Abstract
Immunotherapy has become the standard of care for advanced and resectable lung cancer, and specific mutations may predict immunotherapy response. For example, STK11 mutations, which are more common in African American patients, are associated with immunotherapy resistance. A 68-year-old African American man with stage IIIB lung adenocarcinoma with mediastinal lymph node involvement progressed on first-line concurrent carboplatin-based chemoradiotherapy. Molecular testing of the patient’s subcarinal lymph node tissue revealed STK11 S216F, TP53 R273L, and RB1 splice site mutations; high tumor mutation burden (19.0 mutations/Mb); and high PD-L1 22c3 expression (TPS 70%, 2+ intensity). Treatment with carboplatin-based chemotherapy with radiation therapy failed to control the disease, but the patient has tolerated and responded well to intravenous pembrolizumab. Although STK11 mutations are associated with immunotherapy resistance, our patient demonstrated an exceptional and sustained response to immunotherapy for over two years. The patient’s STK11/TP53 co-mutation, along with high TMB and PD-L1 22c3 TPS scores, may help explain his continued responsiveness to immunotherapy and longer survival. Importantly, incorporating genetic ancestry differences in mutation prevalence and the impact of specific mutations and co-mutations, may help ensure the equitable and optimal treatment of all patients with lung cancers.
Introduction
Immunotherapy, including monoclonal antibodies against programmed death-1 and programmed death ligand-1 (PD-L1), has become the standard of care for both advanced and resectable lung cancer. However, approximately 20% to 27% of patients with advanced non-small cell lung cancer (NSCLC) show primary resistance to immunotherapy, and resistance to chemoimmunotherapy is about 10%. 1 Specific mutations in NSCLC can influence treatment response and survival outcomes. In general, patients with STK11-mutant NSCLC tend to have lower PD-L1 expression and poorer response to immunotherapy.2,3 Recent studies suggest that the impact of STK11 may be modulated by co-mutations, tumor mutation burden (TMB) status, and PD-L1 expression, influencing both immunotherapy response and patient survival. 4 For example, NSCLC patients with co-mutations in STK11 and TP53 tend to respond better to immunotherapy. 4 Compared to STK11-mutated/TP53 wild-type tumors, co-mutated tumors exhibit higher TMB and exome-level neoantigen load, possibly due to accumulated DNA damage from mutated TP53, increasing immunogenicity and immunotherapy efficacy. 5
Emerging data suggest genetic ancestry differences, such as higher rates of STK11 and RB1 mutations in African Americans with lung adenocarcinomas (25% and 14%, respectively), 6 may additionally impact mutation prevalence and treatment response. We describe an African American patient with metastatic lung adenocarcinoma who has responded exceptionally well to pembrolizumab despite the presence of an STK11 mutation.
Case Presentation
A 68-year-old African American man with a 32-pack-year smoking history and chronic obstructive pulmonary disease presented to the hospital with progressive dyspnea, productive cough, and 10 kg weight loss over 6 months. A computed tomography (CT) scan of the chest, abdomen, and pelvis revealed a 6.3 × 4.8 cm mediastinal mass within the azygoesophageal recess.
Esophagogastroduodenoscopy was negative for any masses within the upper gastrointestinal tract. Endobronchial ultrasound-guided transbronchial needle aspiration of the subcarinal lymph node showed metastatic adenocarcinoma. The patient was considered to have T3N2M0 lung adenocarcinoma, pathological stage IIIB. Molecular profiling (NeoType DNA/RNA Lung at NeoGenomics, Fort Myers, FL) on formalin-fixed paraffin-embedded tissue block from the subcarinal lymph node sample detected STK11 S216F, TP53 R273L, and RB1 splice site mutations; and high TMB (19.0 mutations/Mb) and PD-L1 22c3 Tumor Proportion Score (TPS) 70%, 2+ intensity (Figure 1).
