Abstract
Coronary-subclavian steal syndrome (CSSS) is a rare but important complication following coronary artery bypass grafting (CABG) involving the left internal mammary artery (LIMA), typically due to proximal subclavian artery stenosis. We present a 54-year-old male with prior triple-vessel CABG (LIMA to left anterior descending) who developed acute chest pain and elevated troponin levels. Electrocardiogram showed diffuse ST-segment changes. Emergent angiography revealed patent grafts but critical 90% stenosis of the left subclavian artery proximal to the LIMA origin. The lesion was successfully treated with percutaneous angioplasty and drug-eluting stent placement, resulting in the resolution of symptoms and preserved cardiac function. CSSS, though uncommon, should be considered in post-CABG patients with recurrent angina or myocardial injury despite patent grafts. Literature estimates subclavian stenosis in up to 5% of CABG candidates, yet routine screening remains inconsistent. This case highlights CSSS as a reversible cause of ischemia and underscores the value of targeted vascular imaging in selected patients.
Keywords
Case Presentation
The patient is a 54-year-old male with a past medical history of coronary artery disease (CAD) status post coronary artery bypass grafting (CABG) ×3, left internal mammary artery (LIMA) to left anterior descending (LAD), saphenous vein graft (SVG) to posterior descending artery occluded and SVG to obtuse marginal artery 10 years ago, hypertension, dyslipidemia, prior substance use (currently on naltrexone), and a daily smoking habit. He presented with acute substernal crushing chest pain and was transferred from an outside facility. On arrival, his troponin levels were significantly elevated, initially trending at 8, 280, and 2399, and later exceeding 25 000. His electrocardiogram (EKG) showed sinus rhythm with ST depressions in leads II, III, aVF, and V4 to V6, along with ST elevations in V1 (1.5 mm) and aVR (1 mm; Figure 1). He was given morphine, aspirin, Plavix, atorvastatin, nitroglycerin, metoprolol, and Lovenox to stabilize him and was transferred to our facility for further management due to the severity of his troponin elevation and persistent chest pain.

EKG showing ST depressions in leads II, III, aVF, and V4 to V6, along with ST elevations in V1 (1.5 mm) and aVR (1 mm). EKG, electrocardiogram.
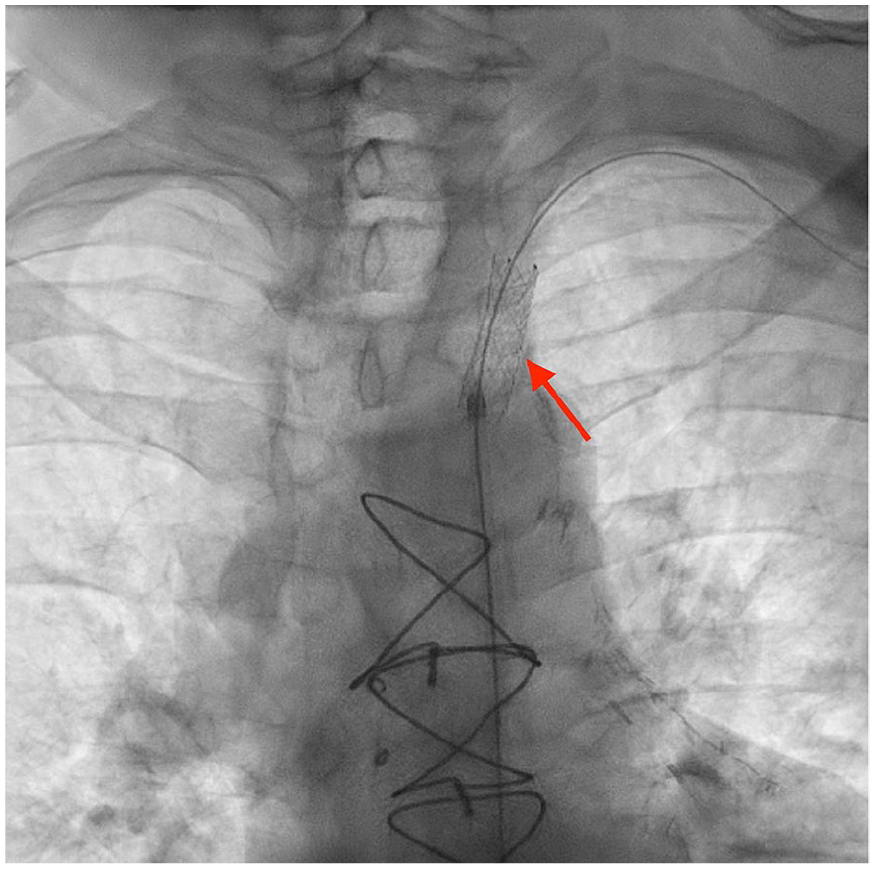
Upon arrival at our facility, the patient continued to experience chest pain, though his vital signs were stable with a pulse of 98 bpm, blood pressure of 127/103 mmHg, respiratory rate of 18, and oxygen saturation of 97%. The EKG revealed sinus tachycardia at a rate of 103 bpm, with resolution of the inferior ST depressions, improvement in lateral ST depressions, and a decrease in the ST elevations in V1 and aVR. Given the elevated troponin and ongoing ischemia, he was started on a heparin drip and immediately taken to the catheterization laboratory. Coronary angiography revealed severe CAD with adequate revascularization via grafts (Figure 2, red arrow, Figures 5-7), However, it also showed 90% stenosis of the left subclavian artery (Figure 2, blue arrow), which was successfully treated with percutaneous transluminal angioplasty and the placement of a drug-eluting stent (Figure 3), leading to the resolution of the stenosis and chest pain (Figure 4).

