Abstract
Breast cancer (BC) accounts for 24.2% of all women’s malignant tumors, with rising survival rates due to advancements in chemotherapy and targeted treatments. However, second primary cancers, particularly lung cancer (LC), have become more prevalent, often emerging approximately 10 years after BC treatment. This study presents a case series of 9 women diagnosed with second primary LC following BC, treated at a high-complexity hospital in Colombia between 2014 and 2019. All initial BCs were ductal carcinomas, 7 were triple negative, 1 was human epidermal growth factor receptor 2 positive, and 1 was estrogen and progesterone positive. Each patient had undergone radiation therapy, and 7 had received chemotherapy, increasing their LC risk. The second primary LCs, all adenocarcinomas, were confirmed using immunohistochemical stains for thyroid transcription factor-1 (TTF-1), Napsin A, and estrogen receptor (ER) status. The interval between treatments and LC detection ranged from 1 to 17 years, with 4 cases identified after 10 years and 3 within 1 to 3 years, underscoring the need for prolonged surveillance. Seven LCs were ipsilateral to the BC and radiation site, while 2 were contralateral, highlighting the necessity of monitoring both sides for potential LC development. This case series enhances the local epidemiological understanding, showing that prior radiotherapy for BC and histological analysis are key in characterizing second primary LC patients. The study emphasizes the critical role of accurate histological diagnosis in guiding treatment approaches for lung lesions in BC survivors.
Introduction
Cancer is a prevalent disease worldwide, with a global incidence of 18.1 million new cases in 2020, according to the latest data reported by Globocan. In addition, 9.8 million people died from cancer that year. 1 However, cancer survival rates have improved in recent years, thanks to advances in early detection strategies and the advent of immunotherapy and targeted therapies, which complement conventional treatments, such as surgery, chemotherapy, radiotherapy, and hormonal therapy. 2
Breast cancer (BC) is the most common malignancy in women worldwide. It accounts for approximately 24.2% of all cancer cases and is the leading cause of death (15.0%), followed by lung cancer (LC) (13.8%). 3 In Colombia, BC is the second leading cause of death in women after cervical cancer. In 2013, it was reported that approximately 7000 new cases were diagnosed annually in this country, with an associated mortality of 2500 women per year.4-6 On the other hand, advances in BC screening and treatment techniques have improved survival rates in recent decades. 7
Despite BC patients experiencing prolonged survival, this has led to an increased incidence of long-term complications, consequently raising the risk of developing a second primary cancer (SPC). 7 It is estimated that 16% (1 of 6) of diagnosed tumors in the United States correspond to an SPC.8,9 Among the factors associated with this occurrence are increased long-term survival, treatment of the primary neoplasm (radiation therapy), and thorough clinical follow-up with routine diagnostic imaging, which allows for the more sensitive detection of a new neoplasm. 10
Large-scale studies have reported that the most common site of SPC after BC is LC.7,11 At least, 10% of patients who undergo post-mastectomy radiotherapy have been reported to have an increased risk of developing an SPC of the lung.12,13 A large-population study highlighted that women with BC who underwent radiation therapy showed a higher incidence of LC compared with those who did not receive radiotherapy and women without BC. 14 Second primary LC is more likely to develop on the same side where the patient received radiotherapy and its primary histological type is non-small cell.12,15
Other associated risk factors for developing second primary LC include smoking, early diagnosis, black ethnicity, triple-negative BC, and undifferentiated histological grade. 15 Prior research has also explored the link between pathogenic genes associated with both BC and LC, including BRCA2, TP53, RAD51D, EXT2, WWOX, GATA2, and GPC3, suggesting a potential genetic predisposition to the occurrence of both primary malignancies. 16 However, no distinction has been observed between primary LC patients with and without a history of BC, which calls into question the presumed predisposition to develop an SPC. 17
A latency period of around 10 years has been described for the second primary LC to appear,12,15 with a 40% higher risk of developing an SPC if the patient has survived 10 years beyond the BC diagnosis. 7 In addition, mortality rates from LC in individuals previously treated for BC have been noted to be higher among those diagnosed with advanced BC. 18