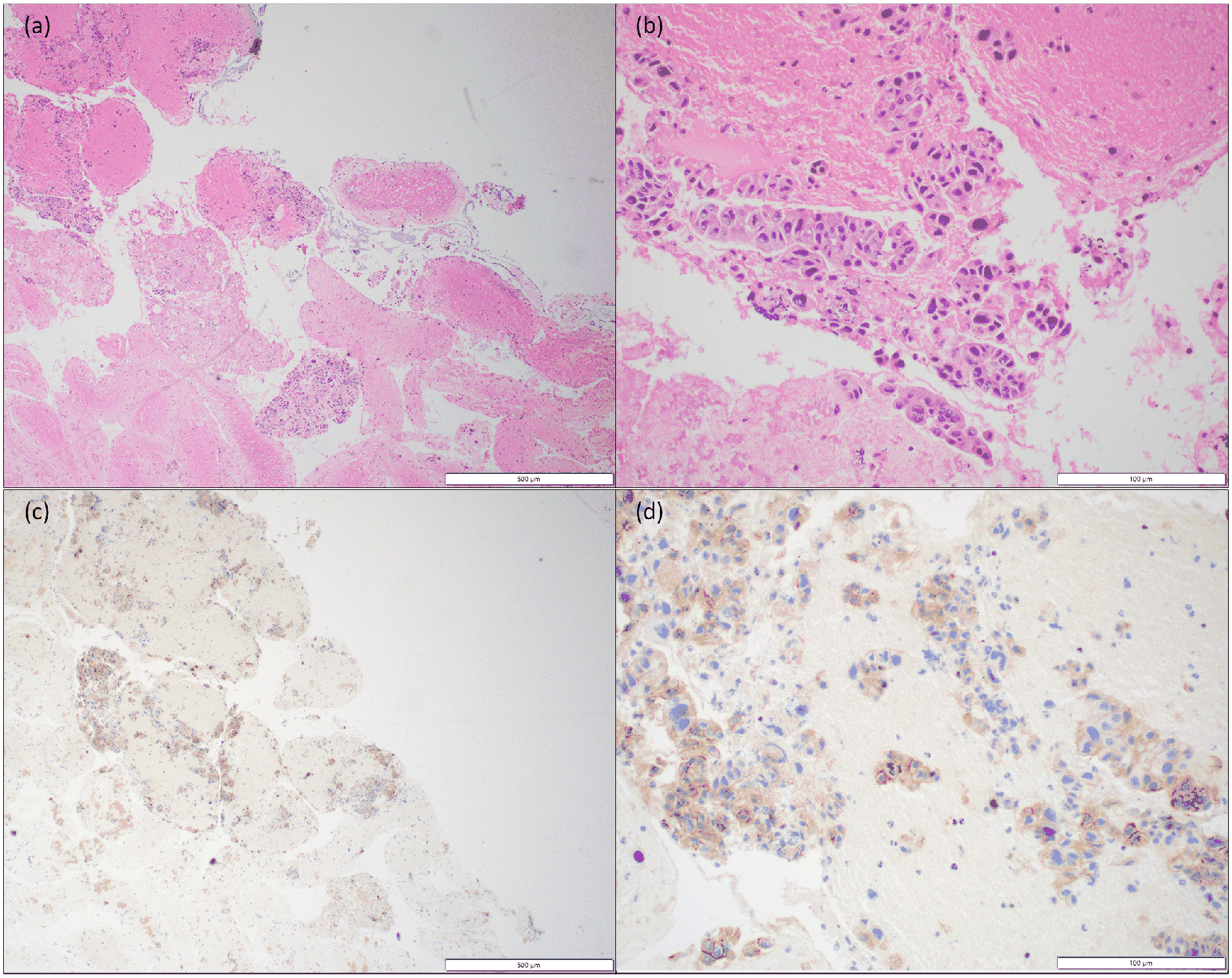
Representative images from the subcarinal lymph node fine needle aspiration cytology cell block (performed 1 month after initial presentation). Hematoxylin and eosin (HE) images at 4× (a) and 20× (b) show clustered and individual tumor cells in a necrotic background. Tumor cells were positive for BerEP4, B72.3, CK7, and negative for CK20, TTF-1, Napsin A, p40 immunohistochemical stains (not shown). The pathologic diagnosis was metastatic adenocarcinoma (i.e., lung adenocarcinoma). PD-L1 22c3 FDA NSCLC showed 2+ intensity and tumor proportion score (TPS) of 70%, as shown at 4× (c) and 20× (d). The white scale bars for 4× and 20× indicate 500 and 100 μm, respectively.
The planned treatment was carboplatin-based chemotherapy and radiation therapy (CRT) for 6 weeks, followed by 1 year of intravenous (IV) durvalumab based on the PACIFIC trial. 7 At the initiation of chemoradiation, the mediastinal mass was 8.5 × 5.8 cm (Figure 2a). The patient tolerated the CRT but developed grade 3 neutropenia, which required holding concurrent carboplatin and paclitaxel during the last 2 weeks. Two weeks after completion, the patient presented with shortness of breath and fatigue and was admitted for anemia work-up. CT exam showed progression of disease with multiple enlarged nodes and a new 2.8 × 2.6 cm left lower lobe lung mass, but his mediastinal tumor had reduced in size from 3 months ago (Figure 2b).

(a) At the start of chemoradiation, computed tomography (CT) scan of the chest, abdomen, and pelvis with contrast showed a large hypo-enhancing mediastinal mass measuring 8.5 × 5.8 cm in axial dimension, increased compared to 6.5 × 4.8 cm measured two months previously. (b) After 6 weeks of treatment with carboplatin-based chemotherapy with radiation therapy, repeat imaging CT of the chest, abdomen, and pelvis with contrast showed a new lung mass and increased size of multiple lymph nodes, but a decrease in size of the mediastinal mass, measuring approximately 5.5 × 4.3 cm compared with 8.5 × 5.8 cm previously. (c) After 3 months of treatment with pembrolizumab 200 mg IV 3 weeks, CT scan imaging of the chest with contrast showed improvement of mediastinal adenopathy and partial improvement of previously noted lymph nodes and lung masses.
