Abstract
Brucellosis is considered one of the most common bacterial zoonosis worldwide. It can affect any organ system and presents with many clinical manifestations related to the organ affected. It is quite uncommon for brucellosis to affect the breast. Here, we present a case of a 41-year-old female who presented with breast abscess associated with generalized symptoms such as fever, fatigue, epigastric pain, and arthralgia. Subsequent pus culture followed by blood culture confirms the diagnosis of brucellosis. This case emphasizes how crucial it is to take brucellosis into account when making a differential diagnosis for breast abscesses, especially in regions where the disease is endemic or in patients who have risk factors for zoonotic diseases.
Introduction
Breast abscesses in both breastfeeding and nonlactating women are often treated empirically as bacterial infections. Nonetheless, the association with universally unusual symptoms need to initiate a diagnostic process.
One of the most prevalent bacterial zoonosis in the world is brucellosis. 1 Humans can contract brucellosis, a zoonotic infection, from infected animals. Common names for it include “Mediterranean fever,” “undulant fever,” and “Malta fever.” The 4 species of Brucella that can infect people are Brucella melitensis, Brucella abortus, Brucella suis, and Brucella canis. Most human cases worldwide are caused by B. melitensis. 2 In many areas with few resources, it is also a serious public health concern. 2
Although a breast abscess and brucellosis are not often associated, breast abscess should be considered a differential diagnosis for brucellosis.
This case report describes a breast abscess caused by brucellosis that was treated with an appropriate antibiotic regimen and early identification. By highlighting this case, we intend to minimize brucellosis sequelae through early identification and treatment.
Case Presentation
A 41-year-old female with a history of hypertension, insulin intolerance, migraine, pituitary prolactinoma, central hypothyroidism, general fatigue, feverish sensation, epigastric pain, and generalized arthralgia of 1.5-month duration with recent 15 kg weight loss. She had a notable history of exposure to unpasteurized dairy products in a rural village with limited access to commercial stores. Presented to the emergency department with right peri-areolar breast pain, swelling, redness, and hotness. On physical examination, she exhibited fever 39 °C, tachycardia with a heart rate of 105 beats/minute, right peri-areolar breast swelling 5 × 5 cm, with erythema, sloughed skin, tenderness fluctuation, no breast masses, no palpable axillary lymphadenopathy, and no nipple discharge.
Normal leukocyte counts and elevated C-reactive protein and alanine aminotransferase were detected through the initial laboratory testing.
Ultrasound of the breast revealed a 5 × 6 cm collection.
An incision and drainage of a breast abscess were performed, thick greenish-yellow purulent discharge came out, and cultures were saved under sterile technique using cup culture. Purulent discharge and blood culture were sent; 2 days’ later, purulent discharge culture grew gram-negative coccobacilli and were finally identified as B. melitensis. Therefore, blood Brucella serology was done with a titer >1:320 (WHO normal reference range is <1:160), and B. melitensis culture was sent for sensitivity. The blood culture result was negative of any growth.
Due to back pain and long-lasting symptoms, further Brucella work-up with normal results on the spine MRI and cardiac echocardiography.
She received daily dressings until her erythematous, indurated open wound healed properly, along with 6 weeks of antibiotics (rifampicin and doxycycline). During the early stages of treatment, the patient’s Brucella serological titer first rose from 1:320 to 1:640, most likely as a result of continued antigenic stimulation. However, by 6 weeks, the titer showed a significant decline to 1:80, indicative of effective treatment.
Discussion
This case highlights the diagnostic challenges of identifying atypical agents, such as Brucella, in breast abscesses. In regions with a high risk of zoonotic infections, a thorough clinical assessment and early microbiological workup are crucial to avoid misdiagnosis and delayed treatment. Despite the initial diagnostic complexity, timely identification, and appropriate management with a 6-week course of antibiotics (rifampicin and doxycycline) resulted in complete recovery without complications or relapse during follow-up.
Brucellosis has been recognized as one of the most common bacterial zoonosis worldwide, despite the lack of accurate human incidence statistics, which is mostly caused by underreporting and inaccurate diagnosis. 1 Most cases of brucellosis are characterized by systemic manifestations such as fever, arthralgia, and hepatosplenomegaly. 2 Although brucellosis can impact any organ system, 3 it is quite uncommon for the breast to be affected. To our knowledge, there have only been 10 cases documented in the literature since 1970 that link brucellosis to breast abscesses 4 ; and no prior case record mentioned a breast abscess as the initial presentation. 5 These manifestations are frequently misdiagnosed as bacterial infections, which postpones necessary medical intervention.
