Abstract
Recurrent deep vein thrombosis (DVT) is a common complication, particularly in cancer patients. We present a case of a 50-year-old male with a history of a previous DVT. Despite therapeutic anticoagulation with apixaban, the patient developed recurrent DVT involving the common femoral, popliteal, and foreleg veins. He was started on a heparin drip in the hospital, but despite this, his DVT worsened. Imaging studies revealed mediastinal and hilar lymphadenopathy, raising suspicion of malignancy. The workup revealed elevated tumor markers, while the thrombophilia panel was unremarkable. Despite aggressive management, including mechanical thrombectomy, catheter-directed thrombolysis, and the placement of an inferior vena cava filter, the patient’s condition continued to deteriorate. A biopsy of the mediastinal lymph nodes revealed poorly differentiated mucinous adenocarcinoma with molecular analysis consistent with hepatobiliary origin, and the patient was diagnosed with stage IVB gallbladder cancer. Given the rarity of gallbladder cancer and its association with hypercoagulability, this case highlights the importance of considering cancer as an underlying cause of recurrent DVT after ruling out common causes. Early recognition and a comprehensive diagnostic approach are essential for managing such cases. The patient was started on chemotherapy while maintaining anticoagulation for recurrent DVT.
Introduction
Recurrent deep vein thrombosis (DVT) occurs frequently, with a recurrence rate of about 26.1% within 5 years. Key risk factors include unprovoked initial DVT, older age, malignancy, and persistent conditions like thrombophilia. 1
Hypercoagulability is a common complication associated with malignancy. Patients with cancer are 4 times more likely to develop venous thromboembolism (VTE), and chemotherapy increases this risk 6 to 7 times compared to the general population. 2 The underlying mechanism is multifactorial, involving the direct secretion of prothrombotic factors by malignant cells, and the subsequent activation of clotting pathways within the host. 3 Cancer-associated thrombosis is a leading cause of death in cancer patients, as fatal pulmonary embolism is 3 times more likely in these patients compared to noncancer patients. 4 Studies have also shown that the risk of VTE is more likely in the first 3 months after cancer diagnosis. 5 Studies have previously indicated that certain cancers of the gastrointestinal tract, including colorectal, gastric, pancreatic, and hepatic cancers, were associated with an increased incidence of VTE. However, gallbladder cancers were not included in this analysis. 6
Mucinous adenocarcinoma, a rare form of cancer characterized by more than 50% extracellular mucin produced by malignant mucin-producing epithelial cells,7,8 represents ~1% of all cancers and can originate in nearly any organ, most commonly affecting the colon and breast. 7
We present a rare case of recurrent DVTs that continued despite optimal anticoagulation, mechanical thrombectomy, and the placement of an inferior vena cava (IVC) filter. This persistent condition was ultimately found to be caused by mucinous adenocarcinoma of the gallbladder. This case highlights the importance of thorough cancer screening in patients experiencing unexplained thrombotic events and provides valuable insights into the connection between hypercoagulability and hidden malignancies.
Case Presentation
A 50-year-old male with a history of hypertension, dyslipidemia, gastroesophageal reflux disease, neuropathy, past upper gastrointestinal bleeding, and a previous diagnosis of deep venous thrombosis (DVT) treated with apixaban presented with left lower extremity pain, swelling, and minimal dyspnea. Despite being on therapeutic doses of apixaban (5 mg twice daily), his DVT recurred a few months after the initial diagnosis, prompting further investigation. He was vitally stable. Laboratory studies supported this concern (Table 1).
Laboratory Results of the Patient Compared to Normal Reference Ranges.
