Abstract
Acute rheumatic fever (ARF) is a complication of streptococcal pharyngitis that can present with cardiac, joint, skin, and neurological symptoms. Cardiac manifestations most often involve valvular dysfunction, but can also include myocarditis or pericarditis. Although advances in healthcare have reduced the prevalence of streptococcal pharyngitis, and subsequently ARF, individual cases and outbreaks can still occur. We present a case of rheumatic myopericarditis in a 60-year-old White male who initially presented to the emergency department with sore throat for 6 days. Initial workup was largely unremarkable, and no microbiological testing was performed at that time. He was diagnosed with presumed viral pharyngitis and discharged home with supportive care. He returned 1 week later with pleuritic mid-sternal chest pain and dyspnea. Laboratory tests were significant for elevated inflammatory markers, cardiac enzyme markers, anti-streptolysin O titers, and Streptococcus pyogenes bacteremia. Further evaluation revealed pericarditis, moderate pericardial effusion without tamponade, and reduced systolic function without valvular disease. The patient was diagnosed with rheumatic myopericarditis. Management included pericardial drainage, guideline-directed medical therapy for systolic heart failure and pericarditis, and primary treatment and secondary prevention of ARF with antibiotics. Currently, the patient’s cardiac function has recovered, and he regularly follows up with his medical care team. Although less common in present times, clinicians are encouraged to consider streptococcal pharyngitis and ARF on the differential diagnosis for patients presenting with pharyngeal symptoms and subsequent cardiac manifestations, with or without valvular dysfunction. Primary and secondary prevention of ARF is paramount to maintaining the low incidence of this disease.
Background
Pharyngitis is a common diagnosis in the ambulatory care setting, most often due to viral etiologies. However, bacterial causes of pharyngitis can also occur. Clinical scores such as the Centor criteria have been used to risk-stratify patients for streptococcal pharyngitis. 1 Advances in healthcare and infrastructure have further aided in proper identification and treatment of streptococcal pharyngitis, reducing the incidence of acute rheumatic fever (ARF) in developed nations. 2 However, individual cases and outbreaks can still occur. 3 The revised Jones Criteria is used to guide the diagnosis of ARF, and involves major and minor criteria that include cardiac, joint, skin, neurological, and inflammatory manifestations.3,4 Although the carditis associated with ARF most often manifests as clinical/subclinical valvular regurgitation, other cardiac findings can occur due to involvement of the myocardium and/or pericardium.3,5 We report a case of rheumatic myopericarditis causing significant pericardial effusion, further complicated by atrial fibrillation and systolic heart failure.
Case Report
A 60-year-old, White male initially presented to the emergency department with symptoms of sore throat for 6 days. His past medical history was significant for hypertension, hyperlipidemia, chronic obstructive pulmonary disease, tobacco use disorder, and a stable 4.1-cm thoracic aortic aneurysm. Home medications included amlodipine, lisinopril, metoprolol tartrate, atorvastatin, fluticasone/salmeterol, and albuterol. He denied alcohol or other substance use. On exam, he was afebrile, with stable vital signs, and was found to have non-exudative pharyngeal erythema and edema. No further microbiological testing was completed at that time. He was diagnosed with presumed viral pharyngitis and discharged with a short course of prednisone and Tylenol.
The patient returned 1 week later with sharp, pleuritic, substernal chest pain, as well as dyspnea. He reported resolution of his sore throat symptoms. Since the onset of the chest pain, he reported taking 2,000 mg of Tylenol every 12 hours. Review of systems was positive for chronic dry cough and diaphoresis, but negative for fever, palpitations, skin rashes, edema, syncope, orthopnea, paroxysmal nocturnal dyspnea, joint pain, and myalgias. He was found to be afebrile, normotensive, and tachypneic, with an oxygen saturation of 99% on room air. On exam, he was noted to be in moderate distress and diaphoretic, but was otherwise unremarkable. Cardiac exam was negative for murmurs, friction rubs, or gallops.