One of the challenges encountered when a second primary LC is suspected is defining whether it is indeed a primary LC or a metastatic cancer from the previous BC. To aid in this differentiation, several biomarkers have been proposed, such as caudal type homeobox 2 (CDX2), cytokeratin 7 (CK7), cytokeratin 20 (CK20), and thyroid transcription factor-1 (TTF-1) panels. Research indicates that a pulmonary tumor positive for TTF-1, a specific marker for primary lung adenocarcinomas, is unlikely to be metastatic. Similarly, a tumor that is CK7 negative is less likely to represent a primary LC.19,20
Other clinically relevant biomarkers for LC include epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase (ALK), mesenchymal-epithelial transition factor (MET), ROS proto-oncogene 1 (ROS-1), and Kirsten rat sarcoma viral oncogene homolog (KRAS). 21 Furthermore, patients with a second primary LC following primary BC have shown higher rates of EGFR mutation compared with cases of first primary LC. 22 Programmed death-ligand 1 (PD-L1) has also become a relevant biomarker as it provides information on whether an LC patient would benefit from immunotherapy directed toward this biomarker. 23
Most of the evidence cited above is based on reports from studies that mainly included Asian and European patients, whose characteristics differ from those of the Colombian and Latin American populations. Currently, there are no epidemiological data on second primary tumors after BC and the associated molecular and histological risk factors that would determine their prevalence in our specific region.
Therefore, we present a case series of 9 patients with second primary LC after BC (BC-LC) in a tertiary care hospital in Colombia. Clinical and molecular aspects are described as reported at the initial diagnosis and during the follow-up period.
Case Series Presentation
This case series describes demographic, diagnostic, therapeutic, and molecular profile data of patients with SPC of the lung with a history of BC diagnosed between 2014 and 2019 at a tertiary referral hospital in Colombia. The data source consisted of digital medical records. Qualitative variables were presented as absolute and relative frequencies. For quantitative variables, measures of central tendency and dispersion were used according to the distribution of the analysis data, evaluated by the Shapiro-Wilk test. Mean and standard deviation were used when the distribution was normal, otherwise median, and interquartile range estimates were applied. No adverse events were encountered.
As this study involved human participants, the ethical principles for research involving human participants, as stated in the Declaration of Helsinki of 2013 and Resolution 8430 of Colombia of 1993, were followed. The Institutional Ethics Committee evaluated and approved this study.
Breast Cancer
Nine female patients with a prior history of BC were diagnosed with LC between 2014 and 2019, corresponding to an SPC after BC. No diagnostic challenges were reported. Their main demographic characteristics are shown in Table 1. Data on the patients’ ages and stage at diagnosis are shown in Table 2; the mean age at diagnosis of BC was 56.2 ± 10.8 years (ranging from 35 to 73 years). All cases were ductal carcinomas; 77.8% were located on the right side of the body and 22.2% on the left. The required data to determine the BC stage were not available for 2 cases as their medical records did not provide sufficient clinical information.
Demographic Characteristics.

Abbreviation: OSA, obstructive sleep apnea.
The patient had no clinical follow-up, only the diagnosis was made at our hospital.
Breast Cancer Characteristics.

Abbreviations: ER, estrogen receptor, PR, progesterone receptor; y, time in years.
The patient had no clinical follow-up, only the diagnosis was made at our hospital.
Time in months from the diagnosis of BC to the last medical examination.
Concerning the hormonal markers, 7 cases were triple-negative ductal carcinoma, with negative progesterone receptor (PR–) and negative estrogen receptor (ER–), 1 case was human epidermal growth factor receptor 2 (HER2) enriched BC (hormone receptor-negative, HER2 score 3+), and 1 case was positive for estrogen (ER+) and progesterone (PR+) only. Six patients received hormonal therapy (anastrozole in 2 cases and tamoxifen in 1), with no further changes. All patients received radiotherapy for BC. Of these, 8 received it before the diagnosis of LC and 1 received it concomitantly. The mean latency between the 2 diagnoses was 8.1 years (SD = 6.2 years; range 0.5-17.5).
