Abstract
Mycobacterium kansasii is a slow-growing mycobacterium commonly affecting the lungs, usually in a structurally deformed lung. We discuss a case of 67-year-old male with a medical history of complicated coal workers pneumoconiosis or coal mine dust lung disease presented to the pulmonary clinic with worsening dyspnea and hemoptysis with imaging findings revealing cavitary lung lesions in the right upper lobe and progressive massive fibrosis (PMF) bilaterally. Patient sputum gram stain showed acid-fast bacilli, and the cultures grew M. kansasii. He was started on rifampin, ethambutol, and azithromycin. This case highlights the importance of thorough workup of cavitary lung lesion, reinforcing the association of mycobacterium species in a patient with PMF and associated cavities.
Keywords
Introduction
Coalmine dust lung disease encompasses various respiratory conditions that arise in miners due to inhalation of respirable dust. The most severe type within this spectrum is progressive massive fibrosis (PMF), marked by significant scarring of lung tissue, which hampers normal lung function and affects gas exchange. An increase in cases has been observed in the Appalachian region, particularly in states such as Kentucky, West Virginia, Pennsylvania, Tennessee, and Virginia, where mining is the main industry. The scarring and inflammation from mineral dust exposure may facilitate the colonization with mycobacterium. Mycobacterium kansasii, identified by Buhler and Pollack as a slow-growing mycobacterium, is one of the common causes of pulmonary infections among nontuberculous mycobacteria (NTM). Clinically and radiologically, M. kansasii resembles Mycobacterium tuberculosis, presenting symptoms such as chest pain, cough, hemoptysis, fever, night sweats, and computed tomography (CT) scans showing cavitary or nodular/bronchiectatic patterns. Some of the clinical and radiological similarities between coal workers pneumoconiosis with PMF, and M. kansasii, make it a diagnostic challenge.
Case Presentation
A 67-year-old man with a history of PMF secondary to coal dust exposure and chronic obstructive pulmonary disease (COPD) presented to the pulmonology clinic with progressive worsening of chronic dyspnea over the past 20 years. His dyspnea was classified as grade 2 on the Modified Medical Research Council scale and was associated with chronic cough and intermittent episodes of blood-tinged sputum for the past 3 years. He was compliant with tiotropium–olodaterol and albuterol as needed and used 2 L/min of supplemental home oxygen. The patient reports being a nonsmoker and has never used any tobacco-related products.
