Abstract
Methemoglobinemia causes hypoxia and can be congenital or acquired through chemical or drug exposure. Standard therapy for non-pregnant symptomatic cases is methylene blue, which poses potential fetal risks in pregnancy. We report a case of a pregnant patient with methemoglobinemia who presented with dyspnea at 18 weeks of gestation. Intravenous ascorbic acid was administered with resolution of dyspnea, and the patient was started on daily oral ascorbic acid upon discharge. With avoidance of potentially inciting agents, the patient proceeded to have a full-term, uncomplicated spontaneous vaginal delivery of a viable female neonate. This report analyzes previously reported cases of methemoglobinemia in pregnancy, its pathophysiology, diagnostic workup, and suggested management. Daily ascorbic acid may be a feasible alternative to methylene blue for the management of symptomatic methemoglobinemia in pregnancy.
Introduction
Methemoglobin (MHb) is an abnormal hemoglobin molecule that contains oxidized ferric iron (Fe3+) instead of the usual ferrous iron (Fe2+) at 1 or more of the 4 iron-containing heme groups. This modification disables oxygen binding at the affected heme group(s) and impairs oxygen release at the unaffected heme group(s). 1 Methemoglobinemia occurs when there is a >1% accumulation of MHb in the blood and results in tissue hypoxia and functional anemia. 2 Accumulation of MHb is normally prevented via enzymes that reduce non-functional Fe3+ to functional Fe2+; however, methemoglobinemia may occur in the setting of impaired enzymatic systems (congenital) or significant exposure to oxidizing agents (acquired). Primary treatment in non-pregnant symptomatic cases includes intravenous or oral administration of methylene blue (tetramethylthionine chloride), which activates the secondary enzymatic pathway of MHb reduction. 3 In pregnancy, however, methylene blue has been reported to be associated with fetal demise or ileal/jejunal atresia of the fetus when administered intra-amniotically. 4 Only a few cases of symptomatic methemoglobinemia in pregnancy have been reported, and its management has varied widely. We discuss this case to raise awareness of this rare hematologic condition in pregnancy, educate on the pathophysiology of methemoglobinemia and diagnostic workup, and demonstrate the possible role of ascorbic acid in the management of persistently symptomatic methemoglobinemia in pregnancy.
Case Description
A 33-year-old woman, G2 P1001 at 18 weeks of gestation, presented for outpatient evaluation by Maternal-Fetal Medicine due to dyspnea described to be chronic and worsening since the beginning of her pregnancy. Her baseline pulse oximetry readings at home were reportedly 92% to 93%.
The patient’s medical history was significant for methemoglobinemia diagnosed 7 years earlier, immediately following the uncomplicated term vaginal delivery of her first child. She had no family history of methemoglobinemia and no symptoms prior to the inciting event. That pregnancy and labor were uncomplicated, and the newborn’s size was appropriate for gestational age. However, after receiving 30 mL of locally injected lidocaine for the repair of a perineal laceration, the patient immediately developed acute dyspnea, cyanosis, and a sudden decrease in her pulse oximetry reading to ~80%. Her MHb level was found to be 18.3% during this episode, and her symptoms resolved after administration of intravenous methylene blue. The patient’s postpartum course was unremarkable, and the patient was discharged home with her infant. As an outpatient, she was started on twice daily oral ascorbic acid (vitamin C) 1 g and daily oral methylene blue 100 mg, which was later titrated up to 200 mg daily. Despite this regimen, the patient continued to have chronic fatigue and dyspnea on exertion, which later led to hospitalization for an acute exacerbation. The patient’s MHb level was 13.5% during this hospitalization, and intravenous methylene blue was again administered with a rapid reduction of her MHb level to 0.9%. In the outpatient setting, her MHb level was found to have increased to 12.5% within the span of 1.5 years. The patient eventually self-discontinued the oral methylene blue and ascorbic acid regimen in preparation for the current pregnancy. The patient described experiencing worsening dyspnea, fatigue, and headaches since becoming pregnant, and at 12 weeks of gestation, she presented to an outside hospital emergency department for an acute-on-chronic exacerbation of dyspnea and a home pulse oximetry reading of 88%. She was placed on a non-rebreather with improvement of symptoms. After ruling out venous thromboembolism, COVID-19 infection, and other cardiopulmonary pathologies, the patient was discharged. Methylene blue was not administered at this time due to concern for potential teratogenicity. Following discharge, the patient started using supplemental oxygen with 2 L nasal cannula at home (6-7 hours in the morning and 1.5 hours before bed).
