Abstract
Coccidioidomycosis, endemic in the southwestern United States, can lead to severe pulmonary complications, particularly in chronic obstructive pulmonary disease (COPD) patients with poor lung reserves. Mycobacterium abscessus has a predisposition for structurally damaged lungs, commonly causing difficult-to-treat bronchiectasis and cavitary lesions. We present the case of a 58-year-old patient with advanced COPD and a remote history of pulmonary coccidioidomycosis diagnosed 30 years earlier, who was found to have sputum and radiographic evidence of M. abscessus cavitary lung disease 4 years prior to admission, but was lost to follow-up. The current presentation is attributed to the progression of untreated M. abscessus infection and reactivation of latent Coccidioides infection. Despite the initiation of antifungal and antibiotic therapy, the subsequent course was complicated by the development of bronchopleural fistula and worsening respiratory failure, leading to an unfavorable outcome. This case highlights the diagnostic challenges associated with overlapping clinical and radiologic features of concurrent infections and devastating outcomes in patients with COPD. Prompt diagnostic testing and prolonged comprehensive therapy are of paramount importance in managing such complex infections.
Introduction
Coccidioidomycosis or Valley fever is a fungal infection caused by the inhalation of spores from the dimorphic fungus Coccidioides. It is endemic in the southwestern United States, including El Paso, with a reported prevalence of 3.2 cases per 10,000 hospital discharges. 1 Diabetes and human immunodeficiency virus (HIV) infection have been reported as the most common risk factors predisposing to this fungal infection. 2 However, chronic obstructive pulmonary disease (COPD) was an independent risk factor for coccidioidomycosis-related hospitalization (adjusted odds ratio 1.59). 3 The spectrum of this infection can range from mild flu-like illness to severe empyema or fibro-cavitary lung lesions. 2 The progression of mild coccidioidomycosis to a more severe or chronic form is attributed to compromised pulmonary function and altered immune responses associated with COPD. 4 Smoking can further exacerbate the risk through impaired mucociliary clearance and immune dysregulation. 5
Mycobacterium abscessus, a rapidly growing nontuberculous mycobacterium (NTM), is an infrequently encountered lung pathogen with geographical variability known to cause pulmonary infections in patients with underlying structural lung diseases such as COPD.6-10 This organism causes a decrease in the oxidative metabolism of lung airway cells and relaxation of epithelial mucosal tight junctions in COPD, contributing to pathogenesis and severity. 11 A diverse range of pulmonary manifestations, ranging from interstitial, alveolar, and reticulonodular patterns to severe bronchiectasis and cavitary lesions, has been reported. 12 It is known to cause biofilms, leading to difficult-to-treat infections. 10 Diagnosis is challenging due to the overlap in clinical and radiological presentations of the 2 entities. Prolonged therapy is necessary for complete eradication to ensure improved survival outcomes, making microbiological and serological testing of paramount importance in informing treatment decisions. We report a case of pulmonary coinfection with Coccidioides and M. abscessus in a patient with COPD, highlighting the challenges in diagnosis and management.
Case Presentation
A 58-year-old male presented with productive cough, exertional dyspnea (New York Heart Association class III), small-volume hemoptysis, significant weight loss, and fatigue for 4 weeks. He denied fever, chills, palpitations, syncope, orthopnea, paroxysmal nocturnal dyspnea, joint pain, rash, diarrhea, hematuria, or bleeding. Past medical history was notable for COPD and pulmonary coccidioidomycosis requiring right upper lobe resection after failure of medical management 30 years before the current admission. Postoperatively, he experienced significant clinical improvement with undetectable titers on follow-up. Four years before this presentation, he developed recurrent symptoms of cough with expectoration, low-grade fevers, and weight loss. Computed tomography (CT) of the chest revealed bronchiectasis in the right middle and lower lobes and centrilobular, peribronchial nodules with a tree-in-bud pattern in the left upper and middle lobes, indicative of an active infective etiology (Figure 1). Two separate sets of sputum culture grew M. abscessus susceptible to macrolides. Coccidioidomycosis serology was undetectable; workup for infections, autoimmune diseases, and malignancy was negative. Infectious disease was consulted and treatment with azithromycin, amikacin, and omadacycline was initiated. However, the patient did not complete therapy and was subsequently lost to follow-up. He had smoked 1 to 2 packs of cigarettes daily for at least 40 years before quitting 3 years ago. There was no history of recent travel or exposure to sick individuals or animals, alcohol usage, or intravenous substance abuse.

High-resolution computed tomography of the thorax (coronal sections) 4 years prior to current admission showing resected upper lobe of the right lung (asterisk) with linear bronchiectasis and bilateral tree-in-bud nodular airspace opacities.
