Abstract
Hyperleukocytosis (HL) in acute leukemia is associated with increased morbidity and mortality, mainly secondary to the effects of leukostasis. Many strategies for cytoreduction exist, however prompt initiation of chemotherapy is paramount. Leukapheresis is an additional strategy, though there is conflicting data for its use in HL in acute leukemia. Below we detail a case of HL and leukostasis in newly diagnosed pediatric T-cell acute lymphoblastic leukemia and the use of leukapheresis. We highlight that leukapheresis can rapidly and safely reduce the white blood cell count, as an adjunct to symptom management, and potentially prevent life-threatening complications while not delaying chemotherapy initiation. However, this case underscores that early recognition of HL prior to the development of severe neurological complications is critical, as once cerebral leukostasis with intracranial hemorrhage is established, neither leukapheresis nor chemotherapy may alter the fatal outcome. The patient’s initial presentation with epistaxis and hematemesis two days prior—without laboratory evaluation—represents a missed opportunity that may have allowed earlier recognition and intervention.
Introduction
Hyperleukocytosis (HL) is defined as a white blood cell (WBC) count > 100 × 109/L. It is an oncologic emergency associated with increased morbidity and mortality in pediatric patients presenting with acute leukemia. HL is present in approximately 10% to 30% of pediatric patients with newly diagnosed acute leukemia. 1 It is associated with young age, especially those less than one year, T-cell acute lymphoblastic leukemia (ALL) subtype, and specific gene arrangements, including KMT2A and BCR-ABL.1-3
HL increases blood viscosity and can lead to leukostasis, a life-threatening condition in which impedance of blood flow in microvasculature may cause decreased tissue perfusion and end organ dysfunction. 4 Symptomatic leukostasis most commonly affects the pulmonary, cerebral, and renal microvasculature. 5 Early recognition and intervention is necessary to avoid life-threatening complications. HL is more common in ALL than in acute myeloid leukemia (AML), particularly in pediatric patients; however, leukostasis is more frequently associated with AML as myeloblasts are larger, less deformable, and have increased endothelial adhesion compared to lymphoblasts. 6 Given this, small vessel occlusion can occur in patients with AML when WBC > 50-100 × 109/L, whereas it is not usually observed until WBC > 400 × 109/L in those with ALL.7,8
Other risks associated with HL include disseminated intravascular coagulation (DIC) and tumor lysis syndrome (TLS). DIC, a coagulopathy characterized by diffuse microthrombosis and consumption of coagulation factors and platelets, is caused by high cell turnover and endothelial damage, leading to release of tissue factor and systemic activation of coagulation. 9 Acute DIC is characterized by a decrease in platelet count and fibrinogen, an elevation of fibrin degradation products (such as d-dimers), and prolongation of the prothrombin time. 10 Patients with HL are at high risk for TLS given their significant leukocyte burden. 11 First-line treatment for HL includes prompt initiation of cytoreductive chemotherapy, along with aggressive supportive care.1,8 Supportive care strategies include administration of blood products (platelet, cryoprecipitate, and/or plasma) to correct coagulopathy, prevention or treatment of TLS which includes hyperhydration, as well as leukapheresis to prevent or treat symptomatic leukostasis.
Leukapheresis, a procedure in which WBCs, including leukemic blasts, are mechanically separated from whole blood, can reduce circulating leukocytes by 20%-50%. 12 Leukapheresis requires specialized equipment and trained personnel. To maintain adequate flow rates, central venous access with a large bore catheter is often necessary, which may increase the risk of bleeding, thrombosis, and infection. The procedure typically uses a citrate-based anticoagulant, which, if not properly managed, can lead to citrate toxicity and symptomatic hypocalcemia. 12
A review of the pediatric oncology literature reveals no consensus on the use of leukapheresis in HL in acute leukemia.13-16 Current apheresis guidelines in transfusion medicine classify therapeutic leukapheresis for HL as a category III recommendation, indicating uncertain benefit and the need for individualized decision-making. 17 National Comprehensive Cancer Guidelines for pediatric leukemia note that leukapheresis has been demonstrated to reduce complications of leukostasis in patients with ALL, but in cases of HL without symptoms of leukostasis, leukapheresis provides no clinical advantage over aggressive chemotherapy, and sometimes may be associated with adverse outcomes.18,19
Despite this lack of consensus, we highlight a case in which leukapheresis effectively and safely reduced the intravascular leukemic burden without procedural complications. However, this case also illustrates that early recognition of HL is paramount.
Clinical Presentation
A previously healthy 11-year-old female with one week of abdominal pain, nausea, epistaxis, and hematemesis presented to the emergency department by ambulance after being found unresponsive at home by parents. She had been evaluated at an outside emergency department two days prior for multiple episodes of epistaxis and an episode of hematemesis, which was believed to be secondary to the epistaxis. She was discharged home without any laboratory evaluation and with supportive care including oxymetazoline nasal spray and air humidification. On arrival to our facility, the patient was unresponsive with a GCS of 6, including withdrawal to pain, and thus was urgently intubated. Other notable exam findings included a 6-mm dilated and nonreactive left pupil, a 3-mm sluggishly reactive right pupil, sinus tachycardia, hepatosplenomegaly, and inguinal lymphadenopathy. There were no signs or history of trauma.
