Abstract
When there is concern that a rash may be a drug eruption reaction, there are many well-known culprits such as antibiotics and phenytoin. When an uncommon or previously unknown offending medication is being considered to have caused a drug eruption, diagnostic skin biopsy can help confirm the diagnosis. Our patient is a 54-year-old female with past medical history of prediabetes and obesity. She had ST segment-elevation myocardial infarction that was treated appropriately with timely percutaneous coronary intervention, which included angioplasty and drug-eluting stent placement. She was maintained on treatment regimen, and over the next 3 months, she developed a worsening maculopapular rash across her torso and upper extremities. Her biopsy showed superficial and mid-dermal perivascular lymphohistiocytic inflammation with focal spongiosis and scattered eosinophils, a common pathologic finding seen in drug eruptions. Although the patient was not taking any well-known offending agents, her ticagrelor was discontinued and she was transitioned to clopidogrel. Shortly after the transition, the patient’s drug rash resolved without recurrence. Drug eruptions are a common etiology for rashes leading to presentation in primary care clinics. Our patient illustrates an exceedingly rare case of ticagrelor-induced drug reaction, as a review of the literature shows only 2 previously published case reports.
Background
Skin rashes can be difficult to diagnose but are a commonly-presenting symptom to primary care internal medicine practices. Despite a thorough history and physical examination, the underlying cause of the rash may remain unclear. For more challenging and complex cases, expert consultation and/or skin biopsy are the next steps in pursuing a diagnosis. When there is concern that a rash may be a drug eruption reaction, there are many well-known culprits such as antibiotics and phenytoin. When an uncommon or previously unknown offending medication is being considered to have caused a drug eruption, diagnostic skin biopsy can help confirm the diagnosis. We present a case of a biopsy-confirmed drug eruption rash from an uncommon offender: the P2Y12 inhibitor and antiplatelet agent ticagrelor.
Case Report
Our patient is a 54-year-old Caucasian female with past medical history of prediabetes and obesity. She had ST segment-elevation myocardial infarction that was treated appropriately with timely percutaneous coronary intervention, which included angioplasty and drug-eluting stent placement. The patient was placed on guideline-directed medical treatment for her infarction with metoprolol, ramipril, aspirin, and ticagrelor. She had an uneventful hospital course and was discharged home. She was maintained on this treatment regimen, and over the next 3 months, she developed a worsening maculopapular rash across her torso and upper extremities. Over the course of several urgent care office visits, she was treated for refractory contact dermatitis with diphenhydramine, triamcinolone cream, and oral prednisone. She was seen by multiple different providers, ultimately choosing to repeat previous treatments as they had resolved the rash for the patient for a short time after treatment. Little thought was given to drug-induced rash at the time because patient had a remote exposure to a hotel stay, so contact dermatitis was highest on the differential.
Despite 6 weeks of conservative management, her rash did not improve. She was seen by her primary care provider who referred to dermatology where they elected to complete a skin biopsy. Punch biopsy was elected in this case as on presentation seemed to be a refractory contact dermatitis since she originally tolerated her oral prescription medications. Since it was high risk to change/stop any of her dual-antiplatelet agents, confirmatory testing was elected for prior to discontinuation. A punch biopsy of the right forearm was performed showing minimal epidermal changes with a superficial and deep predominantly perivascular inflammatory infiltrate. The perivascular inflammatory infiltrate was predominantly lymphohistiocytic with associated eosinophils. The main histologic differential diagnosis for these histomorphologic features is a dermal hypersensitivity reaction to a medication or other internal antigen (Figures 1–3).

Hematoxylin and eosin (H&E) staining 40×: a punch biopsy of the right forearm. There are minimal epidermal changes with a superficial and deep predominantly perivascular inflammatory infiltrate.
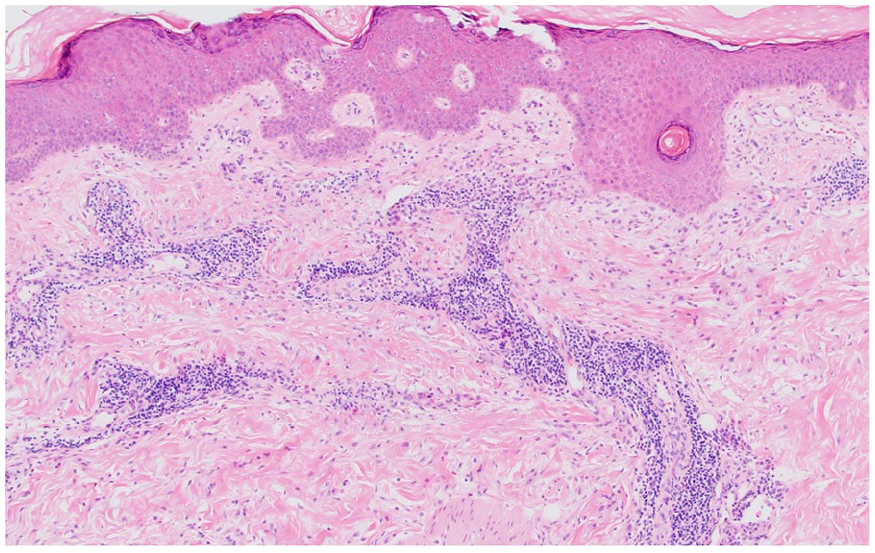
H&E 100×: scattered perivascular and interstitial eosinophils are noted.
