Abstract
Patients with a history of drug abuse and right-sided endocarditis are particularly susceptible to developing septic pulmonary emboli. Rarely, septic pulmonary emboli can cause severe lung injury resulting in parenchymal loss despite appropriate antibiotic therapy. We present 2 cases of severe lung injury associated with septic pulmonary emboli stemming from right-sided infective endocarditis, emphasizing 2 rare complications: bilateral spontaneous pneumothorax and pneumatocele formation.
Introduction
Even with the appropriate antibiotic therapy, the prognosis for infective endocarditis (IE) remains poor, with a global mortality rate as high as 30%. 1 The incidence of IE is around 3 to 10 per 100 000 people with 25% of the cases exhibiting symptoms of embolic events at the time of diagnosis. 2 No evidence currently supports the effectiveness of prolonged antimicrobial treatment in reducing the incidence of embolic events. 2 Septic pulmonary emboli (SPE) represent a significant yet often underrecognized infectious complication of right-sided IE, with a notably high prevalence among intravenous drug users (IVDU). 3 SPE is associated with empyema and pulmonary cavitations; however, pneumothorax (PTX) and pneumatoceles are rare complications.
This report describes 2 patients in whom IE resulted in severe lung injury, including PTX, diffuse parenchymal damage, and pneumatocele formation, which significantly complicated their hospital course.
Case Presentations
Our first patient is a 27-year-old female with a history of IVDU who was admitted to the intensive care unit for acute hypoxemic respiratory failure in the setting of tricuspid valve endocarditis. Multiple cavitary lesions throughout both lung fields were noted in the initial chest X-ray (CXR; Figure 1). Chest CT (computed tomography) angiography revealed bilateral subsegmental pulmonary emboli, extensive cavitary lesions consistent with SPE, and bilateral pleural effusions, larger on the right side (Figure 2). A right-sided pigtail catheter was inserted, draining 900 mL of empyema. Blood, sputum, and pleural fluid cultures grew methicillin-resistant Staphylococcus aureus (MRSA). Transesophageal echocardiography demonstrated large, multilobulated, mobile vegetation on the tricuspid valve (measuring 3.1 cm × 2.6 cm), right heart strain, and a patent foramen ovale with a right-to-left shunt. The patient was deemed unsuitable for surgery due to hemodynamic instability. Her respiratory status improved with antibiotic therapy. However, a repeat CT thorax without contrast on hospital day 13 revealed increased coalescence of bilateral cavities, resulting in extensive bilateral-infected pneumatoceles with air-fluid levels, more prominent on the right side (Figure 3). Continuation of antibiotic therapy with close clinical monitoring was deemed the most appropriate management strategy. On the 27th day of hospitalization, a repeat blood culture came back negative for MRSA. After completion of the 6-week course of antibiotics, follow-up CT thorax without contrast was done and showed persistent posterior left-sided pneumatoceles with air-fluid levels; however, there was a significant decrease in the size of posterior right-sided pneumatocele along with less-prominent SPE (Figure 4).

Multiple cavitary lesions involving bilateral lungs (red arrows) along with nodular opacity in the left lower lung (blue arrow).

Innumerable bilateral cavitating pulmonary lesions with thick irregular walls measuring up to 3.5 cm in diameter (red arrows). Diffuse pulmonary ground glass opacities. Large right pleural effusion (blue arrow). Small left pleural effusion (green arrow). No signs of pneumothorax.

Widespread cavitary lesions throughout the lungs consistent with septic emboli (red arrow). Coalescence of bilateral cavities, resulting in extensive bilateral infected pneumatoceles with air-fluid levels (blue arrows).

Large bilateral posterior pneumatoceles filled with air and soft tissue material (red arrows). The right lesion is slightly smaller and contains less air than prior study. The left-sided lesion contains a larger amount of air and is similar in size. Sequalae septic emboli, less prominent and numerous compared to the previous study.
Our second patient is a 44-year-old female with a medical history of polysubstance abuse and traumatic splenic laceration, initially admitted for acute hypoxic respiratory failure requiring endotracheal intubation. Her initial CT angio of the chest showed extensive cavitation subpleural consolidations surrounded by ground glass opacities suggestive of SPE (Figure 5). Transthoracic echocardiography confirmed the presence of vegetation on the tricuspid valve and blood cultures grew MRSA. Her hospitalization was prolonged due to complications of MRSA bacteremia, including bilateral PTX secondary to ruptured cavitary lesions, necessitating bilateral chest tube placement (Figures 6-8). While the left-sided chest tube dislodged, postdislodgement imaging showed no evidence of PTX reaccumulation, and the tube was not reinserted. Persistent air leakage on the right side raised suspicion for a bronchopleural fistula. Consequently, the patient was transferred to another facility, where she underwent bronchoscopy with placement of 6 endobronchial valves in the right upper lobe. However, the endobronchial valves were not well tolerated, and she was transferred back to our hospital. On the 21st day of hospitalization, a repeat CT thorax without contrast revealed moderate right-sided hydropneumothorax with right subpulmonic effusion, left apical PTX as well as the development of enlarging intraparenchymal and subpleural cavitary lesions (Figure 9). On her 37th day of hospitalization, we repeated the CT thorax to monitor the progression of lung injury, which showed interval development of bilateral extensive pneumatoceles with significant parenchymal loss along with persistent stable bilateral PTX and evident parenchymal scarring (Figures 10 and 11). The patient was not deemed a surgical candidate due to her overall clinical status and malnutrition.

