Abstract
Systemic lupus erythematosus (SLE) is an autoimmune disease targeting multiple organs through immune complexes. Pericarditis and pericardial effusions are often encountered; however, cases of biventricular failure are notably less common in the setting of SLE. We report a 43-year-old male patient presenting with cardiac tamponade with biventricular failure in SLE. Our patient with known SLE, not on medication, presented with exertional dyspnea. He had decreased heart sounds, jugular venous distention, and right-sided inspiratory crackles on physical examination. On further investigation, he was found to have a reduced ejection fraction (EF) of 10% to 15% and moderate pericardial effusion. He was treated with immunosuppressive therapy and goal directed medical theraphy (GDMT), with improvement in his EF to 58% 3 months later. This case demonstrates the importance of early recognition of SLE-induced tamponade and the vitality of prompt medical therapy to reduce any further cardiac deterioration.
Introduction
Systemic lupus erythematous (SLE) is a multisystemic autoimmune disease that can vary in presentation. The cardiac manifestations of SLE can affect all layers of the heart and include pericarditis, myocarditis, endocarditis, and coronary artery disease. Pericarditis is the most common cardiac manifestation, presenting with chest pain and dyspnea. Pericardial effusion, often seen with pericarditis, can lead to cardiac tamponade, a life-threatening condition characterized by fluid accumulation in the pericardial sac compressing the heart. 1 Diagnosis is confirmed via echocardiography, revealing effusion and potential signs of tamponade, such as right atrial collapse. Myocarditis, inflammation of the heart muscle, can cause heart failure, arrhythmias, presenting with symptoms such as chest pain, fatigue, and dyspnea. 1 Echocardiography and cardiac magnetic resonance imaging (MRI) are essential for diagnosis, showing myocardial inflammation and dysfunction. Libman-Sacks endocarditis is a nonbacterial form of endocarditis seen in SLE, involving the formation of sterile vegetations on heart valves. It can lead to valvular dysfunction and embolic events.1 Diagnosis is made using echocardiography, revealing valvular thickening and vegetations. Systemic lupus erythematous also increases the risk of coronary artery disease due to chronic inflammation and accelerated atherosclerosis. Patients may present with typical angina, myocardial infarction, or sudden cardiac death. 1 Risk factors for SLE-related cardiac complications include prolonged disease activity, inadequate control of lupus, renal disease, higher levels of inflammation, and complement depletion.
Case Presentation
A 43-year-old man with a 12-year history of systemic lupus erythematosus, who had not been on medication or seen a rheumatologist for the past 5 years, presented with exertional dyspnea for 6 weeks. He reported orthopnea, intermittent skipped heartbeats, generalized weakness, decreased appetite, and a productive cough. Physical examination revealed decreased heart sounds, a soft systolic murmur, jugular venous distention, and right-sided inspiratory crackles. Vital signs showed blood pressure of 106/71 mm Hg, heart rate of 108 beats per minute, oxygen saturation of 97% on room air, and he was afebrile.