On examination, the patient appeared cachectic (body mass index 16 kg/m2) with a heart rate of 90/min, respiratory rate of 24/min, blood pressure of 110/70 mmHg, and oxygen saturation of 92% on 2 L/min supplemental oxygen. Auscultation revealed severely diminished breath sounds over the right lung and crepitations in the left upper lung. The remainder of the systemic examination was unremarkable. Laboratory evaluation showed normocytic normochromic anemia (9.4 g/dL; normal: 12-16 g/dL), normal leukocyte count (9.97 × 10³/µL; normal: 4.5-11 × 10³/µL), thrombocytosis (554 × 10³/µL; normal: 150-450 × 10³/µL), and elevated C-reactive protein (18 mg/dL; normal: <1 mg/dL). Renal, liver, and thyroid function tests were within normal limits. Chest radiograph revealed a large right-sided pneumothorax with moderate pleural effusion and a cavitary lesion in the left lung apex. Contrast-enhanced CT showed rupture of a thick-walled cavitary lesion in the right lower lobe into the pleural space, resulting in a large hydropneumothorax with bronchopleural fistula, diffuse pleural thickening, and collapse of the remaining right lung. A 4.7 cm thick-walled cavitary lesion with peripheral atelectasis and pleural thickening was identified in the left upper lobe, along with mediastinal lymphadenopathy (Figure 2). Two sets of sputum cultures grew M. abscessus; samples were sent to the central state health department for further processing of antibiotic susceptibility and speciation. Coccidioidomycosis serology was positive (1:4 titer by immunodiffusion [ID]), while urine histoplasma antigen, serum cryptococcal antigen, aspergillus antibodies, serum galactomannan, and sputum Mycobacterium tuberculosis PCR were negative. Blood cultures on prolonged incubation showed no growth. Sputum cultures were negative for bacterial (aerobic and anaerobic) and fungal pathogens. Workup for autoimmune diseases and HIV was negative.
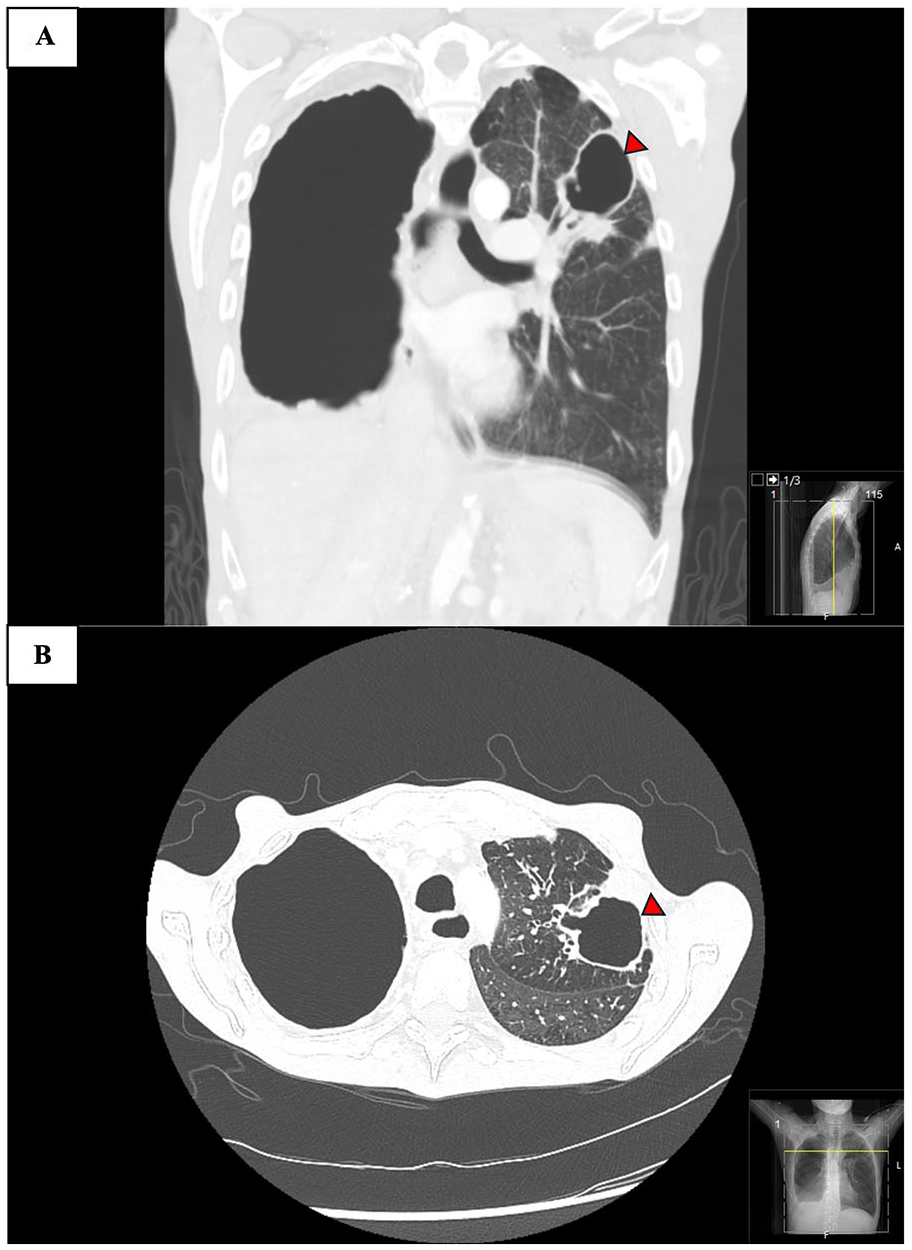
High-resolution computed tomography sections of the thorax (coronal [A] and axial [B] at the time of admission showed large pneumothorax with pleural effusion (hydropneumothorax) in right lung and presence of a cavitary lesion (arrowhead) with bronchiectasis in the left upper lobe.