Angiographic image showing 90% proximal left subclavian artery stenosis (blue arrow) and patent LIMA to LAD graft (red arrow). LAD, left anterior descending; LIMA, left internal mammary artery.
Coronary angiography showing successful placement of drug-eluting stent (red arrow).

Coronary angiography showing resolution of left proximal artery stenosis.

Coronary angiography showing Patent RCA.

Coronary angiography Patent SVG to Obtuse Marginal.

Angiographic image demonstrating Patent LAD.
An echocardiogram performed post-procedure revealed a normal ejection fraction of 60% and normal global systolic function, confirming that there was no significant myocardial damage. He was discharged on dual antiplatelet therapy and scheduled for outpatient follow-up. At his 2nd week and 3rd month follow-up visits, he reported no recurrence of symptoms and was recovering well, indicating a favorable clinical outcome following prompt identification and treatment of subclavian artery stenosis (SAS) as the underlying cause of his ischemia.
Discussion
Coronary-subclavian steal syndrome (CSSS) is a rare complication following CABG, where blood flow is diverted from the coronary artery to the subclavian artery through the LIMA, leading to myocardial ischemia. 1
Approximately 5% of patients undergoing CABG have SAS2,3 and myocardial ischemia after coronary revascularization (0.1%-6%) of cases. 4
In CABG, the LIMA is typically left attached to the left subclavian artery, with its distal end used to bypass a blocked coronary artery. However, significant stenosis of the left subclavian artery can impair blood flow to the LIMA, causing CSSS. This leads to graft failure, with blood diverted away from the myocardium through collateral circulation, resulting in angina, acute coronary syndrome, heart failure, and arrhythmias.5-9
CSSS should be considered in patients with a history of LIMA-LAD bypass who present with angina, heart failure, or myocardial infarction, particularly when symptoms are triggered by exertion of the ipsilateral arm or there is a significant inter-arm systolic blood pressure difference (>15-20 mmHg).10-12
Clinical findings may include upper extremity claudication (fatigue, weakness, or pain), diminished or delayed pulses in the affected arm, and the presence of a supraclavicular or infraclavicular bruit. 10 Acute coronary syndromes, including myocardial infarction, can occur when subclavian stenosis is severe or rapidly progressive. In some cases, impaired LIMA graft flow may also lead to acute systolic heart failure, particularly with large myocardial territories or poor collateral supply.11,12
Risk factors include peripheral arterial disease, prior or concurrent carotid or lower extremity atherosclerosis, and traditional cardiovascular risks such as advanced age, hypertension, diabetes, hyperlipidemia, and tobacco use.13,14
Diagnosis is based on clinical suspicion and physical examination, with particular attention to inter-arm blood pressure difference. Duplex ultrasonography can detect flow reversal, while CT angiography, MR angiography, or conventional angiography confirms subclavian stenosis and retrograde LIMA flow. 10
Currently, there are no established guidelines for routine preoperative or postoperative screening for SAS in patients undergoing CABG with an internal mammary artery (IMA) graft. Although SAS can potentially impair blood flow through the IMA and jeopardize graft patency, its impact on CABG outcomes has not been extensively studied, leading to a lack of emphasis on routine screening for this condition. However, measuring bilateral brachial blood pressures during annual follow-up visits after CABG is crucial for detecting the possible development of SAS. The gold standard for diagnosing SAS remains selective subclavian arteriography. 15
Management involves revascularization of the subclavian artery in symptomatic patients, with endovascular stenting preferred due to high success and low complication rates. 16 Surgical options are reserved for cases where stenting is not feasible or effective. The decision between these methods is influenced by factors such as the patient’s clinical stability, age, and presence of comorbidities.15,17 Asymptomatic patients can be managed conservatively unless future LIMA use is planned. 16
Ongoing follow-up with clinical assessment and imaging is essential to monitor for restenosis. Reappearance of symptoms or evidence of restenosis should prompt timely intervention to maintain graft patency and prevent adverse cardiac events.10,13,14
Conclusion
In conclusion, CSSS is a rare but important complication following CABG with IMA grafts, often caused by SAS. While routine screening for this condition is not well-established, high-risk patients should be monitored with bilateral brachial blood pressure measurements. Selective subclavian arteriography is the gold standard for diagnosis, and percutaneous stenting may be an effective treatment. Clinicians should have a high index of suspicion for CSSS in patients with a history of CABG or peripheral artery disease presenting with chest pain. Further guidelines are needed for routine screening and management of CSSS.
Footnotes
Ethical Considerations
Our institution does not require ethics approval for reporting individual cases or case series.
Consent for Publication
Verbal informed consent to participate was obtained from the patient for their anonymized information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