After an outpatient consultation with Maternal-Fetal Medicine at 18 weeks of gestation, hospital admission was recommended due to worsening dyspnea, with plans for an inpatient evaluation by Hematology. On admission, the patient was afebrile at 98.6°F, with a blood pressure of 125/62 mm Hg, pulse of 81/minute, respiratory rate of 18/minute, and pulse oximetry reading of 92% to 94% on 4 L nasal cannula. Her physical examination was significant for dyspnea while speaking and orthopnea. Cardiovascular and respiratory system examinations were otherwise normal. Her fundus was soft and non-tender. The fetal heart rate was 140 bpm by Doptone. Transabdominal ultrasonographic examination showed a normally developing fetus whose size was appropriate for gestational age. All routine investigations were normal except for an elevated MHb level of 11.3% on arterial blood gas (ABG) analysis. The ABG oxygen saturation was found to be 99%.
Given the elevated level of MHb and dyspnea, Hematology recommended ascorbic acid 2 g IV, which did not significantly decrease the percentage of MHb upon reassessment the following day (10.3%) but did improve her symptoms. The patient was weaned off supplemental oxygen without difficulty and was discharged on hospital day 3 in stable condition without any further dyspnea. On discharge, the patient had a pulse oximetry reading of 97% on room air. A repeat ABG revealed an oxygen saturation of 97.7%. Twice daily oral ascorbic acid 1 g was re-initiated.
The patient was adherent to the daily oral ascorbic acid therapy. One month after admission, the patient followed up with outpatient Hematology and reported fatigue and mild exertional dyspnea of usual severity. She no longer required supplemental oxygen at home. Serum studies showed a MHb level of 8.2% and a quantitative glucose-6-phosphate dehydrogenase (G6PD) enzyme level that was within normal limits. The hematologist recommended obtaining an arterial MHb level every 4 weeks, but the patient was non-adherent to this plan. Genetic testing for cytochrome B5 reductase (Cyb5R) mutations and hemoglobin electrophoresis testing to identify hemoglobin M (HgM) was also ordered to determine inherited causes of her methemoglobinemia; however, the testing was never performed. The patient continued to follow up with her primary obstetrician for prenatal visits without any further complications in her prenatal course. Subsequent ultrasonographic examinations continued to show a normally growing fetus throughout the remainder of the pregnancy.
At 38 2/7 weeks, the patient was admitted to labor and delivery after spontaneous rupture of membranes. At the time of labor, the patient denied headache, chest pain, or shortness of breath. Continuous cardiotocography showed no evidence of fetal heart rate decelerations. ABG demonstrated a MHb level of 9.5% and an oxygen saturation level of 97.6%. The patient had an uneventful labor without regional analgesia, and had a spontaneous vaginal delivery of a 3.25 kg (7 lb 2.6 oz) female infant with Apgar scores of 8 and 9 at 1 and 5 minutes, respectively. A second degree perineal laceration was noted. Local anesthetics, especially benzocaine and lidocaine, were avoided to prevent methemoglobinemia exacerbation, and intravenous fentanyl was given during the repair.
The patient’s postpartum course was uncomplicated. She did not develop cyanosis, dyspnea, chest pain, or headache and was discharged to home on postpartum day 1 with twice daily ascorbic acid 1 g. Her infant had an unremarkable newborn evaluation and was discharged with the patient. The patient was seen in follow-up 1 month after delivery and reported continued adherence to daily ascorbic acid 1 g. She was doing very well without evidence of symptomatic methemoglobinemia.
