Abstract
Hereditary hemochromatosis (HH) is a genetic disorder characterized by excessive intestinal iron absorption, most associated with HFE C282Y homozygosity. In contrast, H63D homozygosity is considered a low-penetrance genotype that rarely leads to clinically significant iron overload. Bariatric surgery, particularly sleeve gastrectomy, may predispose patients to iron deficiency through reduced gastric acid and altered absorption. We report an unusual case of iron overload physiology in a patient with H63D homozygosity following sleeve gastrectomy, highlighting the diagnostic challenges posed by metabolic comorbidities and altered post-surgical physiology. A 51-year-old man with class III obesity, dyslipidemia, hypertension, vitiligo, obstructive sleep apnea, and a history of sleeve gastrectomy presented for weight management. Despite an initial 97-lb postoperative weight loss, he experienced significant weight regain along with fatigue, decreased libido, and skin darkening. Laboratory evaluation demonstrated hypogonadotropic hypogonadism, elevated serum iron (229 µg/dL), and transferrin saturation (60%) with normal ferritin (118 ng/mL). FibroScan revealed F3 fibrosis and grade 3 steatosis, consistent with advanced metabolic dysfunction-associated steatotic liver disease. Genetic testing confirmed H63D homozygosity. Pituitary MRI was unremarkable. The patient had increased alcohol intake prior to surgery, followed by a substantial reduction to moderate weekend use. The patient’s iron overload was notable given his low-penetrance genotype and prior bariatric procedure. This case illustrates the complex interplay between genetic predisposition, metabolic disease, alcohol use, and altered gastrointestinal anatomy in shaping iron indices. It emphasizes the clinical relevance of transferrin saturation and comprehensive metabolic assessment. Clinicians should maintain vigilance for atypical presentations of HH in individuals with metabolic dysfunction or prior bariatric surgery.
Keywords
Background
Hereditary hemochromatosis (HH) is an inherited disorder of iron regulation in which excessive intestinal iron absorption leads to progressive iron accumulation in parenchymal organs, such as the liver, pancreas and heart, and in joints. 1 Normally, iron reserves are closely regulated by the hormone hepcidin and its interaction with the HFE protein. Mutations in the HFE gene, in particular C282Y and H63D, disrupt this control, allowing unchecked accumulation of body iron stores. 2 Homozygous C282Y is likely to lead to clinically relevant iron overload, followed by heterozygotes for C282Y and H63D, while homozygous H63D is associated with a lower risk of iron overload. 1 Over years this iron overload can lead to cirrhosis, diabetes, arthropathy, hypogonadism, and cardiomyopathy. 2
This case presents a patient with homozygosity for the H63D mutation who underwent a prior bariatric gastric sleeve procedure yet developed biochemical evidence of iron overload, contrary to the potential for post-bariatric iron deficiency. 3 The coexistence of a bariatric surgical history – which may reduce iron absorption – and a low-penetrance H63D mutation – which rarely leads to clinical iron overload – creates a diagnostic conundrum, particularly in a patient with normal levels of ferritin but with elevated transferrin saturation. We report this case to highlight the diagnostic complexity of iron overload after bariatric surgery.
Case Report
A 51-year-old man with a history of class III obesity, vitiligo, hypertension, dyslipidemia, and obstructive sleep apnea was evaluated in the endocrinology clinic for weight management and inability to lose weight. He had undergone sleeve gastrectomy in July 2021 for severe obesity, with a preoperative weight of 440 lb (199.6 kg; BMI 58.1 kg/m2). Following surgery, he achieved 97 lb weight reduction; however, over the subsequent 2 years, he regained 25 lb, prompting referral for further evaluation. His post-surgical course was complicated by persistent fatigue, decreased libido, and progressive darkening of the skin, sparing areas affected by vitiligo.
Social history was notable for a prolonged period of alcohol use prior to bariatric surgery. Prior records described a period of heavy intake lasting 4 years, during which the patient consumed up to half a bottle of whiskey daily, followed by a gradual reduction over time. One year before sleeve gastrectomy (SG), he reported drinking ~4 standard drinks/week, with consumption occurring 4 or more times/week and 5 to 6 drinks/session. At the time of SG, intake was documented as ~7 standard drinks/week. Four years postoperatively, he reported limited use of ~1 drink on weekends, supported by a phosphatidylethanol level of 82 ng/mL (<200 ng/mL), consistent with moderate consumption. No formal diagnosis of alcohol use disorder was documented in the available medical record.
