Abstract
In this case report, we describe a case of double-positive rapidly progressive glomerulonephritis among the Lebanese population. The patient, a 44-year-old female, presented with anemia and a recurrent right middle lobe pneumonia. She rapidly developed renal failure with her autoimmune workup being positive for both anti-glomerular basement membrane antibodies and anti-neutrophil cytoplasmic antibodies. The patient was successfully treated with plasmapheresis, intravenous cyclophosphamide, and oral prednisone. The patient regained her normal renal function and did not depend on dialysis for renal and overall survival. At 6-month follow-up, the patient is in remission on a steroids taper. As treatment guidelines are still not well established for such unique presentations, this report highlights the importance of early intervention and combined therapy to treat double-positive rapidly progressive glomerulonephritis.
Keywords
Introduction
In 1919, Ernest Goodpasture described an atypical case of fatal influenza characterized by alveolar wall hemorrhage associated with a glomerular nephropathy with fibrin exudates. 1 Currently, Goodpasture syndrome and anti-glomerular basement membrane (anti-GBM) disease are terms that are used interchangeably to refer to a small vessel vasculitis in which antibodies are produced against the alpha-3 chain of type IV collagen mainly found in the alveolar and GBMs. When these antibodies target type IV collagen, the result is alveolar hemorrhage and rapidly progressive glomerulonephritis that are seen in anti-GBM disease. 2 To date, there is no clear etiology behind anti-GBM disease. However, an 11-year nationwide study in Ireland concluded that the incidence of anti-GBM disease was clustered in time and space, possibly being linked to an unknown environmental trigger. 3 The study also calculated an incidence of around 1.6 cases/million/year. 3 A recent observational study in London, United Kingdom, found an increased incidence of anti-GBM disease between 2019 and 2020 possibly related to the severe acute respiratory syndrome—coronavirus 2 pandemic and the potential of such infections triggering autoimmune diseases such as anti-GBM disease. 4 Our case report discusses a patient with the double-positive variant of anti-GBM disease, that is, also positive for anti-neutrophil cytoplasmic antibodies (ANCAs) and presents with rather more systemic signs compared to the typical picture of hemoptysis and hematuria that are seen in anti-GBM disease.2,5
Case Presentation
A 44-year-old, previously healthy, Lebanese female presented to the emergency department of our hospital for a fever of 2 weeks duration reaching 40 °C and that was relieved with antipyretics. The fever was associated with a dry cough, chills, fatigue, night sweats, loss of appetite, and weight loss. The patient reported a recurring right middle lobe pneumonia despite multiple courses of antibiotics over the past year and requiring multiple hospital admissions. During her last hospitalization, she was found to have anemia and normal renal function (creatinine 0.9 mg/dL).
On admission, the patient was febrile (39 °C), hemodynamically stable. On physical examination, she had good bilateral air entry with no wheezes and a soft but diffusely tender abdomen with hepatosplenomegaly. The patient’s initial laboratory investigations at presentation were pertinent for anemia (hemoglobin 5.4 g/dL), kidney injury (creatinine 1.6 mg/dL), elevated procalcitonin (0.438 ng/mL), and C-reactive protein (233 mg/L). Urine analysis showed mildly elevated red blood cells (RBCs) in the urine (6-10 cells/high-power field).
A computed tomography (CT) scan of the chest without contrast revealed a right middle lobe lateral consolidation and infiltrates which were improving compared to her previous scans. The patient was admitted to the medicine floor for pneumonia and severe anemia, blood cultures were taken. She was started on Piperacillin/Tazobactam 2.25 g intravenous (IV) every 6 hours along with supportive management awaiting primary investigation. The patient was admitted with a hemoglobin of 5.4 g/dL and received a total of 4 units of packed RBCs over a period of 3 days to target a hemoglobin level above 7 g/dL, while anemia workup was still pending. Results did not show any bleeding, iron deficiency, or hemolysis.
On the fourth day of admission, the patient developed dyspnea, desaturation, oliguria with bilateral lower limb and facial edema, and a progressive decline in renal function. Therefore, she was transferred to the intensive care unit (ICU) and a CT scan of the thorax, abdomen, and pelvis without IV contrast was performed. The scan showed a decrease in the right middle lobe consolidation, bilateral basilar pleural effusions, hepatomegaly, and diffuse abdominal and pelvic ascites.