All available MHb levels for this patient are summarized in Table 1. The etiology for this patient’s methemoglobinemia remains undetermined. While a complete diagnostic workup was recommended, the patient did not elect to pursue additional testing following her pregnancy. We hypothesize that the patient’s distance from the testing center and disinterest in pursuing further pregnancies may have been contributing factors.
Patient MHb Levels.

Abbreviations: IV, intravenous; MHb, methemoglobin.
Discussion
Pathophysiology
A single hemoglobin molecule contains 4 heme groups, each bound to a Fe2+ atom that can reversibly bind to oxygen. When hemoglobin becomes oxidized, it loses an electron, which converts a Fe2+ atom into the Fe3+ state and produces the abnormal hemoglobin molecule known as MHb. Fe3+ lacks the electron that is necessary for oxygen binding and is therefore incapable of transporting oxygen. The result is an overall reduced oxygen-carrying capacity and allosteric change in the molecule that causes a leftward shift of the oxygen-dissociation curve. The remaining Fe2+-containing heme sites subsequently have an increased affinity to oxygen that hinders the release of oxygen to tissues. 5 Clinically, this results in methemoglobinemia’s characteristic central cyanosis and hypoxic symptoms (ie, light-headedness, fatigue, headaches, dyspnea, tachycardia) that become more severe at higher concentrations of MHb. MHb levels between 50% and 70% are associated with coma, seizures, arrhythmias, lactic acidosis, central nervous system depression, and dysrhythmias, while levels >70% are considered lethal. 6
MHb is produced as a result of oxidative stress in erythrocytes. The amount of MHb in the blood is physiologically maintained at <1% by 2 enzymatic processes (see Figure 1) that facilitate the reduction of MHb to hemoglobin: an NADH-dependent system that relies on Cyb5R (primary pathway) and an NADPH-dependent system, that is, driven by G6PD (secondary pathway). The primary pathway predominates and is responsible for 95% to 99% of MHb reduction. 3 Methemoglobinemia occurs when there is an accumulation of MHb due to either an impairment in reduction processes (congenital) or, more commonly, increased production of MHb due to significant oxidative stress that overwhelms the reduction processes (acquired). Many drugs and substances found in the environment have been identified as oxidizing agents and may cause acquired methemoglobinemia or exacerbate congenital methemoglobinemia (Table 2). The most common causes of acquired methemoglobinemia include nitroglycerine, dapsone, sulfonamides, phenacetin, and local anesthetics such as lidocaine, benzocaine, or prilocaine. 5
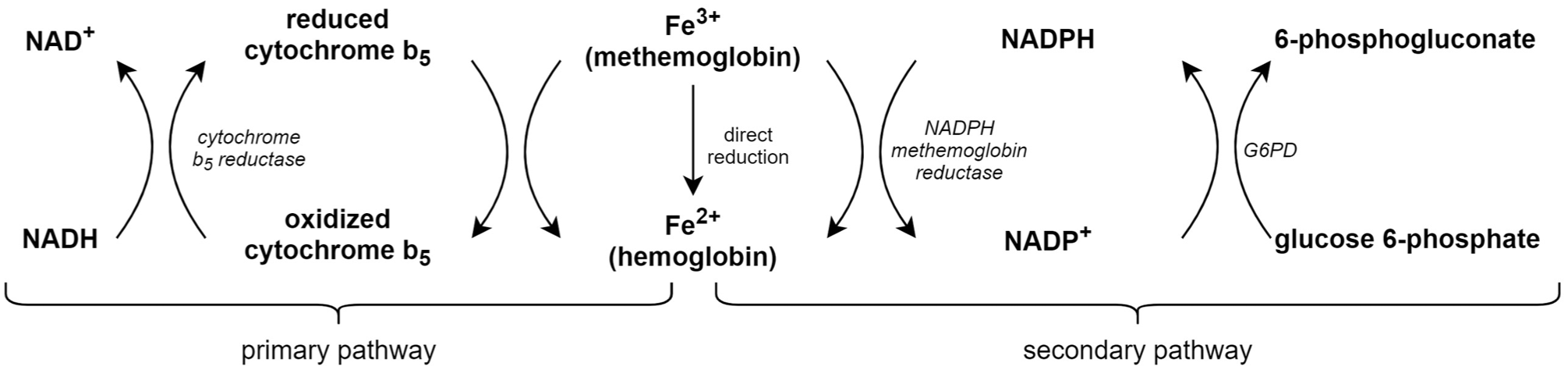
The physiologic amount of MHb in the blood is maintained at <1% by 2 enzymatic processes that reduce MHb to hemoglobin. The primary pathway predominates and is an NADH-dependent system that relies on the enzyme cytochrome B5 reductase. The secondary pathway is an NADPH-dependent system that is driven by glucose-6-phosphate dehydrogenase. Though the secondary pathway plays a minor role in the reduction of methemoglobin under normal circumstances, it assumes a crucial role in patients with cytochrome B5 reductase deficiency. MHb, methemoglobin.