At initial endocrinology follow-up 3 years and 10 months after SG, his weight was 355.6 lb, up from 343 lb earlier that year. Diet modifications had stabilized weight over the prior 6 months, though he reported poor overall dietary quality and inconsistent exercise. His blood pressure measured 137/74 mmHg and pulse 71 bpm. Past medications included semaglutide for 2 months, which was discontinued due to unaffordability; tirzepatide, initiated 3.5 years post SG and discontinued 8 months later due to minimal response; phentermine-topiramate, discontinued 4 years post SG; and metformin. He was also taking atorvastatin, calcium-vitamin D, and cyanocobalamin supplements.
InBody body composition analysis demonstrated significant adiposity, with 36.7% body fat and a visceral fat area of 256.2 cm2. Fat-free mass and skeletal muscle mass were relatively preserved, yet overall body habitus remained consistent with class III obesity (BMI 46.9 kg/m2). A FibroScan performed 3 years and 9 months after SG demonstrated F3 fibrosis (advanced fibrosis) and grade 3 steatosis, indicating severe hepatic fat accumulation and significant liver scarring (Figure 1). These findings were consistent with metabolic dysfunction-associated steatotic liver disease (MASLD) in the setting of severe obesity and dyslipidemia. His family history was notable for obesity in his mother and brother and hepatocellular carcinoma in his father. Social history included prior heavy alcohol consumption with recent reduction to moderate intake.
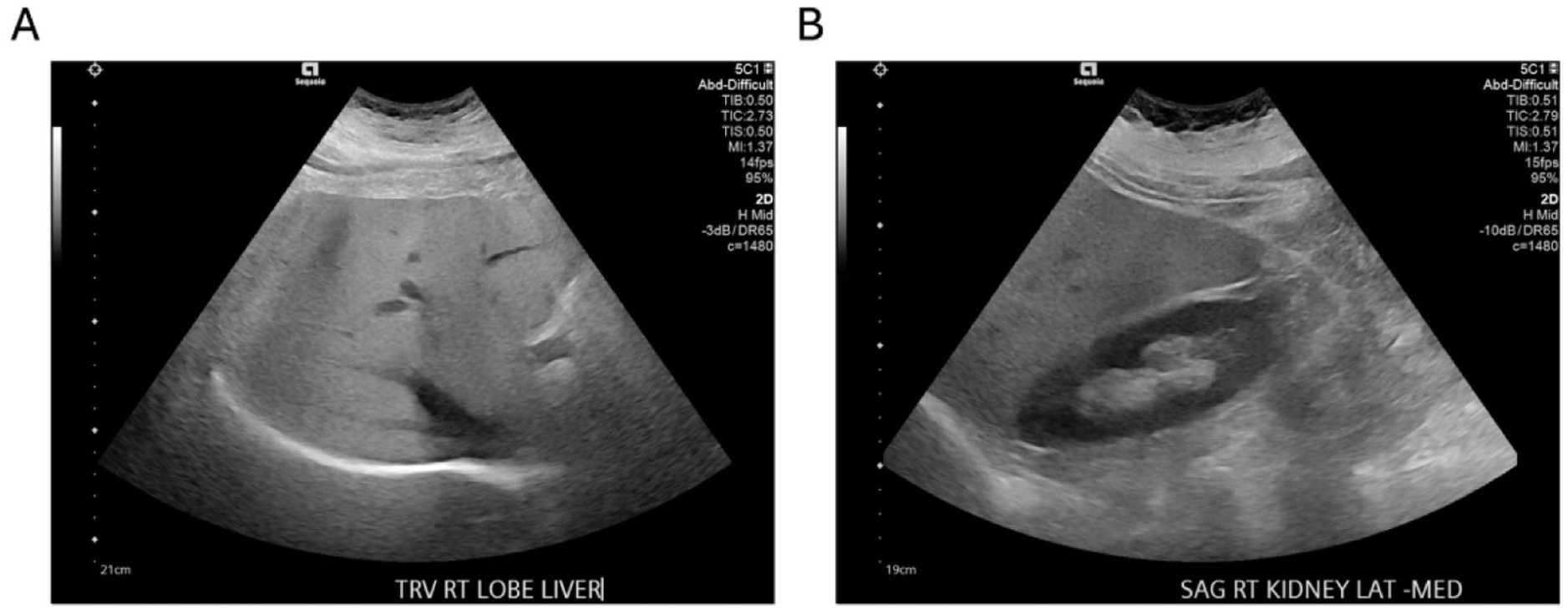
Ultrasound findings demonstrating hepatic steatosis with preserved renal cortical echogenicity for comparison. (A) Transverse right-lobe liver ultrasound showing diffusely increased hepatic echogenicity, consistent with grade 3 steatosis identified on FibroScan. (B) Longitudinal right kidney ultrasound demonstrating normal renal cortical echogenicity, providing contrast with the hyperechoic hepatic parenchyma.