In the ICU, she received appropriate management with IV steroids (methylprednisolone 350 mg/day) for suspected glomerulonephritis, albumin, and furosemide. The results of her initial diagnostic autoimmune workup taken on fifth day of admission are summarized in Table 1, which were strongly positive for anti-GBM (level in serum = 119) and myeloperoxidase (MPO)-ANCAs (level in serum = 81). Elevated urine protein was also evidenced on urine analysis (Table 1).
Summary of the Results From the Autoimmune Workup.

Abbreviations: ANA, antinuclear antibodies; anti-dsDNA, anti-double-stranded deoxyribonucleic acid antibodies; anti-GBM, antiglomerular basement membrane antibodies; c-ANCA, cytoplasmic anti-neutrophil cytoplasmic antibodies; IU, international units; MPO, myeloperoxidase; p-ANCA, perinuclear anti-neutrophil cytoplasmic antibodies; PR3, proteinase 3.
Based on the clinical presentation and laboratory investigations, the diagnosis was suggestive of a double-positive rapidly progressive glomerulonephritis (anti-GBM and ANCA-associated disease). Subsequently, an ultrasound-guided renal biopsy was performed on eighth day of admission and the patient started plasmapheresis and received pulse steroids with 1 g of methylprednisolone/day for 3 days and then switched to daily oral prednisone 1 mg/kg. The patient received 3 doses of cyclophosphamide 400 mg IV every 8 hours, on the fifth day of plasmapheresis. The patient completed a total of 20 plasmapheresis sessions over 25 days.
Figure 1 is a graph that summarizes the progression of the patient’s renal function through serial monitoring of her serum creatinine, anti-GBM, and MPO-ANCA levels during the period of treatment.
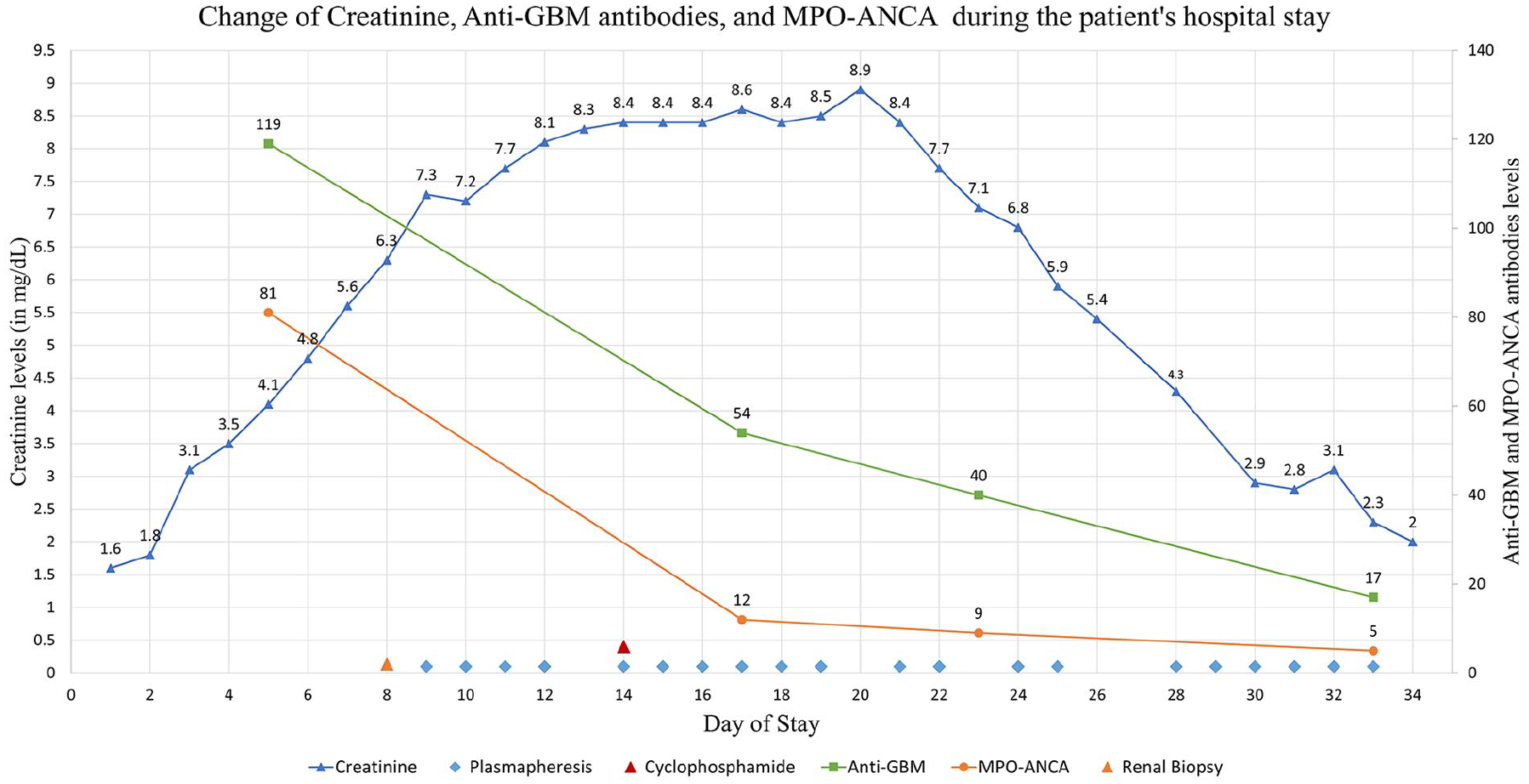
Graph showing the progression of the patient’s disease and kidney function assessed through serial measurement of serum creatinine (mg/dL) during her hospital stay. Anti-GBM antibodies and MPO-ANCA were also monitored to assess Goodpasture syndrome response to treatment. Blue triangle, serum creatinine levels; light blue rhombus, plasmapheresis day; red triangle, intravenous cyclophosphamide day; green square, serum anti-GBM levels; orange circle, serum MPO-ANCA levels; orange triangle, renal biopsy day. Anti-GBM, antiglomerular basement membrane antibodies; MPO-ANCA, myeloperoxidase-antineutrophil cytoplasmic antibodies; admitted on day 1, discharged on day 34.
Results of the biopsy revealed extra-capillary crescentic glomerulonephritis. The immunofluorescence was not in favor of Goodpasture syndrome but showed positive C3 granular and membranous deposits with focally positive fibrin and negative immunoglobulin A (IgA), IgG, and IgM immunofluorescence.