Laboratory tests were significant for leukocytosis to 32.1, lactate level of 2.56, and procalcitonin level of 13.5. Troponin-I and B-type natriuretic peptide (BNP) were elevated at 0.7 and 337, respectively. Erythrocyte sedimentation rate (ESR) was elevated to 23, C-reactive protein (CRP) was elevated to >200. Respiratory viral panel was negative. Rapid streptococcal A screen and culture were negative. Initial electrocardiogram (EKG) was notable for diffuse ST elevations and T-wave inversions in the inferolateral leads (Figure 1A). Bedside ultrasound revealed a small pericardial effusion without cardiac tamponade. Chest X-ray (CXR) and Computed Tomography (CT) chest revealed cardiomegaly, with moderate pericardial fluid and pericardial enhancement (Figure 2A and C).
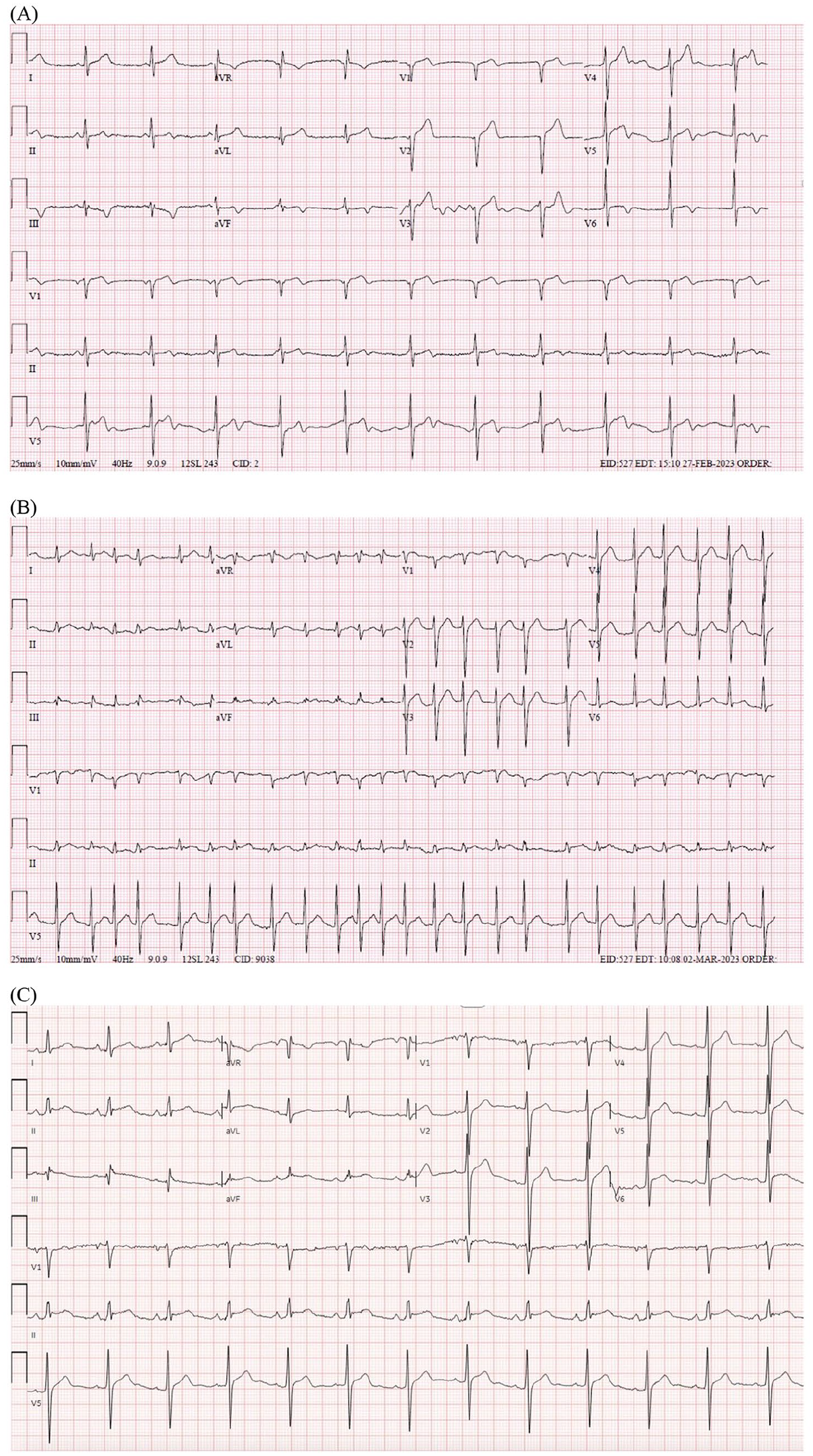
Evolution of EKG. (A) Initial EKG demonstrating accelerated junctional rhythm, with diffuse ST elevations and T-wave inversions in the inferolateral leads. (B) EKG demonstrating atrial fibrillation with rapid ventricular response. (C) Normalization of EKG after pericardiocentesis, but with persistent ST elevations.