Given complaints of decreased libido hormonal evaluation was performed. Fasting morning laboratories revealed elevated estradiol at 44 pg/mL (normal ≤39 pg/mL), reduced free testosterone at 32.4 pg/mL (35-155 pg/mL), and low total testosterone at 204 ng/dL (250-1100 ng/dL). In the setting of low testosterone, gonadotropins (LH and FSH) were within normal limits, representing an inappropriately normal response. This pattern is suggestive of hypogonadotropic hypogonadism, a finding commonly associated with obesity, but also potentially reflective of pituitary dysfunction from iron deposition in the setting of suspected hemochromatosis. Morning cortisol was 15 µg/dL, within normal physiologic range, while ACTH was elevated at 180 pg/mL (6-50 pg/mL), an abnormal but nonspecific finding. Liver enzymes were normal (AST 20 U/L, ALT 19 U/L). Vitamin D was low at 26 ng/mL.
Since preoperative iron studies were unavailable, iron overload physiology prior to sleeve gastrectomy could not be assessed. As part of routine post-surgical monitoring, iron studies were obtained. Indices measured ~5 months after SG were within normal limits, with serum iron of 103 µg/dL (50-180 µg/dL), ferritin of 134 ng/mL (38-380 ng/mL), and transferrin saturation of 32% (20%-48%), indicating absence of iron deficiency or overt iron overload in the early postoperative period. However, preoperative iron overload cannot be excluded. Given that HH progresses slowly and often presents between 40 and 60 years of age, early iron loading may have been present but masked by reduced absorption after SG. 2 In contrast, evaluation ~3 years after SG demonstrated iron overload physiology, with serum iron at 229 µg/dL and transferrin saturation of 60%, while ferritin remained within the normal range at 118 ng/mL. This discordant profile, marked by transferrin saturation elevation with normal ferritin, suggested early-stage or atypical hemochromatosis, particularly in the absence of inflammatory or hepatic abnormalities. 1 While bariatric surgery may predispose patients to iron deficiency due to reduced gastric acid and altered absorption, this patient instead exhibited biochemical evidence of iron loading. 3 This paradox prompted further evaluation.
Genetic testing revealed homozygosity for the HFE p.His63Asp (H63D) mutation, a low-penetrance variant classically associated with minimal risk of clinically significant iron overload. 1 When present alone, H63D rarely leads to symptomatic hemochromatosis; however, homozygosity may increase transferrin saturation or mild iron excess. 4 In this patient, the combination of H63D homozygosity and altered gastrointestinal anatomy from bariatric surgery created a complex clinical picture. Of note, bariatric surgery may lower ferritin levels, potentially masking clinically significant iron overload and complicating early diagnosis. 5
Given the finding of hypogonadotropic hypogonadism, a pituitary MRI was ordered to assess for structural abnormalities or iron deposition. Imaging 3 years and 10 months post SG was unremarkable, showing no mass, infiltrative lesion, or signal abnormality to suggest pituitary iron overload.
For the past 4 years, since the patient’s sleeve gastrectomy surgery, his weight has fluctuated. Lifestyle efforts included a low-salt, low-fat 1800-kcal diet and a goal of 30 minutes of brisk walking daily. Alcohol reduction was encouraged, given its contribution to hepatic iron handling and metabolic derangements. The patient remained asymptomatic from a hepatic or cardiac standpoint, with no signs of arthropathy, diabetes, or cardiomyopathy.