An intercostal drainage tube was placed in the right lung for large hydropneumothorax and revealed constant bubbling of the air column and underwater seal container, suspicious of a bronchopleural connection. Pleural fluid analysis was exudative by Light’s criteria and did not show any specific growth. The patient was empirically started on piperacillin-tazobactam and vancomycin to cover for superadded community-acquired bacterial infections. Fluconazole was added in view of clinical, radiological, and serological evidence of active coccidioidomycosis. However, oxygen requirements continued to escalate, requiring high-flow nasal cannula support. Cardiothoracic surgery was consulted for the repair of a bronchopleural fistula, but the surgical intervention was deferred due to severe structural lung disease, poor pulmonary reserve, and profound malnutrition. On the fifth day of hospitalization, he developed rapidly deteriorating respiratory failure, requiring intubation, mechanical ventilation, and placement of a second chest tube in the right pleural space. Repeat CT imaging of the chest revealed worsening right-sided hydropneumothorax with multiple bronchopleural fistulas and an unchanged left upper lobe cavity without evidence of pulmonary thromboembolism (Figure 3). The family declined further intervention or transfer for specialized care and opted for comfort measures. The patient died the following day. Timeline of the case is summarized in Table 1.

Follow-up high-resolution computed tomography sections of the thorax on day 11 of hospitalization showed persistent cavitary lesion (arrowhead) in the left lung (A) and non-resolving large pneumothorax with pleural effusion (hydropneumothorax) with an intercostal chest tube in the right pleural cavity (B).
Timeline of Clinical Events.

Abbreviation: CT, computed tomography.
Discussion
Coccidioidomycosis is an endemic fungal infection in the southwestern United States. Although it is not a reportable disease in Texas, it is highly prevalent in the border city of El Paso. 13 In contrast, M. abscessus lacks geographic endemicity but is a frequently implicated opportunistic pathogen with a predilection to hosts with underlying structural lung diseases (eg, cystic fibrosis). 14 The mortality rate of pulmonary coccidioidomycosis in critically ill and mechanically ventilated patients remains high (33%-48%) in several multicenter observational studies.15,16 Similarly, the cumulative mortality of pulmonary NTM at 15 years after diagnosis is 36.4%, with etiological agent (M. abscessus) and cavitary bronchiectasis form strongly associated with higher odds of mortality. 17 While mortality is 1.43 times higher in COPD with NTM infection compared to those without NTM infection, there is limited data on morbidity and mortality of pulmonary coccidioidomycosis with underlying COPD.
Pulmonary coccidioidomycosis is often asymptomatic in 60% of cases. Commonly reported symptoms include fever (76%), cough (73%), chest pain (44%), dyspnea (32%), and fatigue.2,18 In contrast, hemoptysis is a common presenting complaint (~31%) in pulmonary infections with M. abscessus, with other nonspecific symptoms such as cough (86%) and low-grade fever (18%). 19 Our patient presented with overlapping symptoms of shortness of breath, productive cough, hemoptysis, weight loss, and fatigue. Chest imaging in chronic pulmonary coccidioidomycosis often reveals solitary nodules in the upper lobe of the lung, masses, thin-walled cavities, consolidation, bronchiectasis, and mediastinal lymphadenopathy. Complications such as bronchopleural fistulas, pneumothorax, and empyema occur in 2.7% of those with cavitary lesions. 20 The predominant radiological pattern of M. abscessus includes bilateral reticulonodular opacities (88%), multilobar bronchiectasis (90%), upper lobe cavitation and consolidation (16%), and tree-in-bud pattern of parenchymal airspace opacities. 21 Imaging of our patient during active M. abscessus infection 3 years prior revealed bronchiectasis in the right middle and lower lobes, along with centrilobular and peribronchial nodules in the left upper and middle lobes. Repeat imaging at current admission showed the development of a large hydropneumothorax with a bronchopleural fistula as a result of a rupture of the cavity in the right lower lobe. In addition, a new thick-walled cavity in the left upper lobe with mediastinal lymphadenopathy was noted. These findings suggest overlapping radiological features of both infections.