Secondary Primary LC
All patients developed primary lung adenocarcinoma, 3 of whom had EGFR mutations, 2 had ROS-1 rearrangements, and 3 had high PD-L1 expression. No ALK gene rearrangements were identified in this series. The average tumor size was 22 mm. Six tumors were peripheral lung nodules (< 3 cm) and the remaining were masses larger than 3 cm in maximum diameter. Seven of 9 LC cases (77.8%) were developed ipsilaterally, while 2 (22.2%) developed contralaterally. The LC characteristics and stage at the time of diagnosis are shown in Table 3. In one of the cases, data were not available because the patient was lost to follow-up.
Lung Cancer Characteristics.

Abbreviations: NYHA, New York Heart Association; y, time in years; IHC, immunohistochemistry.
The patient had no clinical follow-up, only the diagnosis was made at our hospital.
Exon 19 deletion (EGFR mutation).
Time in months from the diagnosis of LC to last medical examination.
Concerning their treatment, all patients received radiotherapy, 3 in combination with chemotherapy, 5 underwent resection surgery, 2 received a tyrosine kinase inhibitor (erlotinib), and 2 received immunotherapy (pembrolizumab), with adequate tolerability. Only 1 patient (no. 9) died during the follow-up period, 69 months after the diagnosis of BC, and 7 patients were followed up in our hospital, with satisfactory recovery. The radiological and histopathological findings from relevant cases, such as patients 1, 2, 3, 5, 7, and 9, are shown in Figures 1–8.

Patient 7. (A, B) Chest radiograph (posteroanterior and lateral views): A homogeneous opacity is seen in the right lung base with the loss of normal borders of the heart and diaphragm, which may suggest right pleural effusion. The trachea appears centered. There are no other visible alterations in the lung parenchyma. (C, D) Hematoxylin and eosin (H&E). 10× and 40×, a pleural metastatic malignant lesion is observed, consisting of large cells of eosinophilic cytoplasm and conspicuous nucleoli, with solid a solid pattern distribution, consistent with solid pattern adenocarcinoma originating in the lung. Mutations for EGFR exons 18, 19, 20, and 21; Echinoderm microtubule-associated protein-like 4 (EML4)-ALK rearrangements, and PD-L1 protein expression were all negative.

Patient 1. Computerized tomography (CT) chest. (A) Mediastinal window. (B, C) A subpleural nodule of 13 × 10 mm is seen in the anterior segment of the right upper lobe, showing irregular borders, which, when compared with previous studies, has increased in size. There are no signs of pleural effusion or thickening. The tracheobronchial tree is normal without endoluminal defects; there are no lytic or blastic lesions in the visible bone structures. (D, E) Lung biopsy was performed via video thoracoscopy. On H&E staining at 10× and 20×, a malignant neoplastic lesion is seen, made of desmoplastic stroma, large cells, clear cytoplasm, arranged in a glandular pattern. An enteric lung adenocarcinoma of classic morphology (70%) was diagnosed based on morphological pattern and immunohistochemical expression profile.

Patient 2. (A, B, C) Positron emission tomography-CT (PET-CT). The following are seen: a 13 mm hypermetabolic pretracheal lymphadenopathy with standardized uptake value (SUV) max 4.2 and 4.8 g/mL. A 12 mm subcarinal lymphadenopathy with SUV max 5.8 g/mL. Hypermetabolic right pulmonary hilum. A 19-mm hypermetabolic nodule in the apical segment of the right upper lobe with irregular spiculated borders with SUV max 10.3 g/mL. Normal metabolic activity in the rest of the visualized structures is seen.

Patient 2. Endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) of the patient described in Figure 3. (A) The tracheobronchial mucosa is seen without endoluminal lesions. (B) Real-time puncture of subcarinal adenopathy. (C, D) Cellblock of a sample obtained using EBUS-TBNA (H&E staining), 20×, and 40×. A metastatic malignant neoplastic lesion of epithelial origin is shown, which is made up of eosinophilic cytoplasm cells, central nuclei, and inconspicuous nucleoli which match an adenocarcinoma. The immunohistochemical technique demonstrates nuclear positivity for TTF-1 and negativity for p40, corresponding to primary lung adenocarcinoma. Study for EGFR gene mutations in exons 18, 19, 20, and 21, EML4-ALK rearrangements were negative and positive for high expression of PD-L1 (TPS: 80%).