Chest X-ray (A) prior to and (B) after pericardiocentesis. CT chest (C) before pericardiocentesis, and (D) after pericardiocentesis. Pericardial effusion (*).
At this time, differential diagnosis included acute coronary syndrome, acute pericarditis, myocarditis/myopericarditis, and ARF. The patient lacked personal/family history of autoimmune disease, malignancy, and exposure to cardiotoxins and/or radiation that could cause non-infectious carditis. He also denied prior symptoms or episodes of ARF in his childhood and adult life.
Given the history and diagnostic findings, acute pericarditis was highest on the differential, and the patient was started on prednisone, colchicine, and ibuprofen. Later in the hospital course, blood cultures returned positive for Streptococcus pyogenes, and anti-streptolysin O (ASO) titers were elevated to 1,320 IU/ml, concerning for ARF. Ceftriaxone was started due to the patient’s underlying penicillin allergy.
On day 3 of hospitalization, the patient experienced worsening substernal chest pain. EKG at that time revealed new atrial fibrillation with rapid ventricular response (Figure 1B). Repeat CXR and CT chest revealed increased pericardial effusion. Given the concerns for impending tamponade, the patient was transferred to another facility with cardiothoracic surgery and interventional cardiology capabilities.
Upon evaluation by cardiology at the new facility, the patient was noted to have newly reduced ejection fraction (EF) and circumferential pericardial effusion without tamponade. The patient underwent pericardiocentesis of 750 cc of serous yellow fluid, with placement of a pericardial drain and subsequent resolution of atrial fibrillation (Figure 1C). Complete transthoracic echocardiogram (TTE) revealed a mildly reduced EF of 41%, trace mitral valve regurgitation, but no other significant abnormalities. Given the reduced systolic function, the patient was started on guideline-directed medical therapy (GDMT) with metoprolol, spironolactone, lisinopril, and Jardiance. Pericardial fluid was exudative, cultures were negative, and cytology was only remarkable for marked acute inflammation. The pericardial drain continued to drain serosanguinous fluid until it was removed 6 days after initial placement. TTE after pericardial drain removal demonstrated resolution of the pericardial effusion (Figure 3). The patient was discharged with plans to continue ceftriaxone for 4 weeks, colchicine for 3 months, and ibuprofen for 2 weeks for ARF and acute pericarditis, respectively.