The patient’s clinical status gradually improved, her pulmonary congestion and pleural effusions as well as her peripheral and facial edema gradually resolved throughout her treatment. However, her renal function took longer to improve, as creatinine levels peaked at 8.9 mg/dL 12 days after beginning plasmapheresis but then gradually improved and reached a level of 2 mg/dL upon discharge. The patient maintained an adequate urine output, had no uremic symptoms throughout this period of elevated creatinine, and did not require hemodialysis. Additionally, both anti-GBM antibodies and MPO-ANCA levels markedly decreased over the course of her treatment.
The patient was hospitalized for a total of 34 days and was discharged on oral maintenance prednisone 50 mg/day. On follow-up 3 months postdischarge, the patient had a stable renal function with a creatinine of 1.2 mg/dL. At 6-month follow-up, the patient’s anti-GBM level is 0.9, and her creatinine is back to normal levels. The patient is considered in remission and is on a steroid tapering regimen.
Discussion
The majority (90%) of anti-GBM disease cases present with Rapidly Progressive Glomerulonephritis (RPGN) manifesting as acute kidney injury with proteinuria and hematuria on urine analysis. Around half of these cases have concomitant pulmonary findings of alveolar hemorrhage presenting with cough, hemoptysis, and shortness of breath. 2 However, there exist multiple variants of the disease including the variant with isolated pulmonary manifestations and the double-positive anti-GBM and ANCA-associated vasculitis variant. The latter double-positive disease variant is suspected in patients who present with additional systemic features including weight loss, malaise, fever, or arthralgia which raises the suspicion of concurrent vasculitis.2,5 In our case, the patient complained of a recent history of weight loss, fatigue, and generalized malaise associated with a chronic fever, raising the suspicion of an underlying autoimmune process. This was consistent with the further discovery of both anti-GBM antibodies and MPO-ANCA in the serum.
A large study on 205 Chinese patients with Goodpasture syndrome found that around one-third of the patients positive for anti-GBM were also positive for MPO-ANCA. These patients, who were mainly aged between 60 and 70 years, had similar histopathologic and immunofluorescence findings as the Goodpasture syndrome patients who were only anti-GBM antibody positive. 6 Another large study reviewing around 500 cases of double-positive Goodpasture syndrome revealed that only 42% of those cases had alveolar hemorrhage in association with the RPGN and that 64% of double-positive cases required hemodialysis on presentation (with 61% of those remaining dialysis dependent at 1-year). 5 Interestingly, our patient did not have alveolar hemorrhage and did not require hemodialysis as part of her treatment.