Patient 3. (A, B) PET-CT (patient no. 9) showing a right paratracheal 10 mm hypermetabolic lymphadenopathy with SUV max 5.8 g/mL; and a 9 mm low right paratracheal lymphadenopathy with SUV max 4.3 g/mL. A 30 × 26 mm hypermetabolic mass is seen adjacent to the right lung fissure, with SUV max 11.23 g/mL, and an adjacent satellite 10 mm node with SUV max 4.7 g/mL. (C, D). Chest CT scan as a guide for needle percutaneous biopsy of the tumor.
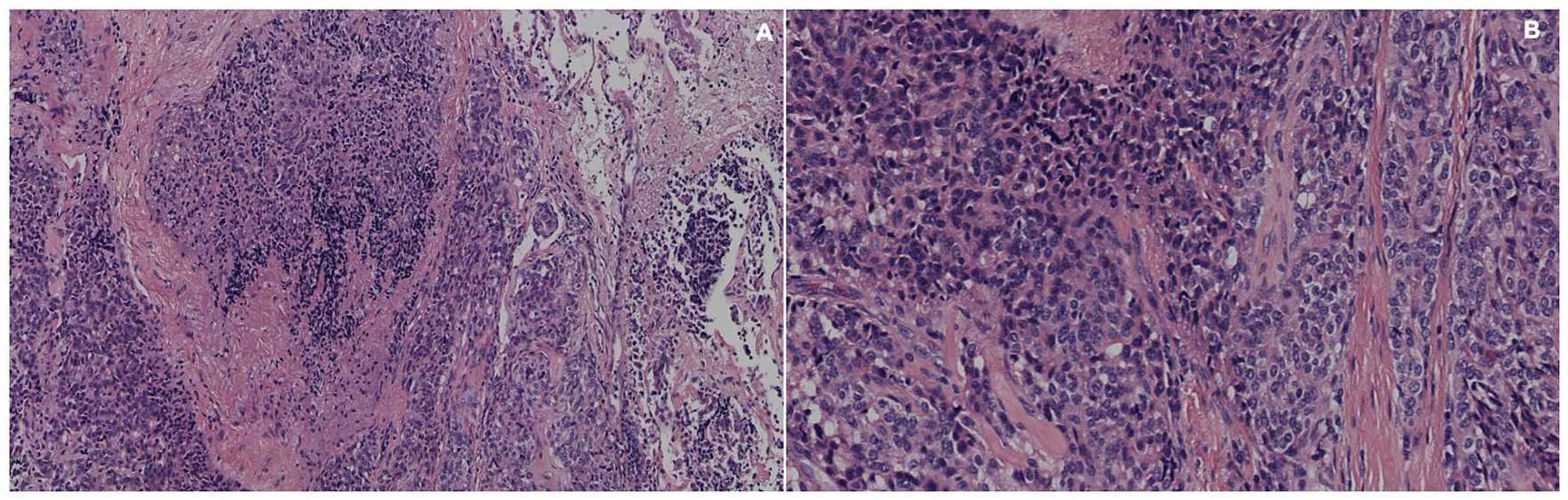
Patient 3. Percutaneous needle biopsy of the patient in Figure 5; H&E staining at 4× and 10×, showing a malignant neoplastic lesion, composed of large cells, eosinophilic cytoplasm, central nuclei, and diffuse morphology. A solid pattern adenocarcinoma of the lung is diagnosed, based on morphological pattern and immunohistochemical expression profile.
