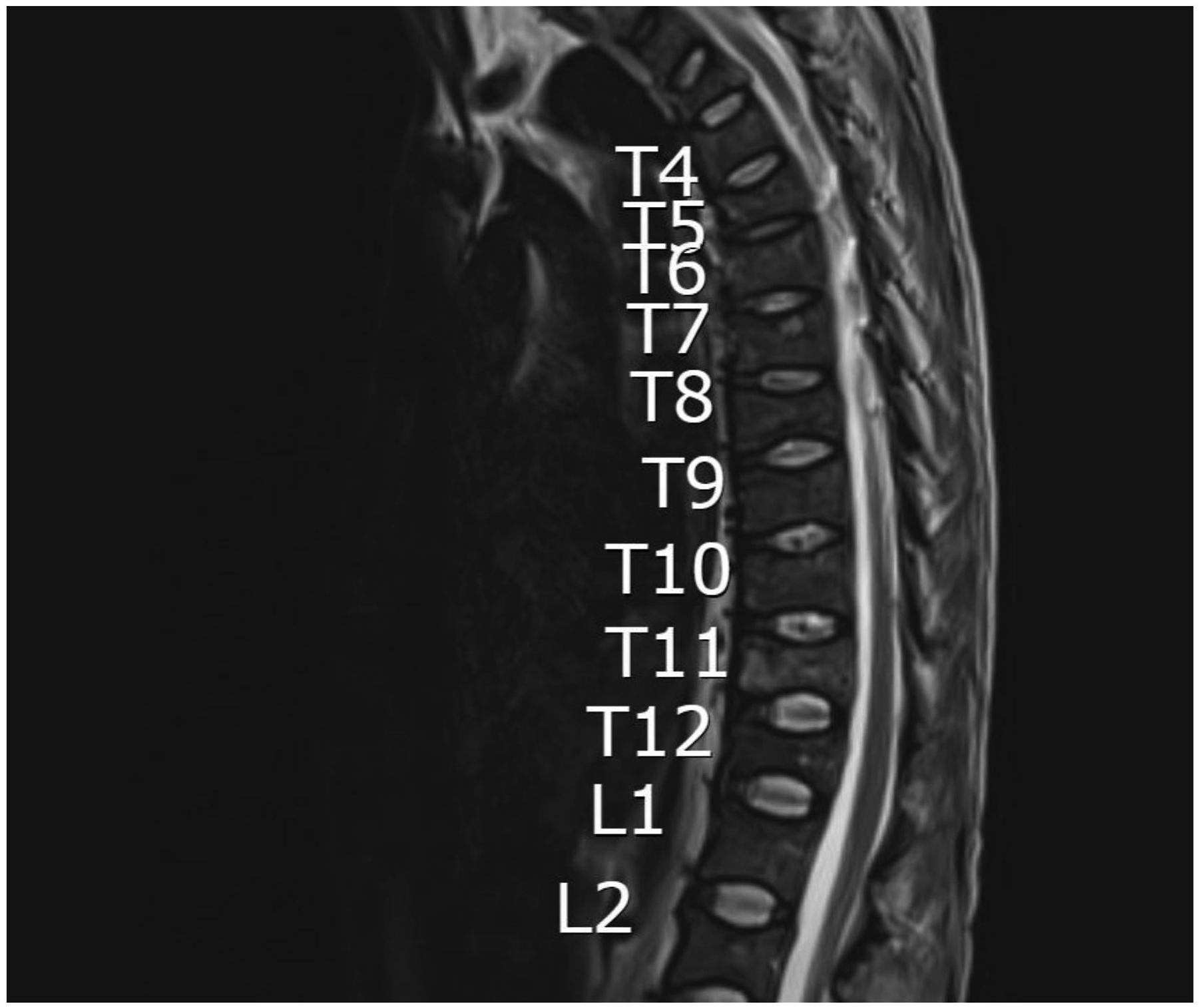
The patient was switched to an intensive salvage regimen of isatuximab, a CD38 monoclonal antibody, in combination with carfilzomib and dexamethasone (Isa-KD). Under the new regimen, he achieved a rapid tumor response, with his IgG paraprotein and bone marrow plasma cell percentage falling to near-normal levels, consistent with a VGPR. Correspondingly, his neurological symptoms and functional status improved. Over approximately 3 months of salvage therapy, the patient progressed from being wheelchair-dependent to walking with a cane. He also regained the ability to drive a car and return to many normal activities; continued to ambulate with a single-point cane for long distances, without any new skeletal events.