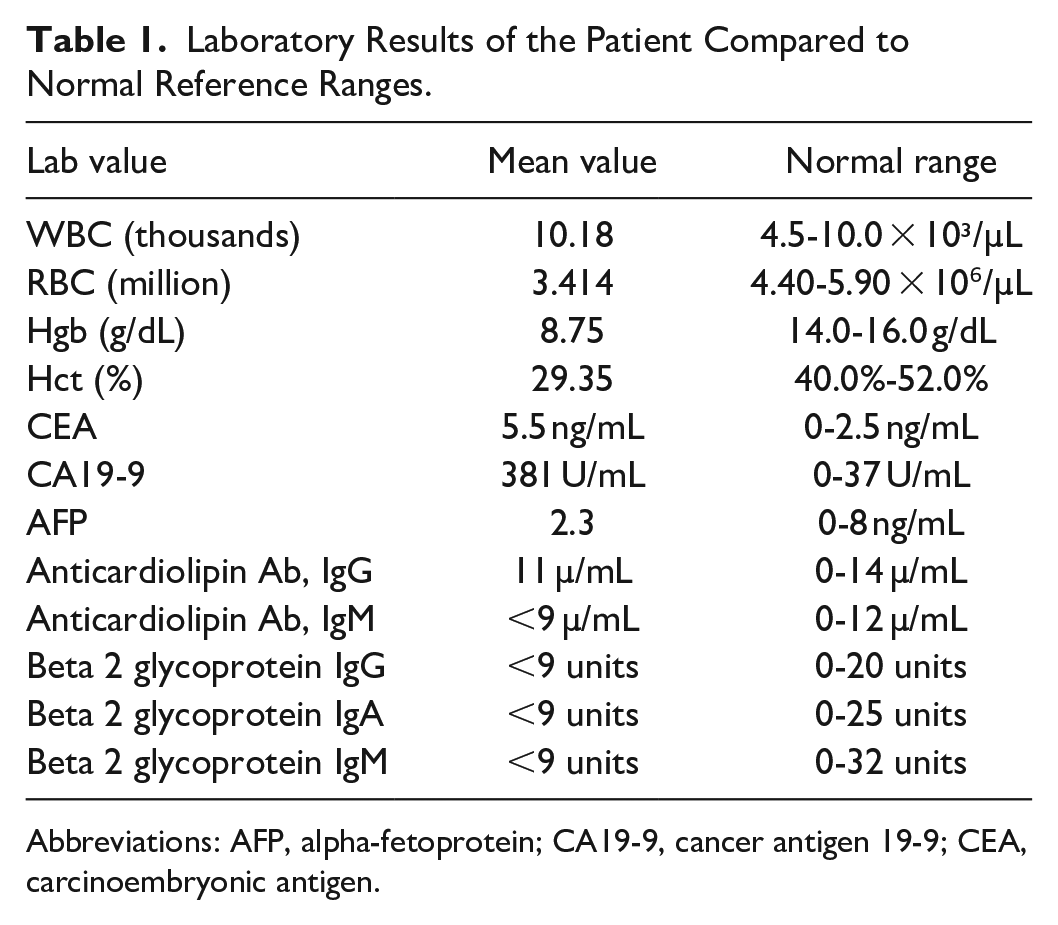
Abbreviations: AFP, alpha-fetoprotein; CA19-9, cancer antigen 19-9; CEA, carcinoembryonic antigen.
Lower extremity venous duplex confirmed extensive DVT involving the left femoral, popliteal, and foreleg veins. A CT chest angiogram of the chest showed no pulmonary embolism yet revealed extensive mediastinal and hilar lymphadenopathy (Figure 1), suggesting possible malignancy such as lymphoma. Subsequent imaging of the chest, abdomen, and pelvis demonstrated widespread adenopathy, hepatosplenomegaly, a nonspecific left adrenal nodule, and lytic lesions in the left ilium. These imaging findings and a 45-pound unintentional weight loss over 6 months strongly indicated an underlying systemic process, likely malignant.

Axial CT chest image, mediastinal window demonstrates significant right paratracheal lymphadenopathy (arrow), measuring ~28 mm.
Despite being anticoagulated with a heparin drip, his DVT worsened, and a subsequent lower extremity venous duplex revealed extensive thrombosis burden extending from the common femoral to the popliteal veins. He underwent a mechanical thrombectomy and the placement of an IVC filter. However, the following day he developed recurrent DVT and escalating left leg pain. Pain management was intensified with opioid medications, including a morphine infusion.
In light of this complexity, catheter-directed thrombolysis with tissue plasminogen activator was initiated, followed by a second mechanical thrombectomy and IVC filter removal. Postprocedurally, he was transitioned from heparin to an argatroban infusion before eventually restarting apixaban. The heparin-induced thrombocytopenia panel, including serotonin release assay, was negative.