It is worth noting that the renal biopsy results were consistent with a diagnosis of RPGN, but the immunofluorescence studies did not show the typical linear deposits seen in anti-GBM RPGN. This is most likely because the patient received high-dose IV steroids daily for 4 days prior to the biopsy coupled with the fact that double-positive patients had evidence of more chronic renal damage. 6 Additionally, the absence of immunofluorescence could hint to a diagnosis of pauci-immune RPGN more specifically ANCA-associated vasculitis with coexisting anti-GBM antibodies based on the serology results. In that scenario, the positive anti-GBM could be attributed to possible cross-reactivity with the ANCA. However, this possibility is rather unlikely due to high clinical suspicion and high serologic titers of anti-GBM compared to ANCA.
Although previous studies showed variable outcomes between single-positive and double-positive anti-GBM disease, a recent large retrospective comparative study concluded that double-positive cases have a higher 5-year relapse rate, a higher tendency to renal recovery, and intermediate long-term renal survival despite having more chronic renal damage on presentation compared to single-positive cases. 7 Those patients had better responses to immunosuppressive therapy but had the same overall survival rate. 7 Another retrospective study on 123 Goodpasture patients concluded that double positivity had similar kidney survival as single positive cases and was associated with less-intensive treatment requirements. 8 Our patient, who was double-positive for anti-GBM antibodies and MPO-ANCA had a very good response to plasmapheresis, prednisone, and cyclophosphamide. She had progressive improvement in renal function and did not require dialysis. Furthermore, her renal function normalized and remained stable 6 months postdischarge. However, the risk of recurrence in double-positive disease is close to 30% at 1 year and 60% at 5 years, which warrant close follow-up and possibly long-term immunosuppressive maintenance therapy for associated vasculitis.5,7 For this reason, our patient was discharged on daily maintenance of oral prednisone with regular measurement of her anti-GBM antibody and MPO-ANCA levels to assess for the need of retreatment with cyclophosphamide in case of relapse.
Treatment of anti-GBM disease mainly focuses on decreasing the load of anti-GBM antibodies; this can be achieved by either blocking the production of antibodies or by eliminating preformed circulating antibodies. This can be achieved by immunosuppressive therapy and by plasmapheresis, respectively.2,9 Plasmapheresis also attenuates inflammation by removing cytokines and other inflammatory mediators. Ideally, plasmapheresis should be initiated as early as possible even before confirmation by biopsy because prognosis is correlated with initial rapid reduction of antibodies. 9 Plasmapheresis should be ideally performed daily for at least 2 to 3 weeks. 9 Our patient received a total of 20 plasmapheresis sessions during a 4-week period. As for the immunosuppressive regimens, studies recommend using oral prednisone 1 mg/kg and oral cyclophosphamide 2 to 3 mg/kg daily with appropriate adjustments based on renal function.2,9 For our patient, we used 1 mg/kg of daily oral prednisone and 3 doses of IV cyclophosphamide because the oral form was not available. Due to insufficient evidence around the use of IV cyclophosphamide in anti-GBM disease, we administered 3 doses of 8 mg/kg (400 mg) IV cyclophosphamide separated by 8 hours on a single day for a total dose of 1.2 g. This dosing is close to that used in other ANCA-associated vasculitis after taking into consideration the patient’s age and renal function. 10
With this combination of daily oral prednisone, daily plasmapheresis, and 3 doses of IV cyclophosphamide, the patient had an initial phase of worsening renal function followed by a progressive improvement and decline in creatinine levels reaching 2 mg/dL 26 days after the initial plasmapheresis session (Figure 1). Also, her anti-GBM antibody and MPO-ANCA serum levels progressively decreased following treatment initiation. Furthermore, recent clinical trials showed that rituximab is better than cyclophosphamide in induction of remission from ANCA-associated vasculitis which also opens the possibility of using rituximab instead of cyclophosphamide in double-positive anti-GBM patients. 11
Conclusion
In this case report, we present a unique variant of RPGN characterized by positivity for both anti-GBM antibodies and MPO-ANCA. The patient was successfully treated and achieved complete renal recovery at 6-month follow-up. This case contributes to the understanding of these uncommon presentations of RPGN in terms of incidence, management, and overall prognosis; especially that the patient did not require dialysis or renal transplant for overall survival. This case also highlights the importance of timely management with a combination of immunosuppression and plasmapheresis for better outcomes. Finally, further research is needed to determine the optimal management approach and long-term outcomes for patients with this variant of RPGN.
Footnotes
Ethics Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for Publication
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