Brucellosis can affect any organ system and presents with many clinical manifestations related to the organ affected. Focal brucellosis most commonly manifests as osteoarticular disease.6-8 It comprises spondylitis, sacroiliitis, and peripheral arthritis. Genitourinary involvement, 9 orchitis and/or epididymitis, tubo-ovarian abscess, cystitis, interstitial nephritis, glomerulonephritis, and renal abscess. 10 Neurologic involvement 9 includes meningitis, encephalitis, brain abscess, myelitis, radiculitis, and/or neuritis. 10 Cardiovascular involvement 9 includes endocarditis, myocarditis, pericarditis, endarteritis, thrombophlebitis, 11 and/or mycotic aneurysm of the aorta. The most frequent cardiovascular consequence and the primary reason for brucellosis-related deaths is endocarditis. 12 Lung nodules, pleural effusion, hilar lymphadenopathy, lobar pneumonia, interstitial pneumonia, bronchitis, and empyema are examples of pulmonary involvement.12-14 Intra-abdominal manifestations are rare; these may include hepatic or splenic abscess, cholecystitis, pancreatitis, ileitis, colitis, and peritonitis. Keratoconjunctivitis, corneal ulcers, iridocyclitis, nummular keratitis, choroiditis, optic neuritis, papilledema, and endophthalmitis are examples of ocular involvement, which is uncommon. 15
Dermatologic manifestations include macular, maculopapular, scarlatiniform, papulonodular, and erythema nodosum-like eruptions, ulcerations, petechiae, purpura, granulomatous vasculitis, and abscesses.12,16
The most frequent ways that brucellosis is spread to people are through the ingestion of contaminated, unpasteurized animal products, skin or mucous membrane contact with contaminated animal fluids (blood, urine, or milk) or tissue (placenta or miscarriage products), inhaling aerosolized particles that are contaminated. 2
Human illness can be caused by 4 different species of Brucella: B. melitensis, which was isolated from small ruminants including sheep, goats, and camels; B. abortus, which was isolated from cattle; B. suis, which was isolated from swine; and B. canis, which was recovered from dogs. B. melitensis is responsible for the majority of human cases worldwide. 2 Human infections caused by B. suis and B. melitensis are typically more virulent than those caused by B. abortus or B. canis.17,18
Although it is uncommon, cutaneous brucellosis has been reported in a few of the cases. The most frequent skin symptom has been widespread papulonodular eruptions, which are followed by lesions resembling erythema nodosum. It was believed that hematogenous spread was the cause of these lesions. B. suis is the primary source of soft tissue abscess formation, which has been documented in all of these cases involving skin abrasions or cuts; in earlier cases, there was neither a history nor any clinical signs of skin abrasions or cuts. This implies that the most likely pathogenic mechanism was hematogenous dissemination. 18
Controlling the disease and avoiding complications, relapses, sequelae, and death are the goals of brucellosis treatment.19,20
In the absence of localized disease from spondylitis, neurobrucellosis, or endocarditis, WHO guidelines for treating brucellosis include doxycycline 100 mg twice a day for 45 days, plus streptomycin 1 g daily for 15 days. The main alternative therapy is doxycycline at 100 mg twice a day for 45 days, plus rifampicin at 15 mg/kg/day (600-900 mg) for 45 days. Experience suggests that streptomycin may be substituted with gentamicin 5 mg/kg/daily for 7 to 10 days, but no study directly comparing the 2 regimes is currently available. The optimal treatment for pregnant women, neonates, and children under 8 is not yet determined; for children, options include trimethoprim/sulfamethoxazole (co-trimoxazole) combined with an aminoglycoside (streptomycin, gentamycin) or rifampicin (Figures 1 and 2).

Sagittal contrast-enhanced T1 MRI showing no significant edema of vertebral bodies or disease to suggest spondylodiscitis.

Brucella colonies on blood agar appearing as small, smooth, translucent, and nonhemolytic.
Conclusion
This case highlights the importance of considering brucellosis in the differential diagnosis of breast abscesses, particularly in endemic areas or in patients with risk factors for zoonotic infections It serves as an awareness of the wide range of clinical manifestations of brucellosis and emphasizes how essential early detection and effective treatment are in preventing brucellosis complications. To improve our understanding of this uncommon presentation of a prevalent zoonotic infection more awareness and reporting of such cases are essential (Table 1).
Laboratory Results During Admission and Follow-Up.