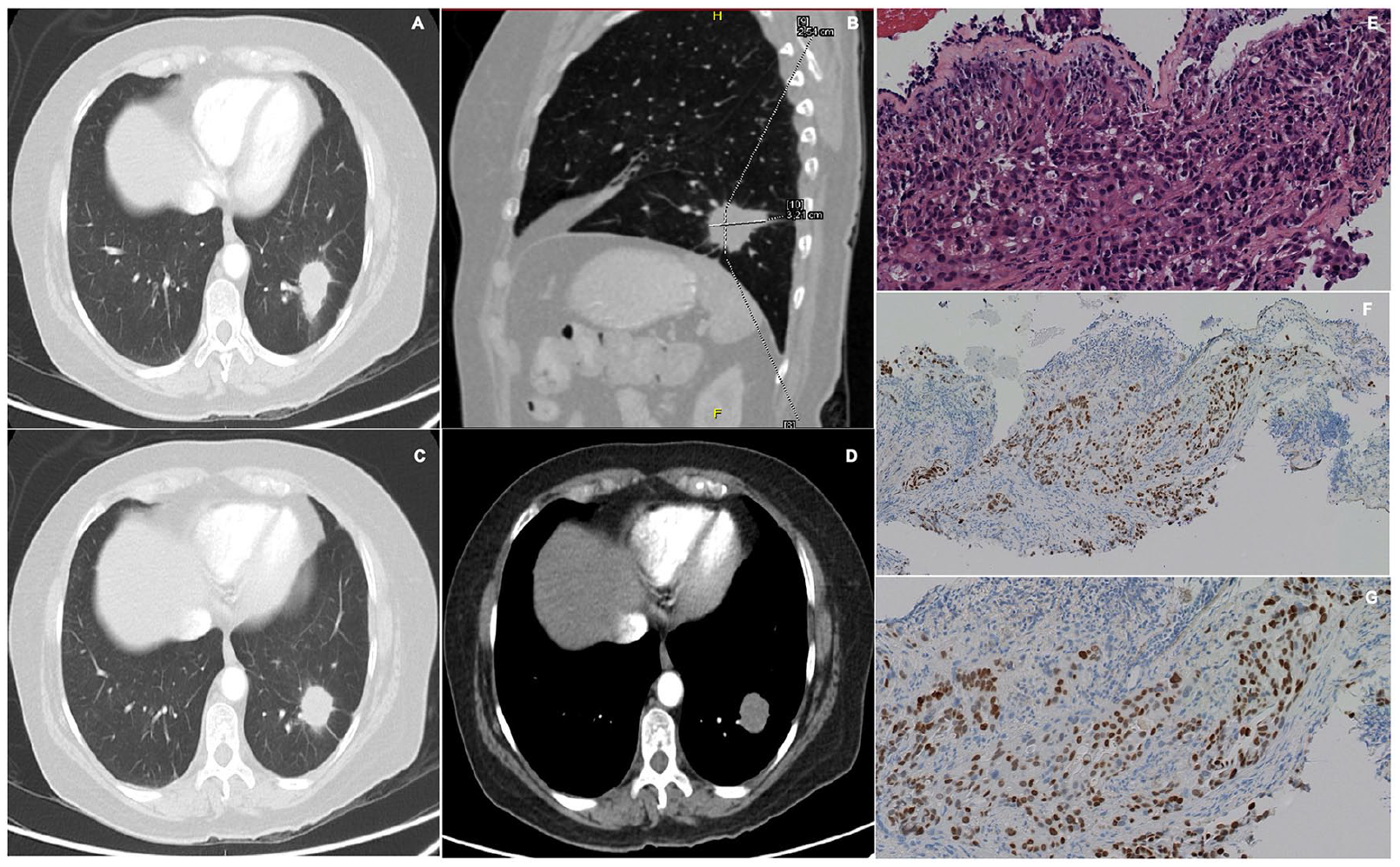
Patient 5. (A, B, C, D) Chest CT showing lung parenchyma with normal morphology and density. A 29 × 34 mm pulmonary nodule is found in the lateral segment of the left lower lobe, with poorly defined borders and spiculated edges associated with linear opacities that distort the adjacent lung parenchyma architecture. (E) Lung biopsy via video thoracoscopy. H&E staining, 20×, showing a malignant tumor of epithelial origin, consisting of large cells, with a solid pattern infiltrating the respiratory mucosa. (F and G) IHC confirmed the pulmonary origin of a solid adenocarcinoma showing nuclear expression of TTF-1 at 4× and 20×. Tumor cells were positive for a mutation of exon 21 of the EGFR gene (L858R), were negative for EML4-ALK rearrangements, and had low PD-L1 expression (TPS: 3%).