Patient 4
A 70-year-old man with newly diagnosed MM (ISS stage III) presented with diffuse bone pain, generalized weakness, inability to walk, bedbound and requiring a gurney for transportation. Staging revealed high-risk disease, with cytogenetic testing positive for t(4;14), t(14;16), and 1q21 gain. He also had a high tumor burden (IgA lambda monoclonal protein >4 g/dL, β2-microglobulin markedly elevated) and moderate cytopenias (hemoglobin 10 g/dL, platelets 100×109/L). Given his high-risk features and frail condition, an aggressive approach was taken. He was started on a quadruple regimen of isatuximab, bortezomib, lenalidomide, and dexamethasone (Isa-VRD), similar to the investigational IMROZ protocol for transplant-ineligible patients. 5 After four cycles, he achieved a VGPR with an approximately 90% reduction in M-protein and significant clinical improvement.
His bone pain diminished substantially, and with physical therapy and assistive devices, he progressed from bedridden status to using a cane. By around 10 weeks into therapy, he was able to ambulate over 700 feet with a rolling walker before requiring rest. Given his improved PS and good response, he was referred for ASCT. He was placed on maintenance lenalidomide with monthly isatuximab. At last follow-up, the patient remained in VGPR with no evidence of progression, and he continued to ambulate independently with a canne, greatly improving his autonomy and quality of life. Given his improved PS and good response, he was referred for ASCT and underwent successful mobilization and collection of his stem cells.
Patient 5
A 70-year-old man with newly diagnosed kappa-restricted MM (ISS stage III) presented with subacute, progressive back pain that rendered him bedbound, and required a gurney for transportation. His condition deteriorated rapidly over several weeks, culminating in hospitalization after developing hematuria and functional decline. Serum protein electrophoresis demonstrated a large M-spike (5.25 g/dL), and imaging revealed extensive myelomatous involvement: a 5.6 × 10.0 × 8.6 cm sacral mass with spinal canal encroachment (Figure 4), lytic lesions in the C5 and T1 vertebrae, and diffuse skeletal disease involving the pelvis and long bones. An adrenal mass concerning for involvement was also noted. Given the degree of marrow infiltration, the patient was unable to stand or walk and was transported to clinic by gurney. Imaging of the pelvis at diagnosis demonstrating a large, destructive soft tissue mass centered in the sacrum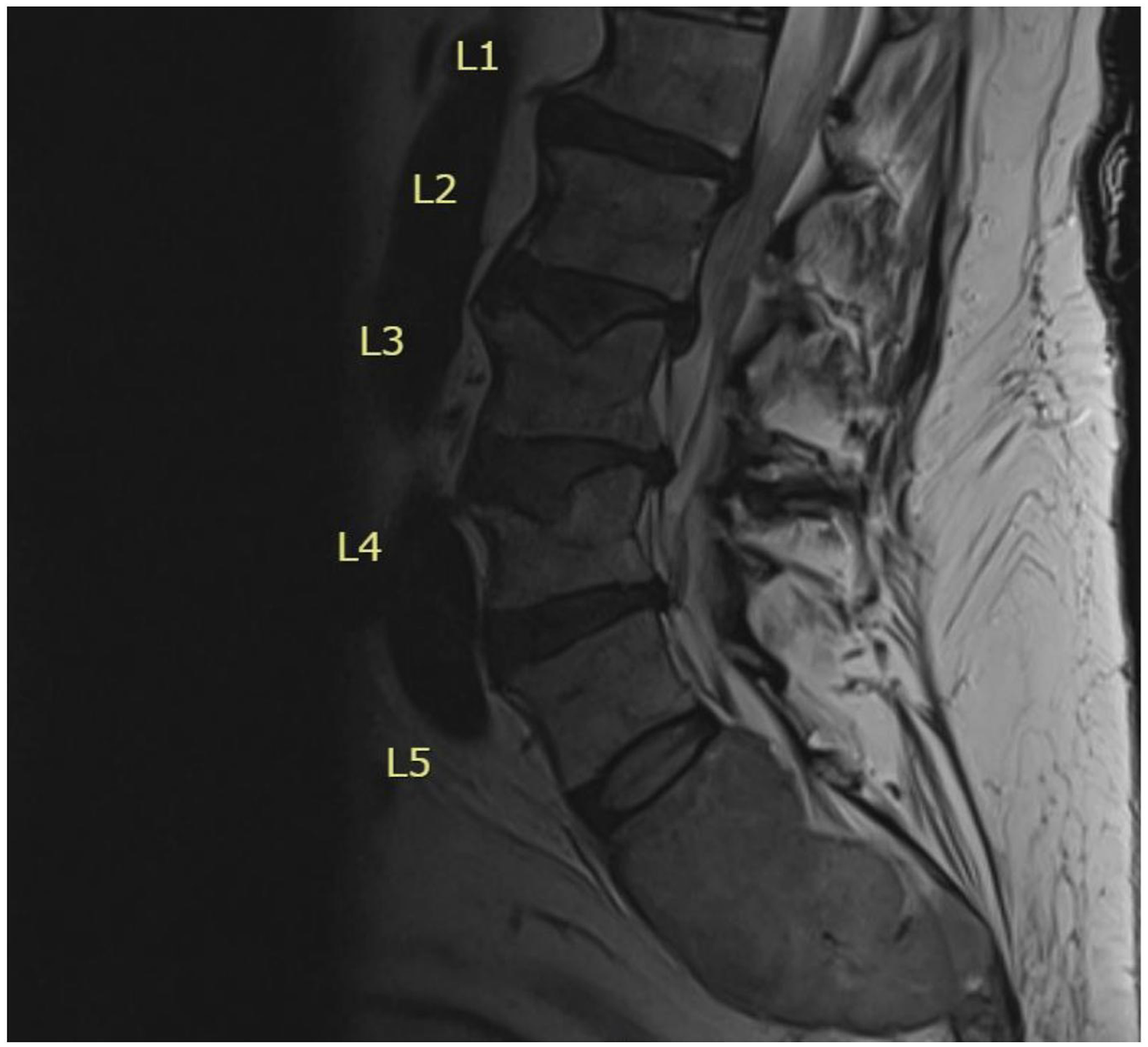