List of Oxidizing Agents That May Cause Acquired Methemoglobinemia. 5

Abbreviation: G6PD, glucose-6-phosphate dehydrogenase.
There are several etiologies of congenital methemoglobinemia. The recessively inherited deficiency of Cyb5R impairs the primary pathway of MHb reduction and is a common cause of congenital methemoglobinemia. In addition, the autosomal dominant HgM disease causes a persistently elevated MHb level due to the production of a structurally abnormal hemoglobin variant that resists enzymatic reduction from Fe3+ to Fe2+ state. 6 Finally, X-linked recessive G6PD deficiency can lead to the accumulation of MHb via impairment of the secondary pathway of MHb reduction. 6 G6PD-deficient individuals are unable to produce adequate amounts of NADPH to reduce MHb to hemoglobin via NADPH MHb reductase. Furthermore, the inability to produce adequate amounts of NADPH in G6PD-deficient individuals impairs the NADPH-dependent conversion of methylene blue to its metabolic product, leukomethylene blue, the therapeutic agent responsible for MHb reduction (see Figure 2); thus, methylene blue therapy is rendered ineffective for G6PD-deficient patients and may paradoxically worsen methemoglobinemia as methylene blue is itself an oxidant. 6

Methylene blue is the standard therapy for both acquired and congenital cases of methemoglobinemia. It utilizes NADPH from the G6PD-dependent hexose monophosphate shunt and secondary pathway enzyme NADPH-MHb reductase to reduce methylene blue to the metabolically active product leukomethylene blue, which ultimately acts as an electron donor to reduce MHb to hemoglobin. G6PD, glucose-6-phosphate dehydrogenase; MHb, methemoglobin.
Review of the Literature of Methemoglobinemia in Pregnancy
Maternal hypoxemia can give rise to fetal hypoxemia and can increase the risk of perinatal and infant mortality. Methemoglobinemia in pregnancy has been associated with exacerbation of existing anemia, oligohydramnios, fetal growth restriction, preeclampsia, placental abruption, and fetal demise.2,7 -10 There are several reported cases of methemoglobinemia in pregnancy (Table 3), and its management varies widely.
Summary of Case Reports of Methemoglobinemia in Pregnancy.