Patient 9. (A, B) Chest CT showing an irregular-looking nodule at the right lung base. (C, D) Lung biopsy via video thoracoscopy. H&E staining at 4× and 20× shows a malignant neoplastic lesion made of large cells with a large eosinophilic cytoplasm, which constitutes a solid and glandular pattern. IHC shows nuclear positivity for TTF-1 and CK7. ER, PR, and CK20 were negative. The definitive diagnosis was a solid pattern adenocarcinoma of the lung, with an L858R mutation in exon 21 of the EGFR gene.
Discussion
The incidence of LC as an SPC is on the rise due to the prolongation of survival in BC as a result of advances in current treatment. As previously reported in the literature, there appears to be an association between ductal carcinoma in situ and primary lung adenocarcinoma as the most common histological types found in these cases. 15 Nine patients with primary LC, predominantly non-small cell lung cancer (NSCLC) adenocarcinoma subtype, who had previously had ductal carcinoma of the breast, mostly triple negative, were identified over 5 years for this study.
All of our patients received radiation therapy for BC, which potentially increased their risk for developing LC. Seven of 9 patients also underwent chemotherapy, which might have further contributed to the development of LC, although a second primary LC could also be solely attributed to breast radiotherapy.24,25 All the LCs identified were lung adenocarcinomas. Immunohistochemical stains for TTF1, napsin A, and ER status (all ER–) confirmed these were LCs, not metastases from BC, highlighting the importance of such immunohistochemistry (IHC) characterization.
The detection of second primary LCs ranged from 1 to 17 years post-BC treatment. Four of 9 cases were identified more than 10 years after BC treatment, while 3 were detected within 1 to 3 years, underscoring the need for vigilant surveillance for second primary LCs throughout BC follow-up. Although 7 of 9 LCs occurred ipsilaterally with respect to the BC and radiation treatment, 2 occurred contralaterally. Therefore, it is crucial to monitor both contralateral and ipsilateral pulmonary lesions for potential LC development.
According to some reports, women treated for BC, especially with radiotherapy, have an increased risk of developing new primary tumors, such as LC compared with the general population.24,25 In addition, second primary LC after radiotherapy for BC is more likely to develop ipsilaterally. 12 All patients from our study received radiotherapy for BC and subsequently developed primary LC.
Among our patients, 7 developed the SPC ipsilaterally, while 2 developed it contralaterally. These findings are consistent with those reported in previous studies. Nevertheless, our study is limited by the relatively small number of patients included. A larger population would be necessary to draw definitive conclusions regarding the impact of the laterality of SPC development after radiotherapy.
In recent years, there has been an improvement in diagnosing patients with BC-LC, particularly NSCLC, at earlier stages. This trend could be attributed to the growing utilization of radiological imaging during the follow-up of BC patients receiving treatment.26,27 Nonetheless, a significant proportion of patients in this study were diagnosed at advanced stages (IIIB and IVB).
According to clinical guidelines in our country, patients are advised to adhere to biannual follow-up appointments during the initial 3 years post-treatment, followed by annual assessments. In addition, bilateral mammography is recommended annually, 6 months post-surgery. Subsequent imaging investigations may be prompted in the event of patient-reported symptoms. 28
There is a lack of consensus regarding incorporating laboratory tests or imaging modalities as screening tools for asymptomatic patients without discernible clinical manifestations during follow-up assessments. 29 However, if a suspicious lesion is detected during the follow-up period, it should be investigated promptly as it could indicate the development of a metastasis or an SPC, with differentiation typically achieved through biopsy.15,30
The latency period for the development of an SPC has been calculated to be approximately 10 years after the treatment of the first primary cancer,31-37 which can vary from 1 to 26 years. 38 In our case series, the median time for the development of a second primary LC was 8.1 years. This is consistent with the latency time reported in several studies.