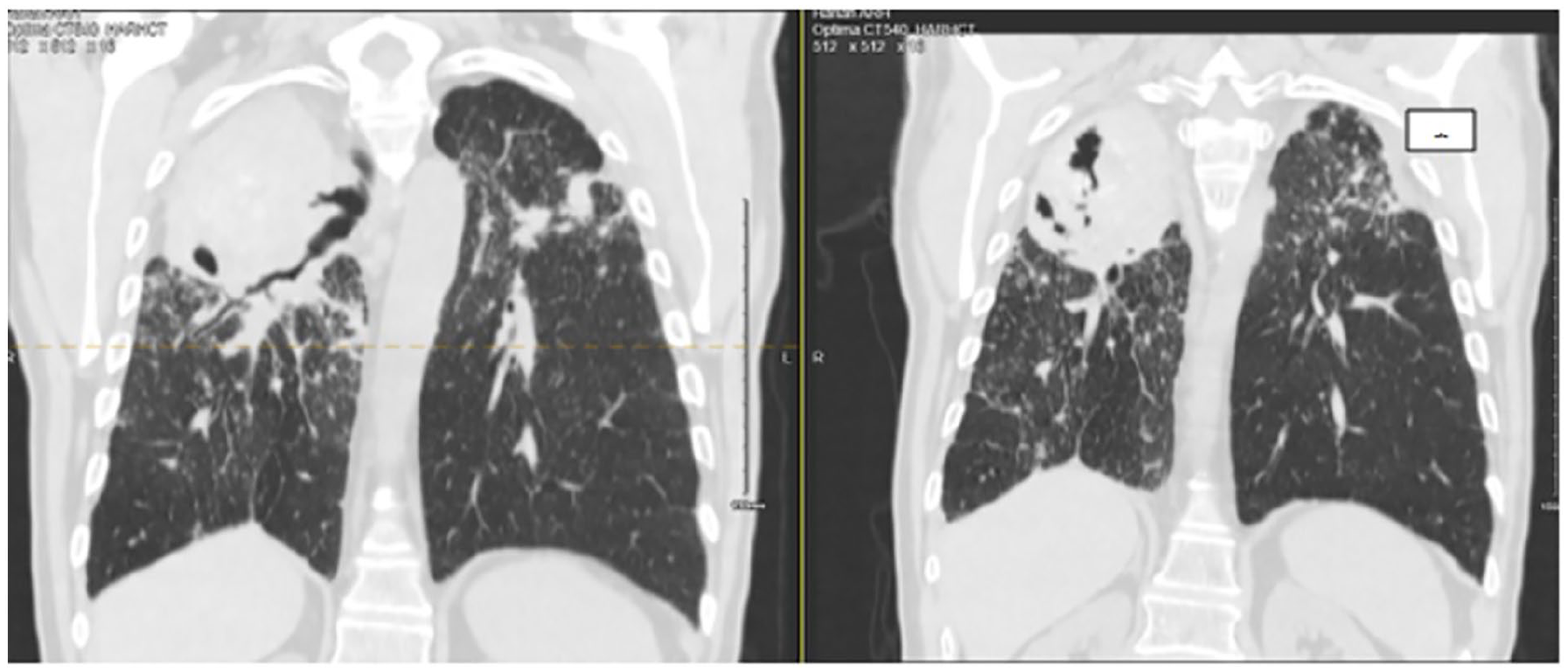
Five days prior to presentation, he reported increased production of yellowish-green, blood-tinged sputum. He had experienced similar episodes of hemoptysis in the past but had not sought medical evaluation or treatment. A CT scan of the chest (Figure 1) before starting the treatment revealed an enlarging cavitary lesion in the right upper lobe in the mass-like area (11.3 cm × 5.9 cm), with small similar structures in the left upper lobe, numerous masses throughout the lungs with diffuse nodular interstitial thickening, calcified mediastinal and hilar lymphadenopathy, and masses typical of granulomas on both lungs compared to a CT scan of chest 6 months ago, representing progression compared to imaging 6 months prior.

Increasing size of the right upper lobe cavitation before treatment (6 months apart).
As a part of the diagnostic workup for cavitary lung disease, the patient underwent extensive testing for infectious, autoimmune, and immunodeficiency-related etiologies. Laboratory evaluation was negative for HIV, Quantiferon-TB Gold, anticyclic citrullinated peptide antibodies, antinuclear antibodies, antineutrophil cytoplasmic antibodies, rheumatoid factor, and serum galactomannan. These findings reduced the likelihood of immunodeficiency, latent or active tuberculosis, systemic autoimmune diseases, vasculitis, and invasive fungal infections. Additional diagnostic studies included sputum culture, Gram stain, acid-fast bacilli (AFB) smear, an upper respiratory viral panel, and pulmonary function testing (PFT). Bronchoscopy was offered but declined by the patient. PFT revealed an FEV1/FVC ratio of 47%, FEV1 of 1.2 L (37% predicted), and FVC of 2.6 L (62% predicted), consistent with moderate-to-severe obstructive lung disease. Sputum AFB smear was positive, and culture subsequently confirmed M. kansasii 6 weeks later. Repeat AFB smear and culture confirmed the diagnosis. Drug susceptibility testing showed resistance to ciprofloxacin and doxycycline, intermediate susceptibility to minocycline, and susceptibility to rifampin. A baseline dilated ophthalmologic examination and laboratory evaluation, including complete blood count and comprehensive metabolic panel, were within normal limits.