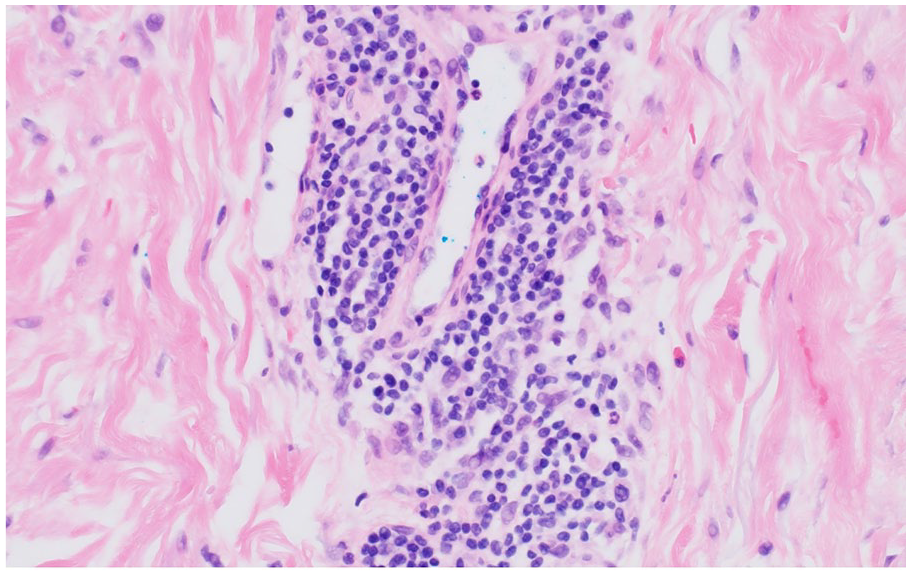
H&E 400×: the perivascular inflammatory infiltrate is lymphohistiocytic predominantly lymphohistiocytic with an associated eosinophil to the right of the vessel.
Although the patient was not taking any well-known offending agents, her ticagrelor was discontinued and she was transitioned to clopidogrel, an alternative P2Y12 inhibitor. In terms of antiplatelet agents, clopidogrel has been around the longest with the most safety data information available, so the providers elected for this transition; little information about the cross reactivity of prasugrel is available at this time. Shortly after the transition, the patient’s drug rash resolved without recurrence.
Discussion
Drug eruptions are a common etiology for rashes leading to presentation in primary care clinics. There are many common drugs or offending agents that have been previously identified. Furthermore, the expansion of new pharmaceuticals in recent years continues to produce more potential offending agents. Our patient illustrates an exceedingly rare case of ticagrelor-induced drug reaction, as a review of the literature shows only 2 previously published case reports. One patient had a whole-body hypersensitivity reaction to ticagrelor that resolved with drug discontinuation, 1 while another patient developed a fixed cutaneous hypersensitivity reaction with bullae. 2 Although our patient had some similarities in skin findings to each of these cases, our case is unique in that the rash was a fixed cutaneous reaction. This highlights the importance for clinicians to have a heightened level of suspicion for a ticagrelor-induced drug reaction given its variability in clinical presentation.
When a drug reaction to ticagrelor is suspected, there are very limited data to suggest the best alternative P2Y12 inhibitor. Clopidogrel or prasugrel are potential options, as are other P2Y12 inhibitors depending on the indication for use and clinical circumstances. Skin reactions with clopidogrel have been previously documented (upward of 6%), but the true cross-reactivity rate with other P2Y12 inhibitors is not unknown due to the rarity of these drug reactions. 1
Conclusion
In patients who present with a skin rash, drug eruptions should be considered as part of the differential diagnosis, even when the medication in question is not commonly known to cause such reactions. Our patient demonstrated a rare drug eruption hypersensitivity reaction to ticagrelor, an increasingly more common antiplatelet agent and P2Y12 inhibitor, that required a skin biopsy for diagnosis. This is one of only a few reported cases of a ticagrelor-induced drug reaction and highlights the variability in rash presentation. As ticagrelor is prescribed more frequently, clinicians should maintain a level of suspicion for ticagrelor-induced drug eruption as an etiology for skin rashes in their patients.
Footnotes
Acknowledgements
We acknowledge the University of North Dakota School of Medicine and Health Sciences for their assistance in publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethics approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
This was not presented regionally or nationally.