Discussion
The present case is notable because the patient exhibited biochemical evidence of iron excess despite H63D homozygosity, which is associated with only mild elevations in transferrin saturation and a very low likelihood of progression to clinically meaningful iron overload, and a surgical and anatomic history, due to his sleeve gastrectomy, that may predispose patients to iron deficiency, not overload.1,3 This combination created a diagnostic paradox that underscores the intricate clinical presentation of HH in a patient who had undergone gastric sleeve surgery and the complexity of interpreting iron indices in post-bariatric surgery patients.
The patient’s iron profile initially appeared discordant, with serum iron and transferrin saturation markedly elevated, yet ferritin within normal limits. In early hemochromatosis, transferrin saturation is often the earliest abnormality, rising before ferritin becomes elevated as iron accumulates. 1 However, several changes after bariatric surgery such as altered eating patterns leading to reduced iron intake, a decrease in gastric acidity altering iron absorption, and decreasing surface area where iron uptake occurs may result in ferritin levels that are lower than expected. 6 Thus, the patient’s normal ferritin level may reflect the combined effects of bariatric-induced changes and the relatively mild iron-loading phenotype associated with H63D homozygosity. This pattern highlights the importance of not relying solely on ferritin to rule out HH in post-bariatric surgery patients.
In addition to altered iron physiology, the patient’s metabolic profile contributed further complexity to this case. He demonstrated advanced MASLD, with F3 fibrosis and grade 3 steatosis on FibroScan. MASLD is highly prevalent in individuals with severe obesity and contributes independently to hepatic inflammation, oxidative stress, and fibrosis. The coexistence of MASLD and hemochromatosis is clinically significant because both conditions can synergistically accelerate liver injury.1,7 Even in low-penetrance H63D cases, the presence of metabolic dysfunction, dyslipidemia, and excess adiposity may amplify iron-related hepatic stress, thereby lowering the threshold at which iron excess becomes clinically meaningful. 8 Although the patient had no current biochemical evidence of liver inflammation and remained asymptomatic, advanced fibrosis (F3) places him at increased long-term risk for cirrhosis and hepatocellular carcinoma, especially in the presence of alcohol intake and genetic predisposition.
Alcohol use represents an additional key factor in this patient’s presentation. While his PEth level indicated only moderate intake, weekend binge-pattern consumption of wine and spirits may still meaningfully affect iron homeostasis and liver health. Alcohol acts in several ways to increase iron levels as it increases intestinal iron absorption by suppressing hepcidin expression while promoting hepatic oxidative injury, both mechanisms that overlap with the pathophysiology of HH. 9 In individuals with HFE mutations, even modest alcohol intake can worsen iron loading and increase the risk of fibrosis. 8 The patient’s strong family history of hepatocellular carcinoma further elevates his risk profile. Although no current evidence of cirrhosis or hepatic decompensation was observed, ongoing counseling regarding alcohol reduction is essential.
Another important dimension of this case involves the endocrine abnormalities suggestive of hypogonadotropic hypogonadism. Iron deposition into the pituitary gland is a well-known complication of HH, often leading to early dysfunction of gonadotroph cells.10,11 Although pituitary MRI revealed no structural abnormalities or signal changes indicative of iron deposition, MRI has limited sensitivity in detecting early or mild iron loading. In many cases of secondary hypogonadism associated with hemochromatosis, the pituitary appears normal radiographically. 12 The patient’s hormonal profile, characterized by low testosterone with inappropriately normal LH and FSH, supported a central etiology. 13 That said, obesity itself can suppress the hypothalamic-pituitary-gonadal axis through estradiol excess from peripheral aromatization of testosterone, leptin resistance, inflammatory signaling, and alterations in kisspeptin-mediated GnRH secretion.13,14 Thus, the patient’s hypogonadism is likely multifactorial, reflecting contributions from obesity, metabolic dysfunction, and possible early iron-mediated endocrine disruption.