Following a multidisciplinary discussion between pulmonology and infectious disease teams, the patient was initiated on a regimen of rifampin 600 mg daily, ethambutol 1200 mg daily, and azithromycin 500 mg daily. He has been on therapy for 10 months, with monthly AFB smears and cultures remaining negative for the past 9 months. A repeat dilated eye examination at 6 months remained normal. The patient reported significant improvement in hemoptysis and dyspnea. Follow-up CT imaging demonstrated a reduction in the size of pulmonary masses (Figure 2). This case highlights the increased susceptibility of patients with underlying COPD and PMF to pulmonary infections with M. kansasii, and the importance of comprehensive diagnostic evaluation and multidisciplinary management in such presentations.
Recent CT scan of chest S/p treatment. Redemonstrated pulmonary masses 10.2 cm × 5.8 cm the right lung apex with cavitation, previously 11.3 cm × 5.9 cm. 7 cm × 6.2 cm mass/conglomerate of masses in the left upper lobe, previously 7.6 cm × 7.2 cm.
Discussion
Coal workers pneumoconiosis is a spectrum of respiratory diseases developing in miners exposed to respirable dust. The most advanced form being PMF, among the spectrum, characterized by extensive fibrosis of the lung parenchyma disrupting normal physiological function and impairing gas exchange. As per study by Almberg et al, from 1970 to 2016, 4679 PMF cases were reported among 314 176 miners who applied for Federal Black Lung Program benefits. Coalmine employment decreased from 250 226 miners in 1979 to 81 485 miners in 2016. However, PMF cases from 1996 to 2016 (2474) exceeded those from 1970 to 1996 (2205). Most PMF cases (83.9%) were from West Virginia (28.4%), Kentucky (20.2%), Pennsylvania (20.0%), Virginia (15.3%), and Tennessee. The highest increase in PMF cases was in Virginia (31.5%), followed by Kentucky (16.6%) and West Virginia (16.8%). The average age of miners with PMF at the time of their claim was 61.6 years, with an average coalmine employment duration of 23.0 years for PMF cases compared to 18.9 years for non-PMF claimants. Among the miners, 1621 (84%) had large opacities indicative of PMF: 915 miners (56%) were classified as category A (opacities >1 and <5 cm); 548 miners (34%) fell into category B (opacities >5 cm); and 158 miners (10%) were in category C (opacities in the right upper lung zone). 1
There are several case reports that suggest presentation of different variants or subspecies of nontuberculous mycobacterial organisms or fungal infections (chronic aspergillus infections, chronic coccidiomycosis, para-coccidiomycosis, histoplasmosis, and melioidosis) that can present as cavitary lung lesions.2,3 The important risk factors for cavitary lung lesions are underlying COPD due to coal workers pneumoconiosis, age of more than 60, and previous history of anti-TB treatment. 2 Prominent bronchiectasis and nodules, along with limited thin-walled cavitary disease, may indicate an NTM infection. 3 Mycobacterium avium complex, Mycobacterium xenopi, M. kansasii, Mycobacterium nebraskense, Mycobacterium szulgai, Mycobacterium kyorinense, Mycobacterium heckeshornense, Mycobacterium brisbanense species nova, Mycobacterium heckeshornense, and Mycobacterium vaccae were some of the nontuberculous mycobacterial causes of cavitary lung lesions.4-11 In immunocompromised individuals (such as those with old age, steroid use, myelodysplasia, or HIV), M. kansasii can cause disseminated systemic infections, leading to necrotizing and nonnecrotizing granulomas in the lungs, heart, liver, spleen, aortic wall, and kidneys. 12 A study by Aviram et al found that among 25 HIV-positive patients with cavitary lung disease, polymicrobial infections were common, with bacterial organisms found in 85% of patients, often alongside other pathogens such as M. tuberculosis, Pneumocystis jirovecii, cytomegalovirus, and various fungal species. Patients with bacterial pathogens alone had a significantly higher mean CD4 count (203) compared to those with nonbacterial pathogens (42). 13