Laboratory findings were notable for an elevated brain natriuretic peptide of 1240 pg/mL (reference <100 pg/mL), elevated erythrocyte sedimentation rate of 75 mm/h (reference <20 mm/h), anti-double-stranded DNA antibodies of 34 IU/mL (reference <30 IU/mL), blood urea nitrogen of 35 mg/dL (reference 7-20 mg/dL), creatinine of 2.04 mg/dL (reference 0.6-1.3 mg/dL), decreased estimated glomerular filtration rate of 36 mL/min/1.73 m² (reference >90 mL/min/1.73 m²), positive antinuclear antibodies, and 3+ proteinuria on urinalysis. A chest x-ray revealed an enlarged cardiac silhouette due to pericardial effusion and right lower lobe infiltrates. His electrocardiogram showed sinus tachycardia. Transthoracic echocardiogram (TTE) demonstrated a reduced ejection fraction (EF) of 21%, severely decreased global wall motion, large pericardial effusion (Figure 1) and buckling of the right atrium, and early diastolic inversion after the T wave suggesting impending tamponade (Figure 2). Computed tomography of the thorax confirmed moderate pericardial effusion, trace right pleural effusion, and patchy opacities in the right upper and lower lobes (Figure 3). The patient received 20 mg intravenous furosemide for fluid overload but repeat TTE showed an enlarging pericardial effusion. He underwent a therapeutic pericardial window with intraoperative transesophageal echocardiogram, which found 350 mL of serous fluid, a normal-appearing pericardium, and a severely reduced biventricular EF of 10% to 15%. Treatment included high-dose corticosteroids (125 mg methylprednisolone), ceftriaxone, azithromycin, metoprolol succinate 12.5 mg, and azathioprine 50 mg twice daily. A renal biopsy revealed class IV lupus nephritis, leading to a switch from azathioprine to mycophenolate 500 mg twice daily for a week, then 1000 mg twice daily. The patient experienced persistent sinus tachycardia with heart rates in the 110s and a drop in blood pressure to systolic 80-90 mm Hg. Swan-Ganz catheterization showed markedly elevated intracardiac filling pressures, a thermodilution cardiac output of 5.1 L/min, a cardiac index of 2.6 L/min/m², an arterial blood gas saturation of 97%, and a pulmonary artery saturation of 72.5%. Calculated cardiac output/cardiac index (Fick’s formula) was 7.3/3.6. Dobutamine was withheld due to the absence of cardiogenic shock, and a Lasix drip was started, later tapered to 80 mg twice daily and then 60 mg twice daily. Follow-up chest x-ray showed resolution of fluid overload and infiltrates, and infectious workup returned negative, leading to the discontinuation of antibiotics. Lasix was held due to worsening kidney function and euvolemic status. Bactrim was initiated for pneumocystis pneumonia prophylaxis. Guideline-directed medical therapy included hydralazine 25 mg 3 times daily, isosorbide dinitrate 10 mg 3 times daily, metoprolol succinate 25 mg, and dapagliflozin 10 mg. The patient was not a candidate for angiotensin receptor-neprilysin inhibitors or mineralocorticoid receptor antagonists due to lupus nephritis and acute kidney injury. He was provided with a LifeVest and discharged. At 1-, 2-, and 3-week follow-ups, he showed clinical improvement and remained asymptomatic. Three months later, the repeat TTE was significant for EF of 58%.

TTE showing pericardial effusion.
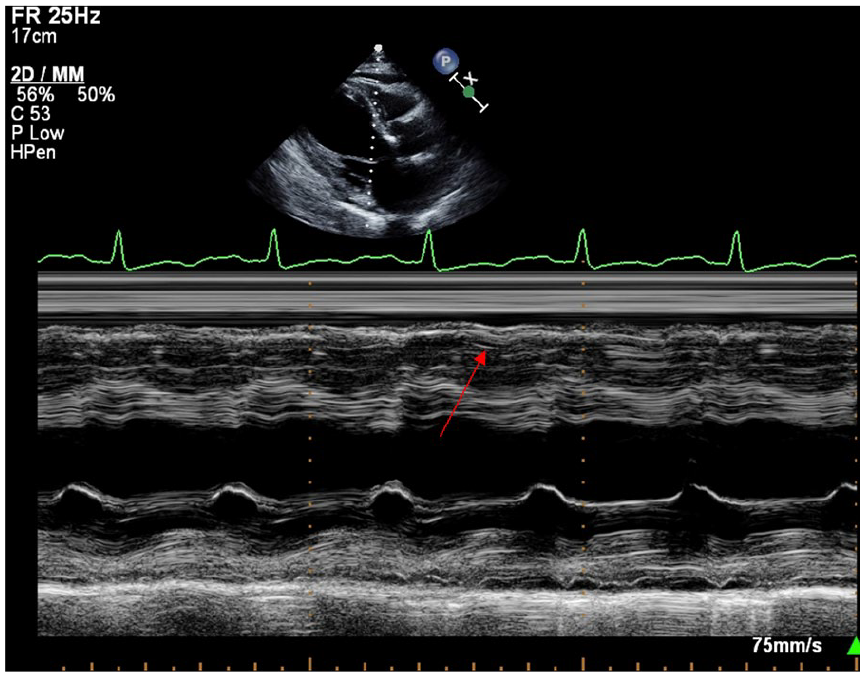
TTE showing early diastolic inversion after the T wave.