Regarding the relationship with hormone receptors, a higher incidence of lung adenocarcinoma has been observed in BC patients with negative ER and PR expression compared with ER+ tumors, suggesting that there may be common etiological factors between the 2 types of cancers.39,40 In contrast, cases of second primary lung adenocarcinomas reported in other studies show a high EGFR expression associated with triple-negative BC compared with patients with lung adenocarcinoma alone, which may also suggest a common pathway.15,22,39
No previous studies that described an association between the development of a typical lung carcinoid tumor and any histological type of BC were found. As the typical carcinoid tumor is a less common neuroendocrine tumor within the NSCLC type, it is usually reported as “another NSCLC,,” and it is not possible to determine a clear relationship or frequency for this type of tumor in the reviewed literature.
The development of lung neoplasms in patients with previous BC often leads to a misdiagnosis, commonly confused with metastasis. Therefore, obtaining biopsies and basic immunohistochemical panels that include CDX2, CK7, CK20, and TTF-1 is recommended to differentiate primary LC from metastatic LC. 19 In our experience, the main requested biomarkers include TTF-1 and CK7, although in some cases, other markers, such as napsin A and CK20 are also requested.
Immunohistochemistry was applied to all of our patients’ LC biopsies, revealing positivity for TIFF-1 in all cases, as well as CK7, CK20, and napsin A when requested. This served as the definitive parameter to classify these LC cases as primary rather than metastatic, as a combination of immunomarkers is recommended for this differentiation. 41
Other biomarkers, such as EGFR, ALK, PD-L1, MET, KRAS, and ROS-1, are also essential to reduce empirical diagnosis, which can lead to inappropriate treatment.21,42 In our institution, additional molecular testing also includes EGFR, ALK, PD-L1, MET, and ROS-1; however, KRAS is not tested since no treatment targeted to this molecular marker has been approved in our country.
On the contrary, PD-L1 serves as a crucial biomarker for guiding immunotherapy as pembrolizumab, an approved medication, has demonstrated efficacy in extending the survival of individuals with NSCLC who exhibit high expression of this biomarker (tumor proportion score [TPS] greater than 50%). 23 Among the 3 patients exhibiting high PD-L1 expression, 2 underwent treatment with pembrolizumab, resulting in positive outcomes. However, the third patient did not return for follow-up at our institution, thus precluding further treatment information.
Making an accurate diagnosis through biopsies and molecular analysis is particularly important in patients with triple-negative BC, as described in this case series. A higher incidence of second primary LC has been observed in these cases, possibly due to common oncogenic pathways.15,43 Moreover, the prognosis of a second primary LC following BC is worse than that of patients with a single primary LC. 44 Therefore, all patients with a history of BC, especially those with triple-negative BC, should be closely monitored and receive a multidisciplinary approach.
Conclusions
This case series describes patients who developed a second primary LC after a primary BC. Histological cancer types appear to play a significant role in this association, particularly ductal BC and lung adenocarcinoma. In addition, it is worth noting that all patients identified with BC-LC had a prior history of radiotherapy for BC treatment. Although our study has limitations, such as relying on medical records for retrospective analysis, it contributes to a better understanding of the local oncological epidemiology. It is important to continue conducting studies on larger samples to establish associations between different histological types in both pathologies and to determine the incidence of second primary LC in the overall population of BC patients, as well as other types of cancer not included in this case series.
Footnotes
Author Contributions
All authors have read and approved the manuscript and significantly contributed to this paper. LFS: Conception and design, literature review, manuscript writing, and correction, final approval of the manuscript. AEO: Conception and design, final approval of the manuscript. VZ-R: Literature review, manuscript writing and correction, final approval of the manuscript. CDI: Data collection, literature review, and final approval of the manuscript. NQ: Data collection, literature review, final approval of the manuscript. LF-T: Conception and design, literature review, manuscript writing and correction, and final approval of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data related to our study have not been deposited in a publicly available repository. Nevertheless, all data and materials will be available upon request by contacting the correspondence author (Liliana Fernández-Trujillo,
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This manuscript was written in compliance with the ethical standards of the institutional ethics committee and with the 1964 Helsinki Declaration. We have the approval of the Ethics Committee in Biomedical Research from Fundación Valle del Lili. This is supported in letter No. 331 of 2019, which is available with the corresponding author if needed.
Informed Consent
Written informed consent was obtained from the patients to publish this case series and any accompanying images. A copy of the written consent is available for review if needed.