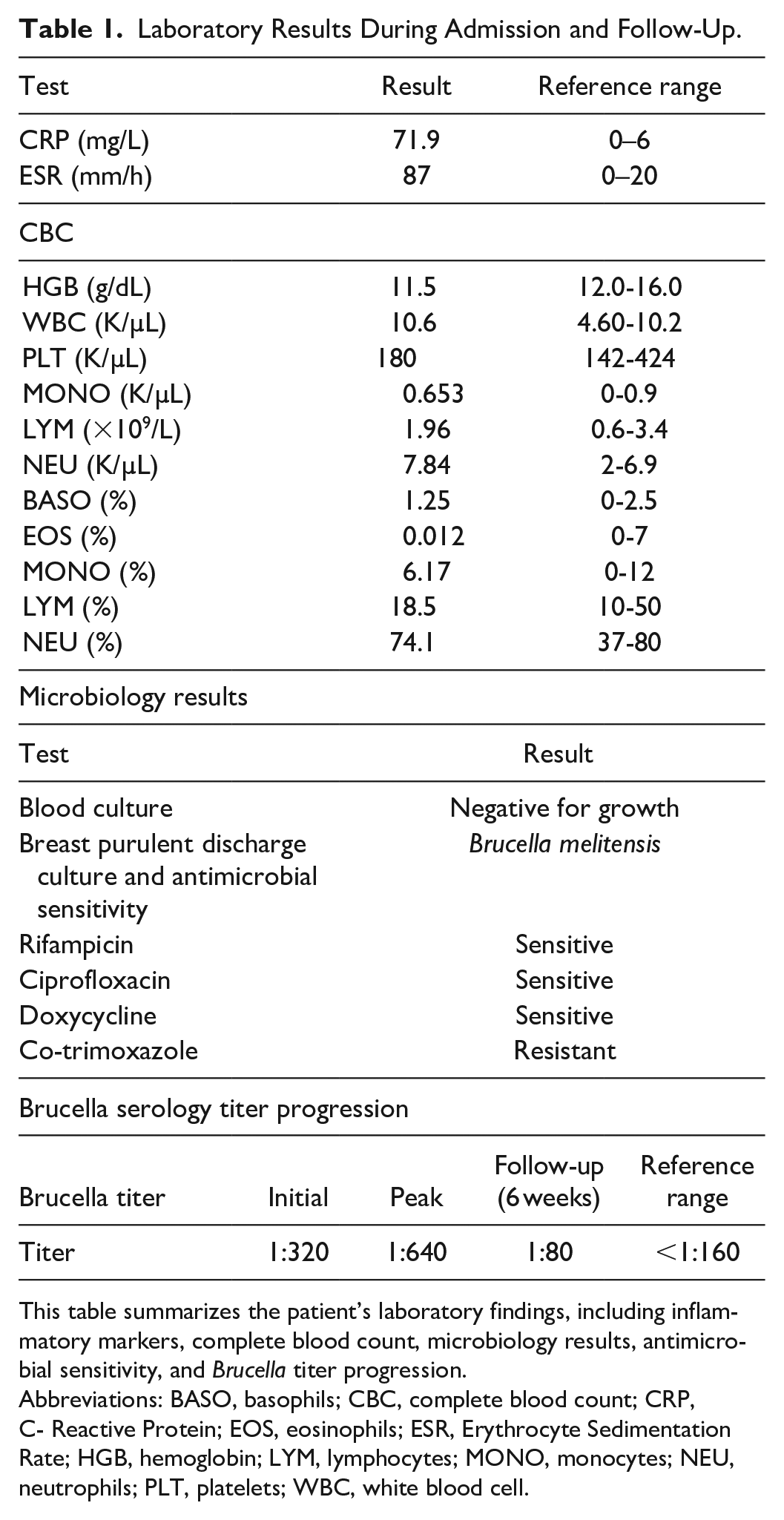
This table summarizes the patient’s laboratory findings, including inflammatory markers, complete blood count, microbiology results, antimicrobial sensitivity, and Brucella titer progression.
Abbreviations: BASO, basophils; CBC, complete blood count; CRP, C- Reactive Protein; EOS, eosinophils; ESR, Erythrocyte Sedimentation Rate; HGB, hemoglobin; LYM, lymphocytes; MONO, monocytes; NEU, neutrophils; PLT, platelets; WBC, white blood cell.
Footnotes
Acknowledgements
We would like to express our appreciation to the Al-Makassed Hospital’s medical and laboratory staff for their tremendous assistance and knowledge during this case study. Their commitment to patient care and their collaboration in diagnosing and managing this unusual presentation of breast abscess secondary to Brucella infection have been crucial. We also acknowledge the academic support provided by Al-Quds University, which facilitated the research and preparation of this report. This case underscores the importance of interdisciplinary teamwork in identifying and managing rare infectious conditions.
Author Contributions
A.D. contributed to writing the first draft and final manuscript. T.A., R.S., and U.G. contributed in the data collection. R.A. supervised the project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for anonymized information to be published in this article.