Second-line treatment using IV pembrolizumab 200 mg was started every 3 weeks according to the KEYNOTE 024 trial. 8 The patient tolerated the infusions with no significant autoimmune toxicity. His dyspnea and fatigue improved, and a CT scan after 3 months of immunotherapy showed radiologic improvement with partial response per iRECIST criteria (Figure 2c). The patient tolerated IV pembrolizumab 200 mg every 3 weeks and has continued immunotherapy for over 2 years (Figure 3).

Discussion
Our patient’s STK11/TP53 co-mutations and high TMB and PD-L1 22c3 may help explain his continued exceptional immunotherapy response. However, more research is needed to develop comprehensive, personalized treatment strategies that account for genetic and environmental factors across diverse populations. Tumor mutation differences among smokers versus nonsmokers and individuals of different ancestry, such as East Asians having higher rates of EGFR mutations, have been well-documented. But differences in tumor biology and treatment response in other groups are less well understood, partly because of the lower representation of minorities in clinical trials and genomic databases. 9 In the United States, African Americans’ worse lung cancer outcomes have largely been attributed to socioeconomic factors, such as inequitable healthcare access, contributing to a later stage of diagnosis, lower rates of appropriate biomarker testing and targeted therapy, and higher mortality rates.10 –12
While socioeconomic factors undoubtedly play a significant role in lung cancer outcomes, it is also important to acknowledge potential biologic differences between patient groups. For example, the frequency of STK11 mutations, which are associated with inferior survival outcomes and resistance to immunotherapy,1 –5 vary across subgroups.6,13,14 Its prevalence has been reported to be 5% in Chinese patients with lung cancers, 13 but as high as 43% among patients in the United States. 14 STK11 mutation is additionally associated with a history of tobacco smoking and African ancestry, and both STK11 and RB1 mutations are reportedly more common in African Americans.6,14
Moreover, the therapeutic implications of tumor mutations extend beyond the mere presence of mutations in candidate genes. In addition to the association of specific genetic mutations for targeted treatment (eg, responsiveness to tyrosine kinase inhibitors in different EGFR mutations), there is also emerging evidence that immunotherapy responsiveness may be associated not only with the specific gene that is mutated but also the specific mutation within individual genes. 15 For example, despite the general acceptance that immunotherapy is ineffective in EGFR-mutant NSCLC patients, those with EGFR L858R appear to benefit more from immunotherapy than those with EGFR 19del.15–17
Conclusion
As our case and the existing literature demonstrate, it is necessary to consider the holistic biomarker status in treating individual patients, especially those from underrepresented populations, where clinical data may be limited. Importantly, designing clinical trials that include historically underrepresented groups, such as African Americans, who have higher rates of STK11 mutations and elevated lung cancer mortality, may improve cancer outcomes globally. Evaluating genetic ancestry would avoid the problematic social constructs of race and ethnicity.18,19 However, given the practical challenges of recording genetic ancestry, existing classification schemes may sometimes be the only available surrogate. 20 Ultimately, it is crucial for the medical and research community to strive for clinical research that integrates genetic ancestry and mutation, and disease prevalence to achieve more personalized and equitable treatment for all patients.
Footnotes
Acknowledgements
Ms Teresa Volk Practice Manager ECU Health Cancer Care, Ms Carla Bass RN, Joycelynn Harrell, RN, OCN, and the entire ECU Edgecombe Cancer Center Tarboro team for coordinating care and helping the patient in a difficult socioeconomic situation. We also thank the patient for providing consent to publish this report.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Since March 2023, Dr Zhou has been expanding her molecular service, as part of a quality improvement initiative made possible by a grant from Eli Lilly and Company; however, Lilly has had no role in the preparation, review, approval, or decision to submit the manuscript. Dr Zhou has received honoraria from medical and life sciences consulting companies for consulting in double-blinded molecular pathology projects. Dr Zhou’s unpaid professional service has included serving as an American Society for Clinical Pathology (ASCP) Choosing Wisely Advisory Board member, and in various capacities for the Association for Molecular Pathologists (AMP).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This patient’s molecular test request was initially evaluated by Dr Zhou as part of her standard molecular pathology review service. Dr Badami subsequently formally ordered a molecular pathology consultation to request Dr Zhou’s review of the patient's clinical history, prior imaging and other studies, pathologic and molecular data, and published literature, to inform his treatment decisions. The molecular consultation service was made possible by a grant from Eli Lilly and Company; however, there has been no dedicated funding for the specific case submitted.
Ethics Approval Statement
Our institution does not require ethical approval for reporting individual cases or case series.
Patient Consent Statement
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