Given the sizable mediastinal lymphadenopathy, fever, weight loss, and recurrent DVT, there was high suspicion of malignancy. Lymphoma remains a strong possibility. The patient underwent an endobronchial ultrasound (EBUS), and cytology and flow cytometry of the mediastinal lymph nodes were obtained through EBUS and transbronchial needle aspiration for further evaluation. Histopathological examination (Figure 2) revealed poorly differentiated adenocarcinoma with mucinous features in the station right 4 and station 7 lymph nodes. Immunohistochemistry results showed that CK7 was strongly and diffusely positive, while PSA, CDX2, CK20, P63, and TTF-1 were all negative. Additional stains showed negative TTF-1, while the mucicarmine stain was positive.

(A–D) Pathology slides show a right, station 4 transbronchial aspirate demonstrating poorly differentiated adenocarcinoma with mucinous features. (A) Shows the H&E stained cell block at 200× and (B) at 400× magnification. (C) Highlights the mucicarmine stain (+) at 400×, while (D) presents CK-7 stain (+) at 400×. Immunohistochemistry revealed negative staining for PSA, CDX2, CK20, P63, and TTF-1. The cancer TYPE ID reference test indicated a 90% probability of pancreaticobiliary origin.
Immunohistochemistry for PD-L1 revealed a tumor proportion score of 1%, while the tumor mutational burden was 16 mutations/megabase, which could make the patient a potential candidate for immunotherapy. However, no actionable mutations such as KRAS, EGFR, or ALK were identified. Microsatellite instability testing revealed that the tumor was microsatellite stable. Furthermore, molecular analysis of the transbronchial aspirates revealed a 90% probability of pancreaticobiliary malignancy. Based on these findings, he was ultimately diagnosed with stage IVB gallbladder cancer and started on Platinol/Gemzar/Imfinzi for 8 cycles for 10 cycles of durvalumab (Imfinzi). According to the patient’s preferences, he was maintained on Apixaban for recurrent DVTs with no events to date.
Discussion
This case involves a 50-year-old male with recurrent DVT, which persisted despite appropriate anticoagulation and prompted investigation into a possible underlying systemic issue specifically possible malignancy. In this case, the DVT recurrence was ultimately traced to a rare malignancy: mucinous adenocarcinoma of the gallbladder.
Cancer-associated thrombosis, often referred to as Trousseau’s syndrome, involves multiple mechanisms, including the upregulation of tissue factor by malignant cells, which promote coagulation and clot formation. Additionally, tumor cells release pro-inflammatory cytokines that activate the coagulation cascade. 9 One understood mechanism involves the upregulation of tissue factor by malignant cells. Tissue factor is a membrane protein generally involved in initiating coagulation when exposed to blood after vascular injury by binding factor VIIa.3,10,11 Tumor cells, however, may display tissue factor on their surface, promoting angiogenesis, local tissue invasion, and the hypercoagulable state seen in cancer. 12 Additionally, tumor cells upregulate several pro-inflammatory cytokines, including tumor necrosis factor, IL-1, and IL-6, which activate the coagulation cascade. Expression of anti-fibrinolytic factors also promotes hypercoagulability and clot formation.9,11,13 Additionally, mucinous adenocarcinomas have been shown to lead to nonenzymatic activation of factor X to factor Xa. 14 The case presented here exemplifies how recurrent thrombosis can be the result of this multifactorial process in rare cancers. Further research is needed to identify additional mechanisms underlying hypercoagulability in such cases.