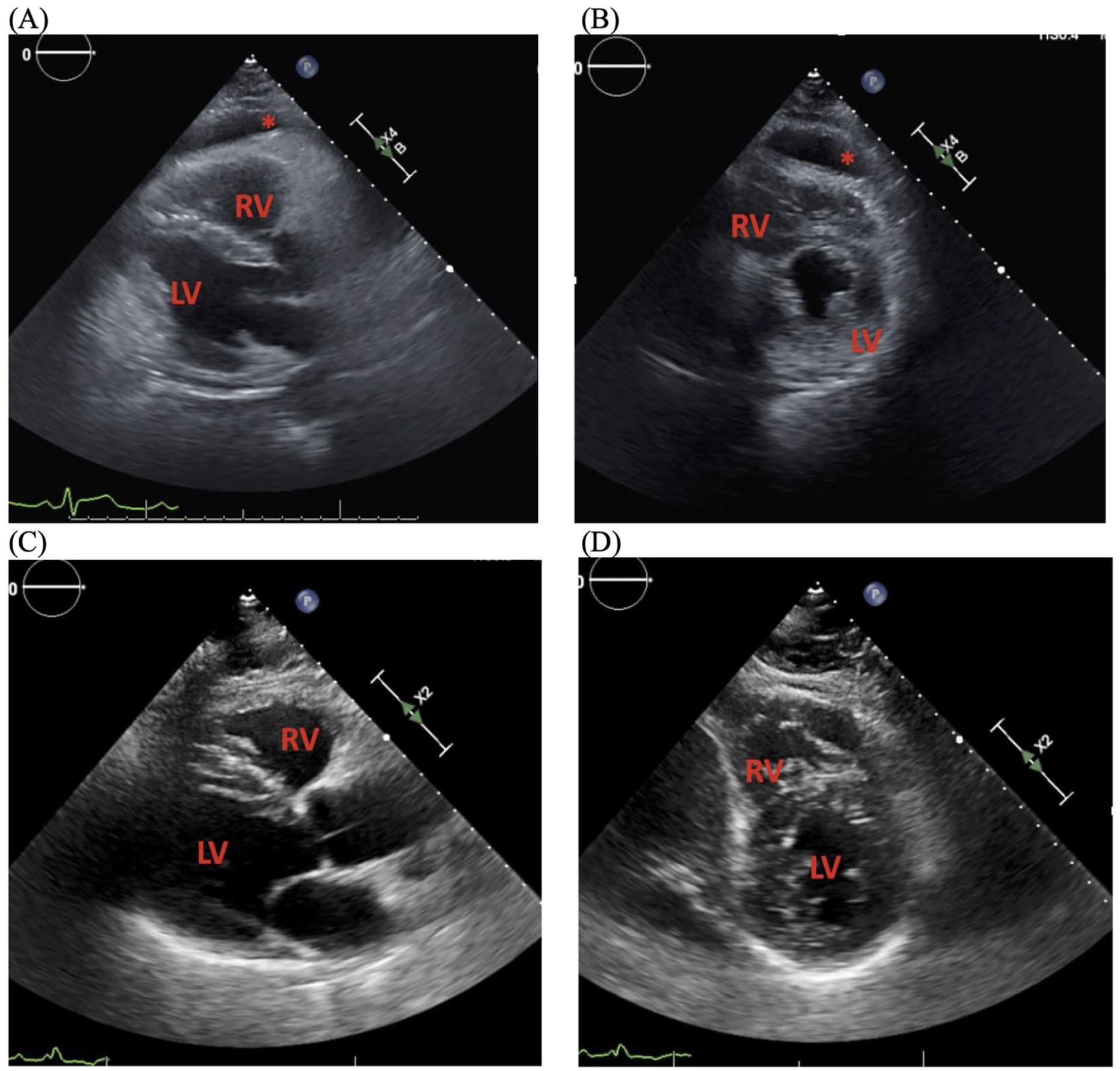
Echocardiographic resolution of pericardial effusion. Residual anterior pericardial effusion shortly after pericardiocentesis and pericardial drain placement (A) parasternal long axis view, (B) parasternal short axis view. Resolution of pericardial effusion after removal of pericardial drain (C) parasternal long axis view, (D) parasternal short axis view. RV, right ventricle. LV, Left ventricle. Pericardial effusion (*).
After discharge, the patient continued to follow up with cardiology and infectious disease. TTE 1-month post-discharge revealed an EF of 35% to 40%, without pericardial effusion. A 4-month post-discharge TTE revealed recovery of EF to 55% to 60%. The patient continues to do well without significant complications (Figure 4).

Timeline of events.
Discussion
Clinical scores such as the Centor criteria can be used to risk-stratify patients for streptococcal pharyngitis, and low scores have been shown to have high sensitivity. 1 In our case, our patient had a Centor score of 0, which has been estimated to have a sensitivity of ~96%. 1 Thus, no further testing for streptococcal pharyngitis was pursued at initial presentation. Unfortunately, his course was later complicated by clinically significant myopericarditis and pericardial effusion, requiring pericardial drainage and GDMT for reduced systolic function and pericarditis.