First-line diagnostic testing of pulmonary coccidioidomycosis involves serological testing in a background of clinical and radiological imaging. The initial serological test is enzyme immunoassay followed by ID for confirmation. 22 Fungal culture of bronchoalveolar lavage (BAL) is more sensitive than sputum but is limited by prolonged duration of incubation for isolation. 22 The gold standard for confirmation is a demonstration of spherules on histopathology specimens of the cavity, but the biopsy was not feasible in our patient due to hemodynamic instability and rapid deterioration. Diagnosis of M. abscessus can be confirmed with positive cultures from 2 sputum samples or 1 BAL. Alternatively, a transbronchial or other lung biopsy showing acid-fast bacilli or granulomatous inflammation, along with positive sputum or BAL culture, is also diagnostic. 23 Our patient had 2 sputum cultures positive for M. abscessus. Serology showed elevated titers for coccidioidomycosis, previously undetectable, suggesting coinfection with both pathogens. Clinical features and radiology supported the diagnosis of coinfection. The timeline suggests that overall poor patient’s immunological status may have contributed to the reactivation of coccidioidomycosis. Previously acquired quiescent and clinically inactive Coccidioides infections have been reported to reactivate in immunocompromised hosts.24,25 Further studies are needed to enhance our understanding of the pathogenesis and disease progression in the rare setting of coinfection in a host with structurally compromised diseased lung.
Both infections require prolonged antimicrobial therapy. For coccidioidomycosis, mild-to-moderate disease is treated with triazoles (fluconazole or itraconazole), while severe disease requires initial treatment with Amphotericin B, followed by oral azoles for 3 to 6 months based on clinical and radiological response. 22 M. abscessus is difficult to treat due to multidrug resistance. Therapy is guided by extended antibiotic susceptibility, the turnaround time of which may be delayed by weeks. In macrolide-susceptible strains, the initial regimen includes azithromycin with an intravenous agent such as amikacin, imipenem, or cefoxitin, and one of omadacycline, linezolid, or clofazimine. This is followed by oral or inhaled maintenance therapy for 1 year until cultures are negative.23,26 Our patient had a history of incomplete treatment for NTM infection, which increased the probability of harboring a resistant, difficult-to-treat strain. Treatment for multidrug-resistant strains is complex and requires robust multidisciplinary support with the aid of public health agencies. Managing drug interactions and ensuring patient compliance can be challenging, particularly in border regions such as El Paso.
We report a rare case of reactivation of pulmonary Coccidiosis in a COPD patient with incompletely treated M. abscessus lung infection. These infections present overlapping clinical and radiological features, which complicate their management. Coinfection potentially amplifies the risk of mortality and poor outcomes, especially in the setting of chronic structural lung disease with compromised pulmonary reserve. In addition, the requirement for prolonged treatment courses further underscores the challenges associated with addressing the coinfection. The presence of 1 diagnosis does not exclude the possibility of others, particularly in patients with underlying structural lung disease. Serial imaging to monitor response to therapy plays a critical role. In cases where clinical or radiological improvement is lacking, further microbiological workup and targeted biopsies should be considered to evaluate coexisting differential diagnoses.
Conclusion
Coinfections with Coccidioides and M. abscessus are rare but can occur in patients with structural lung diseases such as COPD. These patients have an amplified risk of complications and mortality. M. abscessus infections may alter host immunity, potentially triggering the reactivation of quiescent infections such as pulmonary coccidioidomycosis. Due to overlapping presentations, comprehensive microbiological testing, including sputum culture, serological testing, and imaging, is essential for accurate diagnosis and treatment. Prolonged antimicrobial therapy, drug interactions, and poor patient compliance complicate management. Lack of response to therapy should prompt further microbiological workup and targeted biopsies to evaluate other differential diagnoses.
Footnotes
Author Contributions
L.K. and A.Z.: Case report writing and editing. S.R.D. and K.S.: Literature review and case report manuscript editing. A.D.: Supervising and final editing of manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institutions do not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for the anonymized information to be published in this article.