Cancer-associated DVT in patients with exhibits distinct clinical characteristics. Patients with cancer have a significantly higher incidence of DVT and an increased risk of recurrence compared to those without malignancy.15,16 Additionally, cancer-related DVT is often more extensive, involving bilateral lower-limb thrombosis, iliocaval thrombosis, and upper-limb thrombosis, making it more severe than DVT in noncancer patients. 16 The patient’s presentation with extensive recurrent DVT despite anticoagulation aligns with these findings, emphasizing the need for vigilant monitoring and escalation of therapy when cancer-associated DVT is suspected. For further clarification, the Multicenter Advanced Study for a Thromboembolism Registry (MASTER) is a multicenter registry that prospectively enrolled consecutive patients with symptomatic, objectively confirmed acute VTE. In the MASTER registry, 424 patients (20%) had cancer. The study found that cancer patients had a significantly higher incidence of extensive thrombosis compared to noncancer patients. Specifically, the prevalence of bilateral lower-limb DVT was higher in cancer patients (8.5% versus 4.6%; P < .01), as were the rates of iliocaval thrombosis (22.6% versus 14%; P < .001) and upper-limb DVT (9.9% versus 4.8%; P < .001). These findings reinforce the association between malignancy and increased thrombotic burden, emphasizing the importance of early detection, risk stratification, and appropriate anticoagulation strategies in managing VTE in cancer patients. 16 Another distinguishing feature of cancer-associated DVT is the increased occurrence of thrombosis in unusual locations, such as the splanchnic veins and cerebral veins. 15
Gallbladder and other biliary tract malignancies constitute a rare subset of gastrointestinal malignancies and have been increasingly recognized as conditions associated with hypercoagulability. For instance, a retrospective study review of 289 patients with biliary tract cancers revealed that 22% of patients had VTE, suggesting an increased risk of thromboses. However, there was not a statistically significant difference in overall survival in individuals who presented with a VTE. 17 Another retrospective study of 273 cholangiocarcinoma patients identified 10 who presented with VTE at diagnosis, and in this cohort, VTE was linked to poorer prognostic outcomes. 18 These findings suggest that while hypercoagulability is commonly associated with gallbladder and other biliary system cancers, further research is essential to characterize these risks and guide management strategies better.
Common types of gallbladder cancer include nonvariant adenocarcinoma and cholangiocarcinoma. 7 Increased age, female gender, chronic gallbladder inflammation, polyps, and biliary cysts are risk factors for the development of gallbladder and other biliary malignancies. 19 The presentation of gallbladder malignancy is variable, with most patients presenting with generalized right upper quadrant abdominal pain. Additionally, patients may be jaundiced, have weight loss, elevated liver function tests, and nausea. Imaging studies routinely obtained include an ultrasound of the abdomen, as well as a computed tomography(CT) scan for staging. 20 While surgical resection of the disease before metastasis can be curative, these malignancies often present late and with systemic spread and require additional chemotherapy and radiation therapy. 19
Mucinous adenocarcinoma of the gallbladder is an uncommon form of biliary malignancy, representing about 3% of newly diagnosed cases. While more research is needed to characterize these tumors further, current literature suggests they do not favor 1 gender, tend to present at a younger age, and are often diagnosed at more advanced stages than nonmucinous gallbladder cancers. Despite these factors, survival rates at 1, 2, and 5 years appear comparable to those of more frequently encountered gallbladder malignancies. 7
The American Society of Hematology recommends increasing the dose of low-molecular-weight heparin (LMWH) to supratherapeutic levels or continuing with a therapeutic dose if the patient is already receiving LMWH. This recommendation is based on observations that dose escalation can effectively manage recurrent cancer-associated thrombosis, although it should be approached with caution in patients at high risk of bleeding. 21
Similarly, the American Society of Clinical Oncology guidelines suggest using LMWH, edoxaban, rivaroxaban, or apixaban for long-term anticoagulation in cancer patients, with a preference for LMWH due to its effectiveness in reducing recurrent VTE. 22 However, caution is needed when prescribing direct factor Xa inhibitors for patients with gastrointestinal or genitourinary malignancies due to the heightened risk of mucosal bleeding. 22
In instances where recurrent VTE occurs despite therapeutic anticoagulation, it is recommended to either switch from a direct oral anticoagulant to LMWH or increase the LMWH dose.23,24 If these interventions are ineffective or contraindicated, inserting an IVC filter may be considered a last resort.23,24
Conclusion
This case illustrates how recurrent, unexplained VTE can be a sign of an underlying malignancy. In patients presenting with recurrent, unprovoked DVT and lymphadenopathy, it is essential to consider not only common cancers but also rarer diagnoses, such as biliary cancers. This case highlights the importance of early recognition of cancer-related thrombosis and the need for comprehensive diagnostic evaluation, especially when DVT is recurrent or refractory to standard anticoagulation therapies.
Footnotes
Acknowledgements
We would like to express our sincere appreciation to the Research Committee of the Internal Medicine Department at Marshall University, WV, for their invaluable support and guidance during the development of this manuscript. Their assistance has been instrumental in our work. We also want to thank the pathology department at the school of medicine for their contributions; Saroj Sigdel, MD; Logan Lawrence, PA; and Krista L. Denning, MD.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
Prior Submissions/Publications
This material has not been published in any meeting or journal.