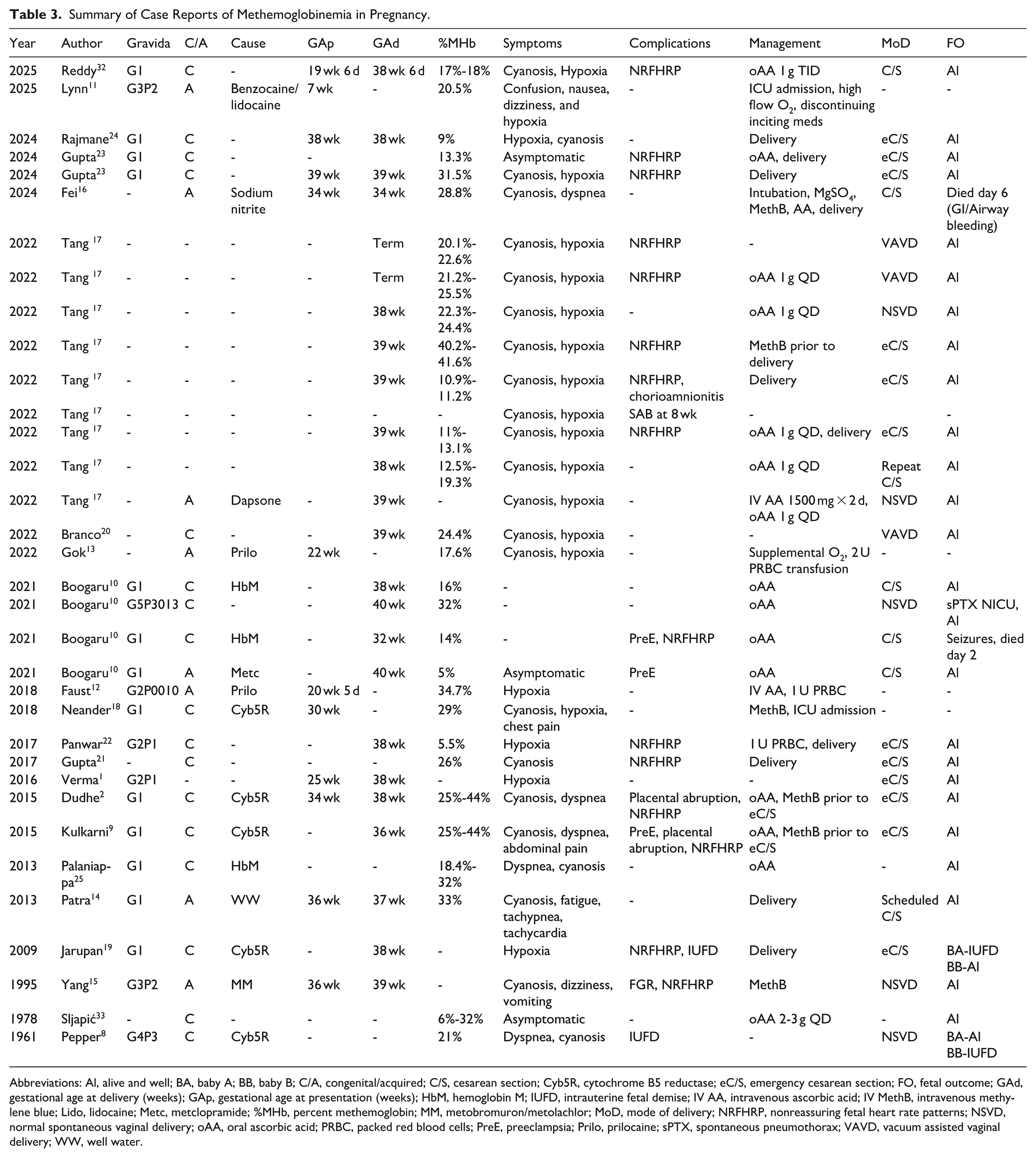
Abbreviations: Al, alive and well; BA, baby A; BB, baby B; C/A, congenital/acquired; C/S, cesarean section; Cyb5R, cytochrome B5 reductase; eC/S, emergency cesarean section; FO, fetal outcome; GAd, gestational age at delivery (weeks); GAp, gestational age at presentation (weeks); HbM, hemoglobin M; IUFD, intrauterine fetal demise; IV AA, intravenous ascorbic acid; IV MethB, intravenous methylene blue; Lido, lidocaine; Metc, metclopramide; %MHb, percent methemoglobin; MM, metobromuron/metolachlor; MoD, mode of delivery; NRFHRP, nonreassuring fetal heart rate patterns; NSVD, normal spontaneous vaginal delivery; oAA, oral ascorbic acid; PRBC, packed red blood cells; PreE, preeclampsia; Prilo, prilocaine; sPTX, spontaneous pneumothorax; VAVD, vacuum assisted vaginal delivery; WW, well water.
A systematic literature search was conducted in PubMed and Google Scholar from January 1, 1970 to January 1, 2025 using free-text parameters “methemoglobinemia” AND “pregnancy.” The search was limited to case reports. Articles for which full-text sources could not be obtained were excluded from this study. A total of 22 publications describing 34 cases were included in this literature review. Basic descriptive statistics are presented below.