Head CT revealed extensive edema and multifocal hemorrhages throughout the cerebral hemispheres and brainstem (Figures 1 and 2), along with early hydrocephalus with compression of the cerebral aqueduct (Figure 3). There was evidence of early herniation of the cerebellar tonsils into the foramen magnum and thus was given 1 mg/kg of mannitol. No acute neurosurgical intervention was offered due to high bleeding risk. She was noted to have some improvement in her neurologic examination following mannitol and other aggressive neuroprotective measures.
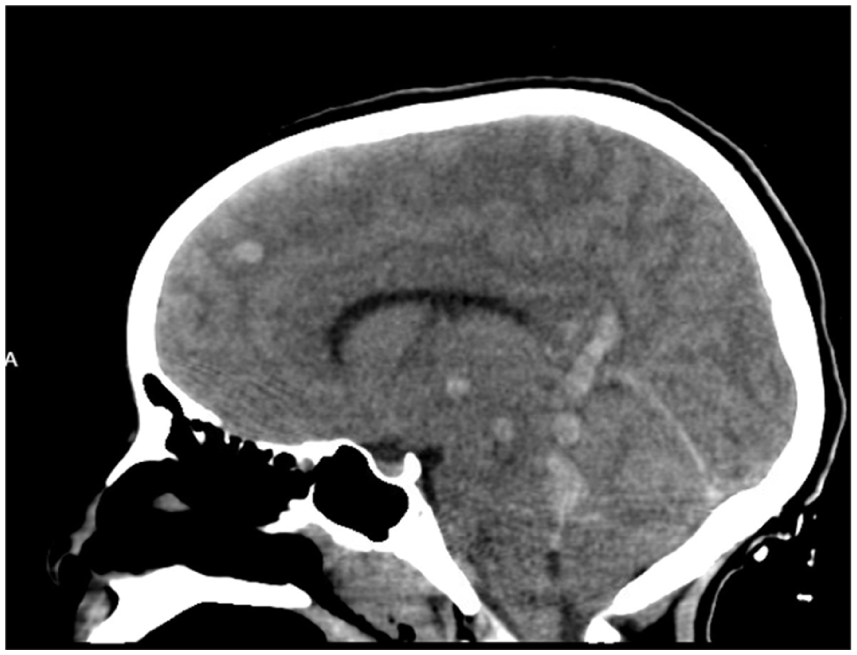
Initial head CT without contrast revealed extensive multifocal hemorrhages and edema visualized in the sagittal plane.

Initial head CT without contrast revealed extensive multifocal hemorrhages and edema visualized in the axial plane.

Initial head CT without contrast demonstrated hemorrhage and edema in the brainstem obstructing the cerebral aqueduct. Early extension of cerebellar tonsils into the foramen magnum is present.
Initial laboratory workup was notable for a CBC with WBC count of 1309 × 109/L (4.5-13.5 × 109/L) including 95% blasts (1244 × 109/L), Hb 8.0 g/dL (11.5-15.5 g/dL), and platelet count of 57 × 109/L (150-400 × 109/L). She was found to have overt DIC 10 given her thrombocytopenia, d-dimer of 22 068 ng/mL FEU (<500 ng/mL FEU), PT > 100 seconds (9.9-13.7 seconds), and fibrinogen of 58 mg/dL (160-450 mg/dL). She received fresh frozen plasma and intravenous vitamin K. There was evidence of TLS with initial chemistry remarkable for K+ 6.9 mmol/L (3.5-5.1 mmol/L), acute kidney injury with Cr 0.62 mg/dL (patient’s baseline was 0.4 mg/dL), uric acid 12.0 mg/dL (3.5-6.8 mg/dL), and LD 13 251 U/L (115-257 U/L). Phosphorous was normal at 4.0 mg/dL. She received 0.1 mg/kg rasburicase, 5 mg (5.9 mg/m2) dexamethasone, a 20 mL/kg normal saline bolus, and was started on intravenous fluids at twice maintenance rate. She received prophylactic levetiracetam and was quickly admitted to the pediatric intensive care unit. Dexamethasone was continued at 5 mg every twelve hours for both cytoreduction and treatment of intracranial edema.
Given the degree of HL with signs of intracranial leukostasis, a 15-L volume leukapheresis was performed via a femoral central venous catheter on the Spectra Optia (Terumo BCT) using anticoagulant citrate dextrose solution A for anticoagulation. Intravenous calcium gluconate was infused throughout the procedure to prevent citrate toxicity. She tolerated leukapheresis well without complications. The patient’s WBC count decreased by 52%, dropping from 1309 × 109/L to 622 × 109/L following the procedure. After further decreases in hemoglobin and platelet counts following the procedure, she received a 5 mL/kg red blood cell transfusion, given in a small aliquot as to not significantly increase blood viscosity, and one unit of platelets.
