Abstract
Wellens’ syndrome is characterized by a distinct electrocardiographic pattern, most notably biphasic or deeply inverted T waves in the anterior precordial leads, particularly V2 and V3. These findings typically reflect transient myocardial ischemia resulting from critical stenosis of the proximal left anterior descending (LAD) artery. They are often a warning sign of an impending anterior wall myocardial infarction. However, conditions such as pseudo-Wellens’ syndrome can exhibit similar electrocardiogram (ECG) changes without LAD occlusion. In this case report, we describe a 71-year-old woman who presented with chest discomfort and dyspnea and was ultimately diagnosed with sepsis-induced cardiomyopathy, which produced ECG changes mimicking Wellens’ syndrome despite the absence of coronary artery disease; cardiac catheterization found no significant coronary pathology. The diagnosis was determined to be stress-induced cardiomyopathy resulting from sepsis. This report highlights the importance of distinguishing between ischemic and nonischemic causes of similar ECG patterns. It emphasizes the necessity for careful diagnosis and management, particularly in complex situations like sepsis.
Keywords
Introduction
Wellens’ syndrome is typically characterized by chest pain along with specific electrocardiogram (ECG) changes, including biphasic or deeply inverted T waves in leads V2 and V3.1,2 This ECG pattern indicates transient ischemia due to significant narrowing in the proximal left anterior descending (LAD) artery, leading to abnormalities in cardiac repolarization and indicating a heightened risk of imminent anterior myocardial infarction. 2 This syndrome serves as a crucial indicator for urgent coronary revascularization and antiplatelet therapy. However, similar ECG changes can also occur in conditions that mimic Wellens’ syndrome, known as pseudo-Wellens’ syndrome, which occurs without LAD occlusion. We describe a 71-year-old woman who developed sepsis-induced cardiomyopathy, presenting with electrocardiographic features mimicking Wellens syndrome in the absence of obstructive coronary artery disease (CAD). This case highlights the difficulty of distinguishing between ischemic and nonischemic causes of similar ECG patterns to guide appropriate clinical management. It shows the importance of careful interpretation in critically ill patients.
Case Presentation
A 71-year-old female with multiple comorbidities, including chronic kidney disease, asthma, pulmonary embolism (on Apixaban), and ovarian cancer, presented to the emergency department complaining of progressive dyspnea and chest heaviness. She was hypoxic and tachycardic upon arrival at the emergency department. Initial evaluation revealed bilateral pulmonary crackles, pulmonary congestion on chest X-ray, leukocytosis, elevated procalcitonin levels, a B-type Natriuretic Peptide of 2271 (reference range 1-100 pg/mL), and a Troponin of 39 (reference range 3-17 pg/mL). A Computed Tomography angiography (CTA) revealed a pulmonary embolism in the right lower lobe without evidence of right heart strain. Anticoagulation with a heparin infusion and diuretic therapy was initiated; however, the patient ultimately required intubation due to worsening hypoxic respiratory failure.
While in the cardiac care unit, she developed new-onset chest pain and ECG changes, showing biphasic T-wave inversions in V2 and V3 leads—findings suggestive of Wellens’ phenocopy, which demonstrated normal R-wave progression and no pathological Q waves (Figure 1). A transthoracic echocardiogram was also done and showed a severely decreased left ventricular ejection fraction of 20% to 25%, grade II diastolic dysfunction, severely dilated left atrium, and mild mitral valve regurgitation. Cardiac catheterization was done and showed no coronary stenosis but confirmed reduced ejection fraction (20%; Figure 2). A positive Legionella pneumophila PCR confirmed an atypical pneumonia; the patient was managed with intravenous azithromycin alongside guideline-directed heart failure therapy. She improved clinically and was discharged in stable condition.

EKG showing biphasic T-wave inversion in leads V2 and V3 (red arrows), a pattern consistent with Wellens’ phenocopy.