M. kansasii is a slow-growing, nontuberculous mycobacterium linked to rare yet severe infections, typically found in urban and industrial settings. It is an uncommon but significant pathogen causing pulmonary and extra-pulmonary infections, particularly in immunocompromised individuals. 14 Among the case–control studies done in Korea and Africa 20% to 30% of the miners had mycobacterial infections out of which 60% were NTM compared to TB.15,16 Tap water being the major reservoir, infection is acquired through the aerosol route. The pathogenesis of M. kansasii involves the inhalation of aerosolized bacteria, leading to respiratory infections that can mimic tuberculosis. 17 Once inhaled, the bacteria evade the host immune system through mechanisms such as the production of mycolic acids in their cell walls, which inhibit phagosome-lysosome fusion in macrophages. 18 M. kansasii’s pathogenicity is largely due to its ability to survive and replicate within host macrophages. The bacterium’s cell wall is rich in lipids, which contribute to its resistance to host defenses and antimicrobial treatments. 18 Corbett et al, 19 Sonnenberg et al, 20 and Lee and Myong, 21 found that dust exposure patients had small opacity profusion and cavity progression pointing toward pneumoconiosis promoting NTM lung disease.19-21
The approximate incubation period for M. kansasii infections varies but generally ranges from several weeks to months following exposure. The clinical presentation often begins with pulmonary symptoms, which can include chronic cough, weight loss, night sweats, and hemoptysis. In about 20% to 30% of cases, the infection can disseminate to other parts of the body, such as the skin, bones, or lymph nodes. 22
The clinical implications are significant, as it underscores the necessity for early and accurate diagnosis of M. kansasii infections. Misdiagnosis or delayed diagnosis can lead to severe complications and prolonged morbidity. Like in our case, the patient has structural lung changes secondary to PMF from a coal worker’s pneumoconiosis which can cause opacities and cavitation. PMF has a high affinity for upper lobes and can cause central ischemic necrosis and cavitation which is very similar to mycobacterial infection making diagnosis difficult. Diagnostic tools such as chest radiograph to assess for cavitation or nodular opacities, CT chest to assess any bronchiectasis changes, sputum cultures, bronchoalveolar lavage, and molecular techniques (e.g., PCR) are crucial for confirming the presence of M. kansasii. 23 This case underscores the need for customized antimicrobial therapy for M. kansasii, as per the latest American Thoracic Society (ATS) guidelines. For pulmonary disease caused by M. kansasii, treatment based on susceptibility to rifampicin is recommended over empiric therapy. A regimen including rifampicin, ethambutol, and either isoniazid or a macrolide is advised for rifampicin-susceptible M. kansasii pulmonary disease. The ATS recommends treating for at least 12 months.23,24
Despite appropriate antimicrobial therapy, cavitary pulmonary lesions may persist or worsen, contributing to increased all-cause mortality. These lesions can serve as reservoirs for secondary infections, particularly in patients with underlying structural lung disease. Superinfections with opportunistic pathogens such as Nocardia spp., Aspergillus fumigatus, Coccidioides spp., Cryptococcus neoformans, and Histoplasma capsulatum have been documented and may complicate the clinical course even after successful treatment of the primary infection and negative AFB results. In select cases, localized surgical resection of cavitary lesions may be considered to reduce microbial burden and prevent recurrent or refractory infections, especially when medical therapy alone is insufficient. Given the complexity and variability in outcomes, there is a critical need for comprehensive, adequately powered studies to better define the role of adjunctive surgical intervention and to establish evidence-based guidelines for the prevention and management of superinfections in patients with cavitary lung disease. 25
Conclusion
NTB mycobacterial infections should be kept in the differential diagnosis, in patients who presents with chronic cough and hemoptysis along with cavitary lesions especially in the setting of coal workers pneumoconiosis with PMF. Early detection of NTB mycobacterial infection by sputum AFB and culture is crucial in reducing disease burden on the patient, preventing lesion progression and avoiding complications. Continued surveillance and reporting of M. kansasii cases will aid in understanding its epidemiology and improving patient outcomes.
Footnotes
Authors’ Note
The abstract of this manuscript was submitted to ATS 2025 and was accepted. I will be presenting the poster at ATS 2025.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