CT thorax without contrast showing pericardial effusion.
Discussion
Cardiac tamponade occurs in less than 1% of patients with SLE. 2 In those with known SLE, those who are more predisposed to developing tamponade are women and those with anemia, renal disease, pleuritis, higher erythrocyte sedimentation rate (ESR) levels, and lower C4 levels. 2 In a study conducted by Goswami et al, 3 of 409 patients with SLE, tamponade was seen in 24 patients (5.9%), and it was the presenting feature for 12 of those patients (50%).Further analysis found that pleuritis, anti-nucleosome antibody, and size of pericardial effusion are predictors for cardiac tamponade. 3
Diagnosis of cardiac tamponade in SLE involves clinical evaluation for symptoms such as dyspnea, chest pain, and signs of hemodynamic instability with hypotension, tachycardia, and physical examination findings of elevated jugular venous distention, diminished heart sounds, pulsus paradoxus (exaggerated decrease in systolic blood pressure [more than 10 mm Hg] during inspiration). Echocardiography is the primary diagnostic tool, revealing pericardial effusion and signs of cardiac compression. Immediate treatment includes pericardiocentesis or a surgical pericardial window to relieve pressure on the heart. High-dose corticosteroids are administered to reduce inflammation, and additional immunosuppressive therapy may be necessary to manage underlying lupus. Immunosuppression in higher doses, such as IV steroids and cyclophosphamide, may reduce the need for surgical intervention.3 Monitoring for recurrent effusion and continuous management of lupus activity are essential to prevent further cardiac complications.
Systemic lupus erythematosus can lead to heart failure through multiple mechanisms, including myocarditis, cardiomyopathy, and valvular disease. Myocarditis may involve long-standing inflammation of the heart muscle, leading to impaired contraction due to fibrosis of the myocardium. In addition, Libman-Sacks endocarditis, a nonbacterial form of endocarditis associated with SLE, can cause valvular dysfunction and contribute to heart failure. Although exact pathogenesis is unknown, autoimmunity, chronic inflammation, and traditional cardiovascular risk factors play a critical role for heart failure in the setting of SLE. 4 Echocardiography is the primary imaging modality used to assess cardiac function in SLE patients. It can identify ventricular dysfunction, myocardial inflammation, valvular abnormalities, and pericardial effusion. Doppler imaging can evaluate diastolic function and detect elevated filling pressures indicative of heart failure. Cardiac MRI provides detailed images of myocardial tissue, allowing for the detection of inflammation, fibrosis, and scarring. It is particularly useful in diagnosing myocarditis and assessing the extent of myocardial involvement.
Treatment involves managing the underlying SLE with immunosuppressants and addressing the cardiac manifestations. For heart failure, guideline-directed medical therapy is applied, including the use of diuretics, angiotensin-converting enzyme inhibitors, or beta-blockers. Controlling underlying inflammation with cytotoxic therapy significantly improves left ventricular function in SLE patients with heart failure. 4 Over 10 years, Naarendorp et al observed 6 SLE patients who, after undergoing cytotoxic therapy, showed a substantial increase in left ventricular EFs. Initially presenting with EFs between 11% and 34%, these patients improved to 25% to 55% within 6 months. Our patient’s EF also improved from 21% to 58% in 3 months following cytotoxic therapy. 5
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
Abstract of this case was presented as titled “Heart Under Siege: Navigating Cardiac Tamponade with Biventricular Heart Failure in the Setting of Systemic Lupus Erythematosus” on April 7, 2024 at 73rd American College of Cardiology 2024 Annual Meeting in Atlanta, GA, USA.