The mechanism underlying ARF is hypothesized to be immune-mediated activation of endogenous markers due to molecular mimicry against bacterial proteins. 3 Clinically, this autoimmune injury manifests into carditis, arthralgias, chorea, skin findings, fever, and elevated inflammatory markers as noted in the revised Jones criteria. 4 In our patient, he met 1 major criterion for carditis and 1 minor criterion for elevated CRP. However, he denied fever, skin, joint, or neurological symptoms. The only notable temperature changes were hypothermia to 35.7 °C and hyperthermia to 38.1 °C on days 3 and 5 of hospitalization, respectively. It should be noted that the patient self-administered antipyretics prior to presentation and continued receiving scheduled doses throughout his hospital stay, possibly masking a fever >38.5 °C that would be included as a second minor criterion for ARF.
Prior case reports have described a non-rheumatic myocarditis/myopericarditis that develops several days after initial streptococcal pharyngitis. 6 The mechanism underlying non-rheumatic myocarditis/myopericarditis has been postulated to be toxin-mediated, as opposed to immune-mediated, in which classic rheumatic carditis presents within 2 to 5 weeks from initial streptococcal pharyngitis.3,5 -7 In this case, clinical symptoms of myopericarditis developed 2 weeks after the onset of pharyngitis. By the time the patient returned to the emergency department for chest pain, his symptoms of pharyngitis had resolved. At that time, he was found to have a negative rapid streptococcal A screen and culture, arguing against an active streptococcal pharyngitis. He did however demonstrate supportive criteria for ARF, including S. pyogenes bacteremia and elevated ASO titers 2.5 weeks after initial pharyngeal symptoms.
Importantly, ARF is rare in older patients, especially those over age 50, and other differentials should be considered, as was done in this case. 2 Diagnostic testing for autoimmune disease, malignancy, and other viral etiologies of myopericarditis, such as coxsackievirus, can be obtained if clinical suspicion warrants such testing. In our case, because a respiratory viral panel was negative, there was no personal or family history of autoimmune disease, no exposure to cardiotoxins and/or radiation that could cause noninfectious carditis, and the pericardial fluid studies were negative for malignant cells, alternative etiologies were less likely. Taking all of this into account, the etiology of the myopericarditis was presumed secondary to ARF.
The cardiac manifestations of ARF have most notably been clinical or subclinical valvular regurgitation. Myocardium or pericardium predominant cardiac involvement has been noted, but is generally rare. 3 Our patient was noted to have trace mitral regurgitation, but significant myopericarditis leading to reduced systolic function and classic signs of pericarditis, including sterile pericardial effusion, pleuritic chest pain, and diffuse ST elevations.8,9 The management of myopericarditis includes treatment of pericarditis with nonsteroidal anti-inflammatory drugs, colchicine, and steroids, as well as supportive management of myocardial injury. 9 If associated pericardial effusions result in cardiac tamponade or lead to significant discomfort or arrhythmias, pericardial drainage is recommended, as was done in our case. 10
Once ARF has been diagnosed, many patients warrant secondary prophylaxis to prevent further streptococcal infections and episodes of ARF. Monthly penicillin injections are generally preferred, although daily oral penicillin can also be used.2,3,5 The duration of secondary prophylaxis depends on various factors, including the number of prior ARF episodes, the time elapsed since the last episode, and the extent of cardiac involvement. Secondary prophylaxis is recommended for 10 years or until the age of 40 for severe valvulitis. 2 In patients with penicillin allergies, alternatives include macrolides or sulfonamide. 2
Conclusion
Advances in healthcare and social infrastructure have largely eradicated ARF in developed countries such as the United States. However, it is prudent for clinicians to recognize preceding streptococcal pharyngitis and associated rheumatic disorders as possible etiologies of myopericarditis. Primary and secondary prevention remain key to maintaining low incidence and prevalence of rheumatic heart disease.
Footnotes
Acknowledgements
None.
Authors’ Note
Prior Presentation of Abstract Statement: This abstract was previously presented at the 2025 American College of Cardiology Conference in Chicago, Illinois, on March 29, 2025.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