Extensive cavitating subpleural lesions with areas of peripheral consolidation surrounding ground glass most consistent with septic emboli (red arrows).

Large left-sided pneumothorax with irregular collapse of the left lung (blue arrows).

Left-sided chest tube (green arrow). A new pneumothorax at the right lung base (blue arrow). Slight decreased left pneumothorax (red arrows).
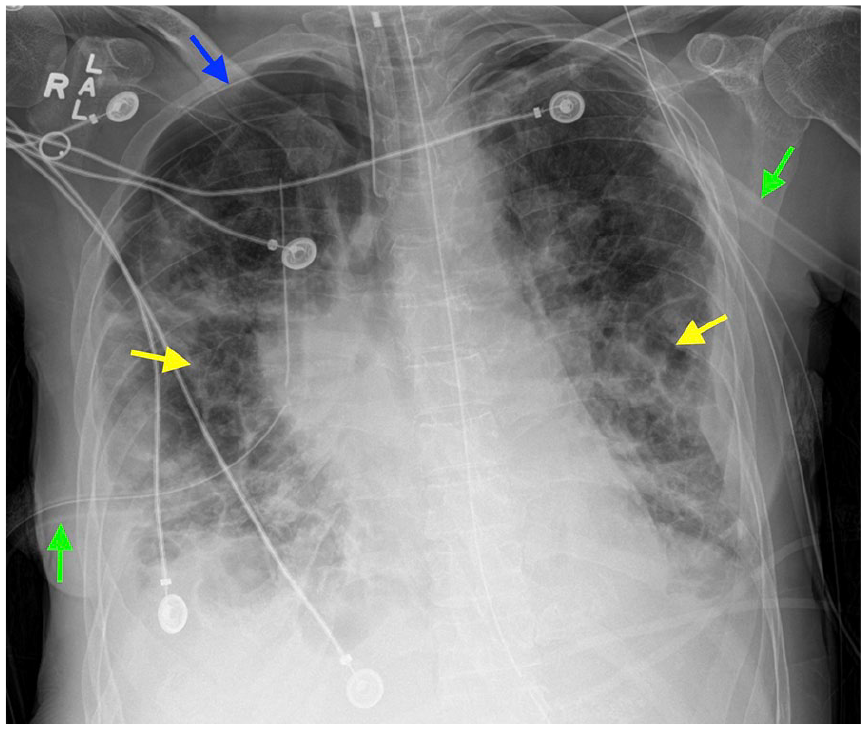
Bilateral surgical chest tubes (green arrows). Small right apical pneumothorax (blue arrow). Again noted are interstitial infiltrates involving the lungs (yellow arrows).

There is a moderate right hydropneumothorax, possibly partially extrapleural (blue arrow). Right subpulmonic effusion. Widespread cavitary lesions are likely due to septic emboli (red arrows).

Interval development of bilateral extensive pneumatoceles/intraparenchymal bulla (red arrows). Stable left-sided apical pneumothorax. Stable right-sided hydropneumothorax. Bilateral parenchymal scarring is again evident.