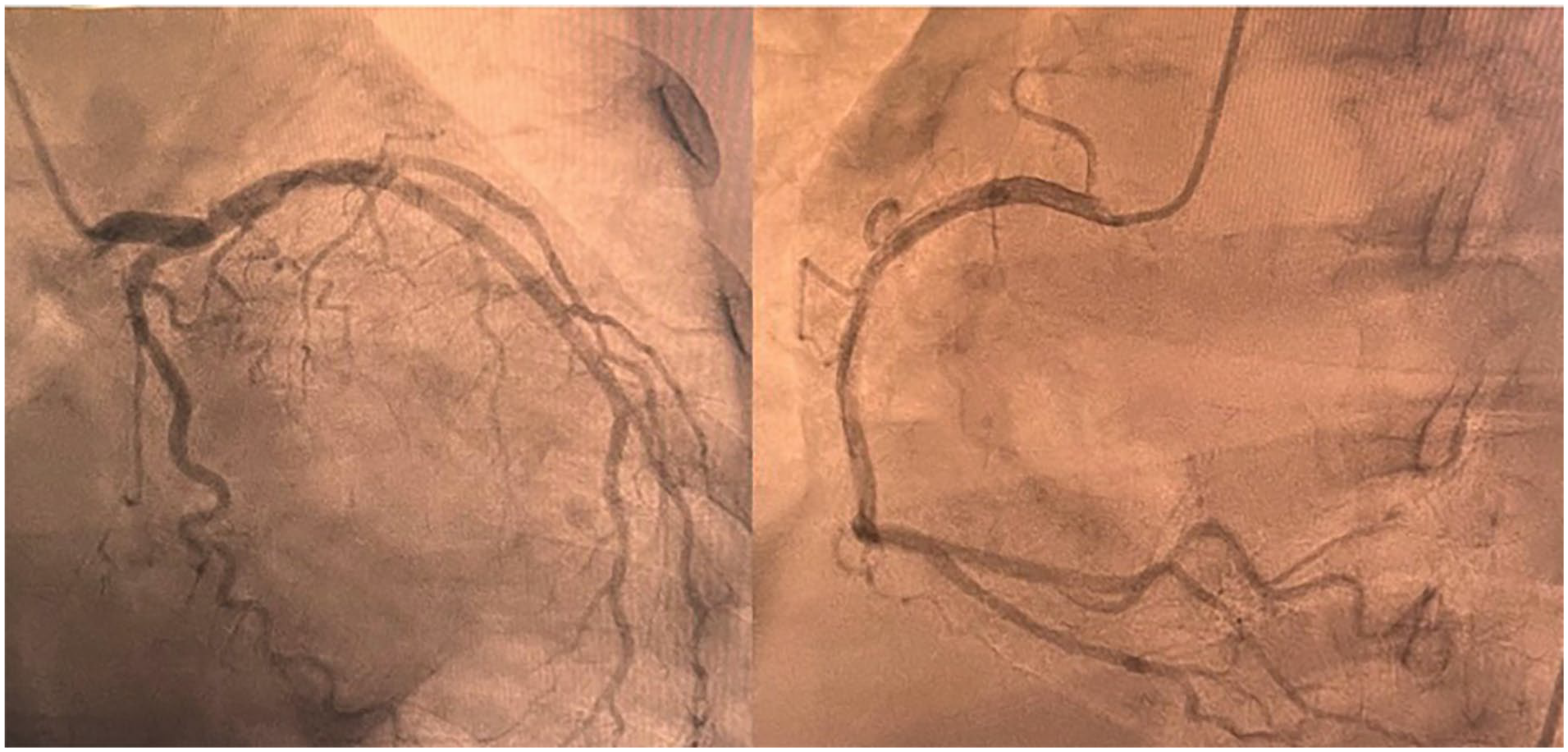
Coronary angiogram showing no coronary occlusion.
Discussion
Wellens’ syndrome is a well-recognized marker of significant proximal stenosis of the LAD artery, particularly in patients who present with symptoms of unstable angina. 2 This syndrome was first described in 1982 when de Zwaan and Wellens’ published a case series of 145 patients with unstable angina, 26 of whom (18%) exhibited similar ECG findings. These findings included an isoelectric or minimally elevated ST segment (1 mm) in leads V2 and V3. 1 The ECG pattern was described as originating from the QRS complex with either a straight or mildly concave ST segment, leading into a sharply angled and symmetrical T-wave inversion. 1 In a later prospective study involving patients with unstable angina, de Zwaan et al found that 128 of 1260 individuals exhibiting this type of ECG abnormality were confirmed to have significant narrowing of the proximal LAD artery. 3
Today, the diagnostic criteria for Wellens’ syndrome include the following: a history of anginal chest pain, minimal or no elevation of cardiac enzymes, and no significant ST-segment elevation (<1 mm). Additionally, there should be an absence of pathological precordial Q waves, no loss of precordial R-wave progression, and the presence of profoundly inverted or biphasic T waves. These T wave abnormalities are primarily observed in leads V2 and V3, and may occasionally appear in leads V1, V4, V5, and V6.2,4
Typically, Wellens’ syndrome results from the rupture of an atherosclerotic plaque, leading to an obstruction of the LAD artery. This is often followed by clot lysis or other disruptions of the blockage before a complete myocardial infarction occurs. As a preinfarction condition, Wellens’ syndrome signifies an unstable coronary perfusion state, increasing the risk of major anterior infarction and serious clinical deterioration. 5
In contrast, pseudo-Wellens’ syndrome is a term used to describe a clinical feature and ECG changes that closely resemble those of classic Wellens’ syndrome. However, it occurs due to temporary disruptions in coronary blood flow rather than the severe fixed stenosis of the LAD artery. Nevertheless, it still presents with similar ECG changes, making it crucial to differentiate between them for accurate diagnosis and appropriate management. 6
This temporary obstruction can result from various conditions, including cocaine use, pulmonary embolism, stress-induced cardiomyopathy, and uncontrolled hypertension.7 -11
In the literature review, pseudo-Wellens’ syndrome involved patients ranging in age from 22 to 81 years, averaging around 50, with most cases presenting with chest pain that lasted from a few hours to a few months, accompanied by transient ECG changes that resolved alongside symptom improvement, and angiography that was unremarkable. 7
The electrophysiological basis of pseudo-Wellens’ syndrome remains an active area of research. Some consider intermittent vasospasm causing transient myocardial ischemia and ECG changes like the cocaine effect. 9 While others have suggested that myocardial edema, rather than ischemia, may be responsible for the Wellens-like ECG pattern, irrespective of the underlying cause. 12