Due to his age, frailty, and the extent of skeletal disease, he was deemed ineligible for transplant and initiated on Isa-VRD. Supportive care included supplementation with calcium, magnesium, and vitamin B12, as well as planning for intravenous bisphosphonate therapy with zoledronic acid. During treatment, he experienced chemotherapy-related toxicities including constipation, peripheral neuropathy, and diarrhea. Despite these complications, the patient remained adherent and motivated. After four months, he completed three full cycles of Isa-VRD and achieved a VGPR with marked improvement in cytopenias.
Functionally, the patient showed a striking recovery. Initially non-ambulatory, he regained strength and coordination with supportive care and physical therapy. Approximately 5 months after diagnosis, he could ambulate more than 700 feet with a rolling walker. His ECOG performance status had improved from 4 to 2. At last follow-up, he remained in VGPR, independent in daily activities, and continued on Isa-VRD maintenance with close monitoring.
Patient 6
A 60-year-old man with high-risk IgA κ MM (ISS stage III) presented with acute encephalopathy, intracranial hemorrhage, pneumonia, acute hypoxic respiratory failure, which requited intubation and ICU care. Initial laboratory evaluation revealed hypercalcemia and anemia, with serum free light chain analysis demonstrating a markedly elevated κ/λ ratio >100. Bone marrow aspiration and biopsy showed >90% clonal plasma cells, confirming IgA κ multiple myeloma with gain of 1q, consistent with high-risk disease.
Neuroimaging revealed bilateral cerebral microhemorrhages with associated vasogenic edema, without evidence of mass effect. The patient required endotracheal intubation and underwent plasma exchange for hyperviscosity, resulting in neurologic improvement. He was initiated on intravenous bortezomib and dexamethasone, achieving a rapid reduction in serum free light chains from 111 to 26 mg/L, with complete resolution of encephalopathy and successful extubation.
Clinical Characteristics, Treatment Regimens, Hematologic Responses, and Functional Outcomes of Six Patients Included in This Case Series

Abbreviations: MM = multiple myeloma; ISS = International Staging System; ECOG = Eastern Cooperative Oncology Group; ASCT = autologous stem cell transplant; CR = complete response; sCR = stringent complete response; VGPR = very good partial response; Dara = daratumumab; VRD = bortezomib, lenalidomide, dexamethasone; KRD = carfilzomib, lenalidomide, dexamethasone; CyBorD = cyclophosphamide, bortezomib, dexamethasone; Rd = lenalidomide, dexamethasone; Sel-Pd = selinexor, pomalidomide, dexamethasone; Isa = isatuximab; KD = carfilzomib, dexamethasone; Vd = bortezomib, dexamethasone; BM = bone marrow.