Interval development of bilateral extensive pneumatoceles/intraparenchymal bulla (red arrows). Stable left-sided apical pneumothorax. Stable right-sided hydropneumothorax. Bilateral parenchymal scarring is again evident.
Discussion
The lungs are susceptible to injury from illicit drug use. IVDU has been associated with a wide spectrum of acute and chronic pulmonary complications, including cardiogenic and noncardiogenic pulmonary edema and SPE, all driven by both the toxic effects of the drugs and the harmful properties of filler materials. 4 SPE is an uncommon but well-defined complication of IE mostly involving the tricuspid valve, particularly prevalent among IVDU due to the high incidence of right-sided IE. 5 CXR findings of SPE are usually nonspecific lung consolidations mimicking bronchopneumonia, but may indicate ill-marginated peripherally located lung nodules. 6 The most common CT findings suggest an extrapulmonary infectious focus and raise suspicion for septic emboli are multiple peripherally located nodular opacities with or without accompanying pulmonary cavitations.6,7
Septic emboli originating from IE frequently lead to lung abscesses and empyema. Additionally, IVDU is a risk factor for the development of PTX, mostly secondary to direct pleural trauma caused by self-injection into the subclavian and jugular veins. 8 Rarely, the rupture of subpleural cavities secondary to SPE can cause spontaneous PTX as in our first patient who developed bilateral PTX requiring bilateral intercostal chest tube placement. Likely, the use of positive pressure ventilation also contributed to her increased risk of PTX.
To the best of our knowledge and based on the literature review, since the first case of spontaneous PTX in the setting of SPE was described by Aguado et al 8 in 1990, only 17 cases were reported in the literature, which we summarized in Tables 1 and 2.
A Summary of 17 Documented Cases of PTX as a Complication of SPE Stemming From IE.
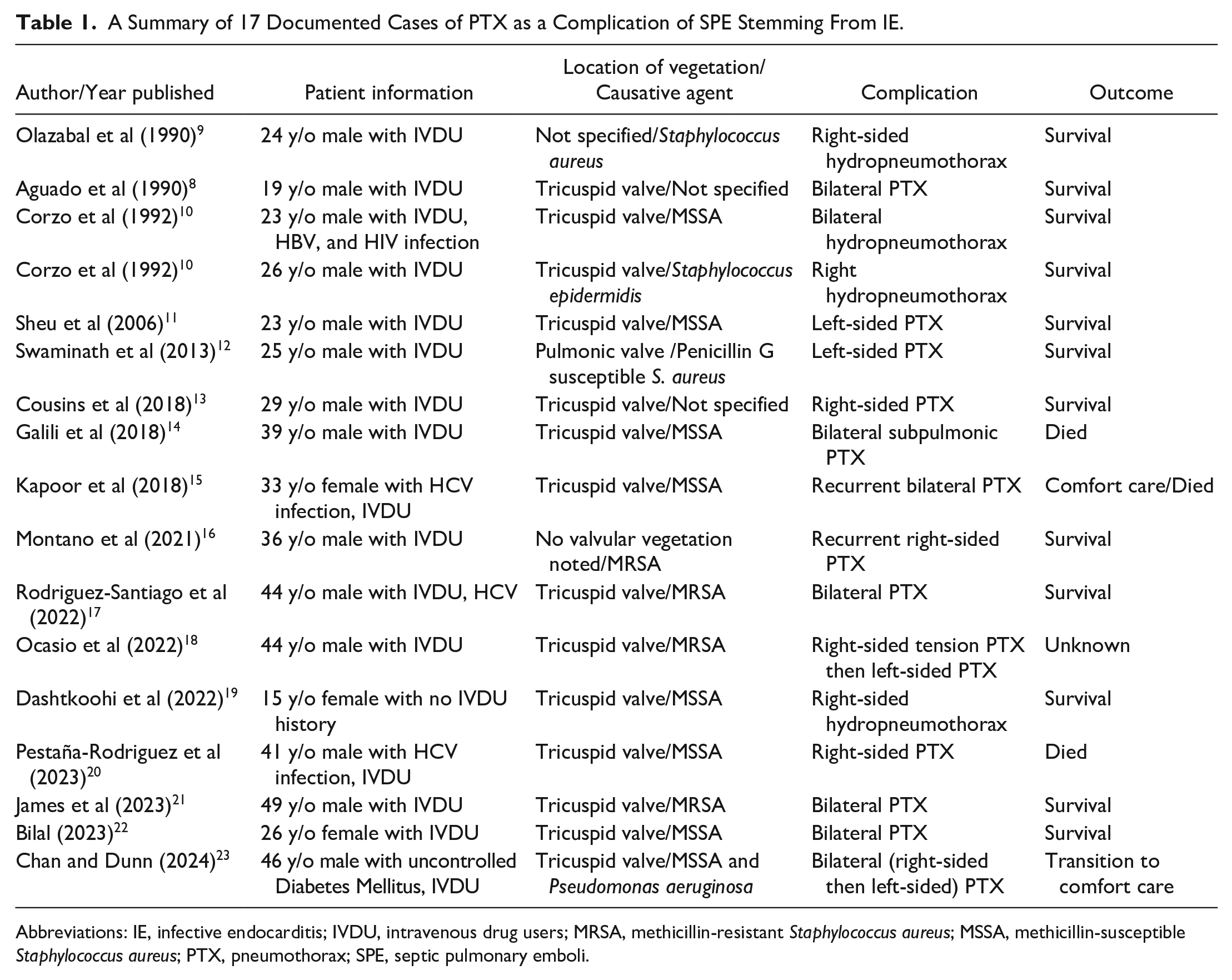
Abbreviations: IE, infective endocarditis; IVDU, intravenous drug users; MRSA, methicillin-resistant Staphylococcus aureus; MSSA, methicillin-susceptible Staphylococcus aureus; PTX, pneumothorax; SPE, septic pulmonary emboli.
A Summary of 3 Reported Cases Describing Cystic Parenchymal Changes (Pneumatoceles/Multiloculated PTX) of the Lung in the Setting of SPE Stemming From IE.