Eight cases described acquired methemoglobinemia in pregnancy, which occurred after exposure to topical lidocaine/benzocaine, 11 prilocaine,12,13 well water, 14 metobromuron/metolachlor, 15 metoclopramide, 10 sodium nitrite, 16 and dapsone. 17 Seventeen cases described congenital methemoglobinemia, with 29% (5/17) attributed to Cyb5R deficiency and 18% (3/17) due to HgM disease. The remaining 53% (9/17) did not have any confirmed etiology of congenital methemoglobinemia but were suspected based on past episodes of cyanosis or significant family history. There were 9 additional cases that described methemoglobinemia in pregnancy of uncertain etiology.
Sixty-eight percent (23/34) of cases describe patients presenting with cyanosis. Fifty-nine percent (20/34) of cases reported hypoxia. Fifteen percent (5/34) of cases reported dyspnea. Nine percent (3/34) of cases described patients as asymptomatic. Fifteen percent (5/34) of cases described other symptoms, including confusion, nausea, vomiting, dizziness, chest pain, and tachycardia. All cases report MHb levels averaging 22.01% (range 5.5%-44%), with no statistically significant differences between the acquired and congenital cases (23% vs 22%, P = .77). Of cases describing patients with congenital methemoglobinemia, 47% (8/17) were unaware that they had congenital methemoglobinemia at presentation,18 -25 which supports Dudhe et al’s hypothesis that congenital methemoglobinemia may be asymptomatic and diagnosed for the first time during pregnancy when symptoms can manifest as a result of increased oxygen consumption to meet the metabolic demands of the fetus. 2
Seventy-nine percent (27/34) of cases described delivery of the patient, with 38.0 weeks being the average gestational age at delivery (range: 32-40). There was no difference between the gestational ages at delivery between patients with acquired versus congenital methemoglobinemia (37.8 vs 37.6, P = .95). Seventy percent (19/27) of deliveries were via cesarean section, 71% (12/19) of which were reported to be emergent. Thirty percent (8/27) of deliveries were vaginal deliveries, 38% (3/8) of which were vacuum-assisted. Of the prenatal and peripartum complications described, non-reassuring fetal heart rate patterns occurred in 44% (15/34) cases, preeclampsia occurred in 9% (3/34) cases, and placental abruption occurred in 6% (2/34). One case reported fetal growth restriction, 2 cases described intrauterine fetal demise, and 1 case described spontaneous abortion.
Management of patients varied widely throughout cases. Forty-four percent (15/34) described management with oral ascorbic acid, and 9% (3/34) described use of intravenous ascorbic acid. Eighteen percent (6/34) of cases reported administration of methylene blue. Of these patients who received methylene blue, 50% (3/6) were immediately prior to emergency cesarean section. Yang et al 15 described the administration of intravenous methylene blue for acquired methemoglobinemia at 36 weeks of gestation with rapid resolution of symptoms, avoiding an emergency cesarean section. This patient went on to have an uncomplicated spontaneous vaginal delivery 17 days later with birth of a vigorous infant that remained normal and healthy at 4-year follow-up, suggesting that the one-time use of parenteral methylene blue might be considered for resolution of acute methemoglobinemia in the late third trimester. Methylene blue was administered in 1 case at 30 weeks of gestation for a MHb of 29% and severe hypoxic symptoms, but there was no follow-up regarding the rest of the pregnancy course. 18 Thirty-five percent (12/34) of cases described proceeding with delivery as a part of management, 9% (3/34) described transfusion of packed red blood cells, and 6% (2/34) of cases described admission to the intensive care unit. These different approaches for managing cases of acquired and congenital methemoglobinemia illustrate the need for further studies to generate formal recommendations and a standardized approach to manage the acute hypoxic symptoms of methemoglobinemia in pregnancy.