In our case, the development of Stress-induced cardiomyopathy secondary to sepsis, with a subsequent massive release of pro-inflammatory cytokines, likely contributes to myocardial edema, which can serve as the underlying mechanism for both cardiomyopathy and pseudo-Wellens’ syndrome. 13 Sepsis-induced cardiomyopathy is a complex condition resulting from the systemic inflammatory response to infection, where a cytokine storm plays a central role in myocardial dysfunction. This cytokine release can lead to mitochondrial dysfunction, which impairs cellular energy production and disrupts normal myocardial contraction and relaxation, resulting in a range of abnormalities in myocardial function that may present as global or regional wall motion abnormalities observed during systole and diastole on echocardiography.7,14
These abnormalities are often reflective of myocardial edema and impaired contractility rather than an issue stemming from obstructive CAD. Interestingly, although the pattern of the ECG in pseudo-Wellens’ syndrome can resemble that of true Wellens’ syndrome, it is often not accompanied by the critical stenosis of the LAD artery.
While it is crucial to assess underlying CAD, the presence of ECG changes mimicking Wellens’ syndrome in the setting of sepsis should prompt clinicians to consider stress-induced cardiomyopathy as a possible etiology. Given the unstable nature of sepsis-induced myocardial dysfunction, patients may remain at considerable risk for major cardiac events even in the absence of obstructive CAD. Early recognition of this condition, along with careful management of the underlying infection and appropriate cardiac monitoring, is essential. Physicians should consider early cardiac catheterization when there is suspicion of CAD, particularly in patients with severe septic shock or those showing ECG patterns suggestive of Wellens’ syndrome, to rule out LAD stenosis or occlusion.
To conclude, while Wellens’ syndrome is a critical indicator of proximal LAD stenosis and impending myocardial infarction, pseudo-Wellens’ syndrome, including cases due to sepsis-induced cardiomyopathy, underscores the importance of differentiating between ischemic and nonischemic causes of ECG changes. Early recognition and careful management of underlying conditions, such as sepsis, are essential to prevent adverse outcomes and ensure appropriate treatment. Timely diagnostic interventions, including coronary angiography, when necessary, remain crucial for optimal patient care.
Conclusion
In conclusion, Wellens’ syndrome is a critical indicator of significant proximal LAD coronary artery stenosis and a potential myocardial infarction, necessitating prompt intervention. On the other hand, pseudo-Wellens’ syndrome, which can be caused by conditions such as sepsis-induced cardiomyopathy, underscores the importance of distinguishing between ischemic and nonischemic causes of ECG changes. It is essential to identify the underlying conditions like sepsis early and manage them appropriately to avoid misdiagnosis and adverse outcomes. When necessary, timely diagnostic evaluations, including coronary angiography, are crucial for providing the best possible care for patients with these complex presentations.
Footnotes
Acknowledgements
We thank the patient and her family for allowing us to share this case with our colleagues.
Author Note
All authors, including the corresponding author, may be reached using the contact information above.
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
As this is a case report, consent was obtained for this paper.
Consent for Publication
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Author Contributions
All authors performed the literature review, and all authors contributed to the writing, the final editing, and the patient’s clinical data collection. All work was performed at St Joseph’s University Medical Center at the following address.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