Discussion
This case series demonstrates that selected patients with multiple myeloma (MM) presenting with severe functional impairment (ECOG 4) can achieve meaningful recovery of mobility following early and aggressive therapy. All six patients were bedbound at presentation yet regained ambulation within a median of 10 weeks, coinciding with rapid tumor cytoreduction and substantial reductions in paraprotein levels. This temporal relationship suggests that effective disease control played a central role in functional recovery, likely through alleviation of bone pain, neurologic compromise, and cytopenias. Such deep responses, including two sCRs are noteworthy, as achieving ≥VGPR is associated with improved survival and likely contributed to durable functional improvements. 8 Notably, our patients’ functional gains were sustained alongside ongoing therapy or maintenance, suggesting that quality of life improved in parallel with disease remission, rather than being compromised by treatment side effects. 11
Reversal of myeloma-related paralysis or immobility has been reported in the literature but historically was relatively uncommon. Classic reports documented that even on older chemotherapy, such as melphalan/prednisone, some patients with myeloma-induced paraplegia due to spinal cord compression could regain ambulation. 14 Our series builds on this concept with contemporary therapies: for example, Patient 1 and Patient 5, who were gurney-bound from sacral tumor involvement, recovered the ability to walk with assistance after induction therapy. This mirrors other modern observations such as a recent case of a tetraplegic myeloma patient with cervical cord compression who fully recovered neurologic function after bortezomib-based chemotherapy. 9 In that case, five cycles of VTD (bortezomib, thalidomide, dexamethasone) yielded a 98% tumor reduction and enabled complete resolution of paralysis prior to stem cell transplant. Our Patient 5 similarly achieved ∼90% tumor reduction (VGPR) with an anti-CD38–based quadruplet and went from unable to sit upright to ambulating 700+ feet with a walker. Indeed, chemotherapy and immunotherapy directly target the myeloma cells driving bone destruction and nerve compression, thereby alleviating the root cause of disability. Early intervention is key, therefore if treatment is initiated promptly, even serious neurologic deficits can improve or resolve before becoming permanent. 15
From MM management perspective, these cases also illustrate the power of novel multi-agent regimens in inducing fast and deep responses. Daratumumab- and isatuximab-containing quadruplets in the upfront setting have set new benchmarks for response. In the GRIFFIN trial, adding daratumumab to VRd yielded a post-transplant sCR rate of ∼42% vs 32% with VRd alone, and significantly prolonged progression-free survival. 8 The Phase III IMROZ study likewise showed that isatuximab plus VRd produced higher CR or better rates (75% vs 64%) and superior 5-year PFS (63% vs 45%) compared to VRd. 5 These data support using four-drug induction, particularly in patients who need a rapid maximal response, such as those with end-organ damage or severe symptoms. In our series, three patients received quadruplets, two with anti-CD38 mAbs, and all three achieved ≥VGPR with substantial early improvement. One patient (Patient 1) achieved such a deep hematologic and functional response that she was able to proceed to autologous stem cell transplantation ASCT, a goal often considered unfeasible in patients presenting with such profound disability. The one patient who started on a triplet (Patient 2 on CyBorD/Rd) required escalation to a quadruplet (with selinexor) to achieve sCR, further suggesting that more intensive regimens were key to reversing her disease and disability. Additionally, the use of second-generation novel agents in relapse, including carfilzomib, pomalidomide, selinexor, and isatuximab proved invaluable for Patients 1–3 in either managing toxicity or overcoming refractory disease. Of course, this must be done with careful patient monitoring and supportive care, as these powerful therapies can have significant side effects. In our cohort, toxicity was managed through regimen adjustments, as described, and did not preclude ultimate success.
While these results are encouraging, we acknowledge the limitations of a small case series. The improvements in mobility cannot be solely attributed to chemotherapy, as improvements may also result from supportive care and aggressive physical therapy over time. However, in our series the temporal association between therapy initiation and functional gains suggests a strong contribution from rapid tumor suppression. We did not systematically quantify quality-of-life scores or use formal mobility scales, which is a limitation; future prospective studies could evaluate functional endpoint, such as ambulatory status, frailty indices, or quality of life measures, in parallel with traditional response criteria. Additionally, long-term follow-up will be needed to determine if these functional improvements are sustained, especially if disease were to relapse. This study is also limited by inherent selection bias, as only patients who demonstrated substantial functional recovery were included. As such, the findings do not reflect the overall frequency of recovery among all patients with ECOG 3–4 MM treated during the study period. The proportion of patients who fail to achieve similar outcomes remains unknown. Therefore, these results should be interpreted illustrative rather than representative, underscoring the potential, but not the predictability, of functional recovery in this high-risk population.
Conclusion
Early and aggressive treatment of multiple myeloma, in conjunction with appropriate supportive care, can reverse profound disability in select patients. In this series, bedridden or wheelchair-bound patients achieved significant ambulatory recovery alongside deep hematologic responses. These cases underscore that restoration of mobility and improvement in quality of life are attainable goals in MM management, even for patients presenting with severe functional compromise. Clinicians should strive for timely, intensive therapy in patients with disabling myeloma, while also providing multidisciplinary support, to maximize the chance of both disease control and functional rehabilitation.
Footnotes
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for Publication
Informed consent for patient information to be published in this article was not obtained because all patient information was fully de-identified, no protected health information was included, and no individual patient can be identified from the data presented.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Prior Presentation of Abstract Statement
This abstract was previously presented in abstract form at the Lymphoma, Leukemia & Myeloma Congress, New York, New York, USA, October 13 – October 16, 2025.