Evaluation and Diagnosis
While the characteristic clinical manifestation of methemoglobinemia is central cyanosis due to the uniform lack of tissue oxygenation throughout the body, patients may exhibit a range of symptoms, including subtler signs of hypoxia. These symptoms, especially dyspnea or fatigue, may be overlooked in pregnant patients, given physiologic changes in pregnancy that cause similar symptoms. As such, clinicians should be familiar with how to interpret oxygenation status and assess for methemoglobinemia to efficiently arrive at a diagnosis and avoid unnecessary testing.
Because MHb is unable to bind to oxygen, methemoglobinemia leads to a reduction in oxygen saturation (the percentage of hemoglobin bound to oxygen). Routine measurement of oxygen saturation with a standard pulse oximeter offers readings that may misrepresent a patient’s oxygenation status in methemoglobinemia. Pulse oximetry is a noninvasive spectrophotometric test that calculates oxygen saturation by utilizing the absorbance measurements of light at only 660 and 990 nm, which correspond to deoxyhemoglobin and oxyhemoglobin, respectively. 26 The pulse oximetry conversion algorithm does not account for waveforms of dyshemoglobins such as MHb; therefore, pulse oximetry readings for methemoglobinemia may not be accurate. In addition, higher quantities of MHb cause the pulse oximeter to become insensitive to hypoxemia such that when MHb levels are 30% or greater, pulse oximetry readings may not record levels below 85%. 26
Arterial blood of patients with methemoglobinemia may exhibit a distinctive “chocolate-brown” color at higher concentrations of MHb. Similar to pulse oximetry, oxygen saturation levels on ABG may appear falsely normal because its computation involves the assumption of a normal oxygen dissociation curve (which does not apply to patients with methemoglobinemia) and does not account for dyshemoglobins. 27 Another commonly utilized indicator of oxygenation status obtained during blood gas analysis is the partial pressure of oxygen in blood (pO2), which informs clinicians of the total oxygen that is dissolved in blood plasma (not that which is bound to hemoglobin). Because oxygen diffusion across the alveoli to blood plasma is not impaired by methemoglobinemia, pO2 may remain normal in those with methemoglobinemia despite many clinical signs of hypoxia and may even be elevated if the patient is receiving supplemental oxygen. A clinician should suspect methemoglobinemia (or another dyshemoglobinemia) in a patient with decreased oxygen saturation as measured by pulse oximetry with a concomitant arterial pO2 value that is within normal limits.
CO-oximetry provides the most accurate measurement of methemoglobinemia. CO-oximeters spectrophotometrically examine blood at 4 wavelengths. 27 This allows for the identification and quantification of hemoglobin species beyond just deoxyhemoglobin and oxyhemoglobin, including dyshemoglobins such as MHb and carboxyhemoglobin. CO-oximetry additionally provides an accurate calculation of oxygen saturation (unlike pulse oximetry or blood gas analysis, as discussed above).
Many modern blood gas analyzers have an integrated CO-oximeter, which allows for MHb to be measured at the time of either arterial or venous blood gas analysis. Some laboratories automatically test for and report MHb percentages for all arterial and venous blood sent for blood gas analysis. If not automatically reported, the MHb level may be a separate test that a clinician must order. Alternatively, noninvasive and continuous readings of MHb can be obtained via pulse CO-oximeters, a specialized pulse oximetry device capable of 4-wavelength detection through the skin.