The patient’s elevated ACTH level further added to diagnostic considerations. Plasma ACTH measured 2 years before SG was within normal limits at 36.9 pg/mL (6-50 pg/mL) with a normal cortisol at 7.3 μg/dL (6-19 μg/dL). Repeat testing ~3 years after SG demonstrated marked elevation to 180 pg/mL, while morning cortisol remained within the normal physiologic range (15.0 μg/dL). Subsequent testing 3 months later demonstrated improvement with ACTH at the upper level of normal (50 pg/mL) and cortisol at 9.7 μg/dL, supporting a transient rather than progressive abnormality. In the absence of hypocortisolism or clinical features of adrenal insufficiency, evolving primary adrenal failure was considered unlikely. Although iron overload can rarely contribute to adrenal dysfunction, the lack of biochemical progression and potential laboratory variability supported an alternative explanation.
Moreover, obesity, alcohol use, chronic stress, and metabolic illness can dysregulate the hypothalamic-pituitary-adrenal (HPA) axis, leading to intermittent elevations in ACTH without clear adrenal pathology.8,15 Additionally, advanced MASLD may influence cortisol clearance and HPA axis function. 16 In the absence of hypercortisolism or adrenal insufficiency, this pattern likely reflects physiologic stress rather than primary endocrine disease. 17 Nonetheless, the interplay of metabolic stress, hepatic dysfunction and potential early iron effects merits continued monitoring.
The patient’s bariatric surgery history also interacts meaningfully with the endocrine and metabolic findings. Sleeve gastrectomy, unlike Roux-en-Y gastric bypass, preserves duodenal continuity but still significantly alters gastric acid production and nutrient absorption. 18 Postoperative weight regain, as seen in this case, is not uncommon and may reflect environment and behavioral factors, metabolic adaptation, or hormonal changes.19,20 To address this issue, medications including semaglutide were trialed with good efficacy. The reinstatement of semaglutide with resulting weight loss is encouraging and aligns with current evidence demonstrating benefits of GLP-1 receptor agonists in obesity, glycemic control, and MASLD improvement.21,22 Given his F3 fibrosis and ongoing metabolic risk, durable weight reduction remains a critical therapeutic priority.
The constellation of findings in this case – iron overload with normal ferritin, hypogonadotropic hypogonadism, advanced MASLD, bariatric surgery history, and H63D homozygosity – illustrates the challenge of diagnosing hemochromatosis in patients with complex metabolic disease (Figure 2). This case reinforces several important clinical principles. First, clinicians should rely on a comprehensive assessment in identifying presentations of HH, particularly in individuals with prior bariatric surgery or depleted iron stores. Second, H63D homozygosity, though traditionally considered low risk, can still manifest biochemical or clinical features of iron loading when compounded by metabolic dysfunction, alcohol intake, or hepatic disease. Third, endocrine abnormalities such as secondary hypogonadism may still serve as early indicators of iron-related pituitary effects, even in the absence of overt MRI findings. Finally, the coexistence of advanced MASLD heightens the importance of early recognition, risk factor modification, and ongoing surveillance for progressive liver disease.

Multifactorial contributors to iron overload physiology in an H63D-homozygous post-sleeve gastrectomy patient. Diagram illustrating the intersecting genetic, surgical, metabolic, lifestyle, and non-modifiable factors shaping the patient’s iron overload physiology. Despite the low-penetrance HFE H63D genotype, elevated serum iron and transferrin saturation emerged in the context of MASLD with advanced fibrosis, metabolic dysfunction, moderate alcohol consumption, and physiologic changes following sleeve gastrectomy. Physiologic changes following sleeve gastrectomy are depicted as modifying factors that may mask iron loading rather than promote iron accumulation. Together, these factors increased iron absorption and impaired hepatic iron handling while masking ferritin elevation, resulting in an atypical presentation of hemochromatosis physiology. MASLD, metabolic dysfunction-associated steatotic liver disease.
Footnotes
Author Note
Prior Presentation of Abstract Statement: This abstract was previously presented in poster form at ENDO 2025, held in San Francisco, CA, on July 13, 2025. The abstract was also published in the Journal of the Endocrine Society, volume 9, issue Supplement 1 (October-November 2025), Abstract bvaf149.202, and is available online at ![]() (published October 22, 2025).
(published October 22, 2025).
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
Verbal consent for participation of this case report and any accompanying images was obtained from the patient.
Consent for Publication
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