Later that evening, there was concern for worsening neurologic status and risk of herniation, so she received 5 mL/kg hypertonic saline in addition to continued aggressive neuroprotective interventions. Several hours later she had worsened hypertension, likely signifying a herniation event, and developed fixed and dilated pupils with loss of all neurologic responses. Subsequent physical exams were consistent with brain death and life support was ultimately withdrawn. Peripheral blood flow cytometry later confirmed the diagnosis of T-cell ALL.
Discussion
HL is an oncologic emergency in acute leukemia with high risk of morbidity and mortality, primarily due to neurologic and pulmonary leukostasis. Despite its clinical significance, there is no established consensus regarding the use of leukapheresis in pediatric patients with HL. Current American Society for Apheresis (ASFA) guidelines (Figure 4) classify leukapheresis for HL as a category III, grade 2B recommendation, reflecting uncertain benefit and the need for individualized clinical decision-making based on moderate-quality evidence. 17 The ASFA guidelines suggest that leukapheresis may be considered for symptomatic leukostasis in patients with WBC counts >100 × 109/L in AML and >400 × 109/L in ALL. These guidelines are derived from retrospective, observational based studies, and no randomized controlled trials have been conducted to date.13,14,16

2023 ASFA Guidelines on the Use of Therapeutic Apheresis in Clinical Practice. The use of leukapheresis in hyperleukocytosis is overall a category III recommendation where the optimum role of apheresis therapy is not established, and decision-making should be individualized. Its use is also a grade 2B recommendation which is described as a weak recommendation with moderate-quality evidence.
Available retrospective studies have not demonstrated a clear survival benefit from leukapheresis in patients with AML.14,16,20 A meta-analysis including thirteen studies and 1743 total adult patients with AML similarly found no improvement in early mortality among those who underwent leukapheresis for HL. 16 Interpretation of these data is challenging, as leukapheresis is often reserved for the most critically ill patients, introducing significant selection bias. Data in ALL are even more limited, as leukostasis is less common and typically occurs only at very high WBC counts (>400 × 109/L). Consequently, the role of leukapheresis in pediatric ALL remains particularly difficult to define. While large pediatric randomized trials would be valuable, their feasibility is limited by ethical and safety considerations. A Children’s Oncology report suggests that leukapheresis does not reduce induction mortality in pediatric AML. 21
Leukapheresis also has practical and clinical limitations. The procedure requires specialized equipment, experienced personnel, and adequate venous access, often necessitating placement of a large-bore central venous catheter. Citrate-based anticoagulation may result in electrolyte disturbances, particularly hypocalcemia, and careful monitoring of fluid balance is required. In addition, leukemic blast counts often rebound rapidly, and repeated procedures may be necessary to achieve sustained cytoreduction.
The present case illustrates the technical effectiveness of leukapheresis in the setting of symptomatic leukostasis, with a rapid reduction in circulating leukocyte burden from 1309 × 109/L to 622 × 109/L without procedural complications or delay in therapy initiation. However, despite successful cytoreduction, leukapheresis was clinically insufficient to alter the patient’s outcome given the severity of disease at presentation. Her death resulted from catastrophic complications of leukostasis and DIC that were already established upon arrival.
This case underscores the critical importance of early recognition and intervention in HL. A retrospective single-center study of pediatric patients with acute leukemia demonstrated that neurologic symptoms, coagulopathy, and WBC counts >400 × 109/L at presentation are strong predictors of early death due to leukostasis. 22 Once severe cerebral or pulmonary involvement has developed, neither leukoreduction with chemotherapy nor leukapheresis is likely to meaningfully improve survival. 22 In our patient, the presence of profound neurologic deficits and coagulopathy at admission indicated an already guarded prognosis. It is unclear whether a focused physical examination, including assessment for organomegaly, was performed during her initial emergency department visit for epistaxis and hematemesis two days earlier.
Leukapheresis can serve as a temporizing adjunct to rapidly reduce intravascular leukocyte burden and potentially improve tissue perfusion in select patients with symptomatic leukostasis. 16 Nevertheless, prompt initiation of definitive chemotherapy remains the cornerstone of treatment for HL, regardless of whether leukapheresis is employed. In this case, leukapheresis safely achieved cytoreduction but did not result in neurologic recovery or improved survival, as irreversible complications were present prior to intervention.
In summary, while leukapheresis may be a useful adjunctive therapy for symptomatic leukostasis in carefully selected pediatric patients, its benefits are limited once advanced organ dysfunction has occurred. This case highlights a systems-level opportunity for earlier laboratory evaluation and recognition of HL in pediatric patients presenting with unexplained mucosal bleeding, which may allow intervention before the development of irreversible complications.
Footnotes
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series
Consent to Participate
Verbal informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