Management
Once methemoglobinemia is diagnosed, identification and removal of potentially oxidizing agents to prevent further production and accumulation of MHb should be the first step in managing methemoglobinemia. In non-pregnant patients, the standard therapy of both acquired and congenital cases of symptomatic methemoglobinemia is methylene blue, which utilizes the secondary pathway of MHb reduction to efficiently reduce MHb to hemoglobin. Once converted to the metabolically active product leukomethylene blue, it ultimately acts as an electron donor to reduce MHb to hemoglobin (see Figure 2). Intravenous methylene blue is administered to non-pregnant patients whose MHb level exceeds 20% to 30%, or at lower levels if the patient is symptomatic. 6 The therapeutic dose of methylene blue is 1 to 2 mg/kg intravenously, which may be repeated after 1 hour if there is no clinical improvement. 26 In a non-acute setting, daily oral methylene blue 100 to 300 mg may be given to non-pregnant patients with congenital methemoglobinemia to maintain low levels of MHb. Side effects of methylene blue include nausea, diarrhea, green or blue-tinged urine, oral dysesthesia, dyspnea, and excessive perspiration. 26
It is unclear whether intravenous or oral methylene blue can be given in pregnancy. In pregnancy, intraamniotic methylene blue dye has been associated with fetal death and intestinal atresia of the fetus. 4 A retrospective cohort study on 303 pregnant patients in New South Wales, Australia who received intraamniotic methylene blue dye during mid-trimester amniocentesis between 1980 and 1991 revealed that fetal death occurred in 31.8% of pregnancies that had exposure to a high concentration of methylene blue as compared to 14.5% of pregnancies exposed to a low concentration and 4.3% of pregnancies that had no exposure to the dye. 4 Due to the lack of safety evidence and definitive studies, oral and intravenous methylene blue is best avoided in pregnancy at this time.
Ascorbic acid is typically used to treat methemoglobinemia in patients with contraindications to methylene blue or if treatment with methylene blue has failed. It is a strong reducing agent that directly reduces MHb. 5 Its capacity to reduce MHb is inferior to that of endogenous enzymatic systems and methylene blue. 5 Therefore, it is generally not considered a first-line agent for management of symptomatic methemoglobinemia, but it can be effective in situations where methylene blue is contraindicated, such as in pregnancy. A meta-analysis of randomized studies examining 24 300 pregnant patients who took oral ascorbic acid at doses ranging from 100 to 2000 mg/day between 4 and 35 weeks of pregnancy showed no increased risk of miscarriage, preterm premature rupture of membranes, preterm delivery, or pregnancy-induced hypertension when compared to placebo. 28 Effects of doses higher than 2000 mg/day have not yet been elucidated; however, clinicians should consider that high doses of ascorbic acid can be associated with increased oxalate excretion in the urine, which may predispose a patient with renal insufficiency to renal failure due to hyperoxaluria. 29
Non-pregnant patients with severe methemoglobinemia that is non-responsive to methylene blue may also be treated with exchange transfusion and hyperbaric oxygen, 30 though the utility of these treatments for methemoglobinemia in pregnancy remains unclear. Animal models have demonstrated that the hyperoxia induced by prolonged high partial pressures of oxygen causes altered blood flow in the fetus; specifically, premature closure of the ductus arteriosus. 31 However, there is other evidence demonstrating that short durations of hyperoxic exposure during hyperbaric therapy for the treatment of carbon monoxide poisoning in pregnant patients can be tolerated by the fetus in all stages of pregnancy. 31
There are no studies to suggest whether vaginal delivery or cesarean section is more ideal for patients with methemoglobinemia. However, in a hemodynamically stable patient with no evidence of hypoxia and with a normal fetal heart rate tracing, cesarean section should be reserved for traditional obstetric indications.
Conclusion
For patients with known congenital methemoglobinemia, coordination of care efforts between Hematology, Obstetrics, Maternal-Fetal Medicine, and Anesthesia is imperative to create a multidisciplinary plan for pregnancy and delivery that limits exposures to oxidizing agents. Patients should additionally be adequately counseled on drugs and environmental exposures to avoid to decrease the risk of methemoglobinemia exacerbation. As the percentage of MHb generally correlates to symptom severity, clinicians may want to trend MHb levels to determine the efficacy of a chosen treatment or to ascertain the need for more aggressive treatment. Our case report suggests that the use of oral ascorbic acid may be a reasonable alternative to methylene blue during pregnancy in gravidas with symptomatic methemoglobinemia.
Footnotes
Acknowledgements
We would like to express our gratitude to the clinical staff at Cooper University Hospital for their exceptional care of the patient described in this case report. We also thank our colleagues for their insightful discussions and contributions to the development of this manuscript.
Author Note
Prior Presentation of Abstract Statement: This abstract was not previously presented at a meeting.
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
Written informed consent was obtained from the patient to participate in the clinical evaluation and management described in this case report.
Consent for Publication
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