Abbreviations: CXR, chest X-ray; IE, infective endocarditis; IVDU, intravenous drug users; MRSA, methicillin-resistant Staphylococcus aureus; MSSA, methicillin-susceptible Staphylococcus aureus; PTX, pneumothorax; SPE, septic pulmonary emboli.
The majority of patients in the reviewed literature underwent chest tube placement, with an overall favorable outcome. Notably, males seem to be more frequently affected by this rare complication, accounting for 82.35% of the cases (14 out of 17 patients). Furthermore, a remarkable 94.12% of the reported cases had a history of IVDU, stressing this population as being potentially more vulnerable to developing severe lung injury in the setting of SPE.
Pneumatoceles or severe cystic changes are also rare lung injuries associated with SPE that were scarcely reported in the previous literature. We were able to find only 3 cases describing destructive parenchymal changes and the development of large pneumatoceles or multiloculated pneumothoraces in the setting of SPE secondary to IE; 2 of which were severe to the extent of radiologically mimicking vanishing lung syndrome (bullous emphysema) as we encountered in both cases we reported.24-26
CT is the gold standard for assessing pneumatoceles, providing significantly higher sensitivity—approximately 96%—compared to conventional radiography. 27 Air-filled pulmonary lesions exhibit similar characteristics on imaging, and their terminology is occasionally used interchangeably. Pulmonary cavities, cysts, and bullae are the primary differential diagnoses for pneumatocele, requiring careful consideration of clinical presentation, underlying etiologies, and radiographic features of both the lesions and surrounding lung parenchyma. Pulmonary cysts present as round lesions with well-defined, thin walls (<2 mm) surrounded by normal parenchyma, whereas bullae, another type of thin-walled cystic lesion, are typically larger (>1 cm) and often associated with centrilobular and paraseptal emphysema. 27 Cavities are common lung lesions, typically resulting from lung infections. On CT scans, cavities appear as air-filled lesions with thick, irregular walls, typically located within consolidations, masses, or nodules. 27 Pulmonary pneumatoceles are thin-walled and gas-filled sacs within the lung parenchyma that are also considered pseudocysts due to walls formed by lung parenchyma and absent epithelial lining.28,29 A key distinguishing feature of pneumatoceles, setting them apart from other cystic lesions, is their transient nature, typically developing after pneumonia or trauma, along with their tendency to rapidly change in size and appearance. 30 They may be associated with adjacent consolidations or ground-glass opacity due to recent pneumonia and often show an increase in size over time on repeated imaging.27,31 Pneumatoceles formed due to trauma, chronic infections, or prolonged inflammation may display varying levels of fibrosis around the cystic space, marked by dense collagen deposition or fibrous tissue encircling the lesion. 31 There are multiple proposed mechanisms explaining the development of pulmonary pneumatoceles including focal pulmonary overinflation secondary to inflammatory exudate in the airway lumen causing bronchial or bronchiolar obstruction of the check-valve type, drainage of the necrotic lung parenchyma with subsequent enlargement of the sac due to check-valve bronchiolar obstruction, and collection of the air in the interstitial tissue, particularly in the pleura. 28 The most frequently reported mechanism is the formation of an endobronchial check-valve in the setting of bronchial obstruction that leads to air trapping and enlarging cystic spaces with prolonged and severe blockade. 32 Pneumatocele formation can be part of the lung’s reparative process, with fibrotic changes around the cystic space developing in traumatic cases as a healing response to pulmonary laceration and tissue disruption. 31
While postpneumonia pneumatocele formation is a well-established entity in the pediatric population, only a few cases have been reported in adult patients.30,33-35
Despite the strong connection with Staphylococcus aureus pneumonia, Streptococcus, Haemophilus influenza, Klebsiella, Escherichia coli, and viral pneumonia (particularly COVID-19 infections), positive pressure ventilation, and pulmonary infarcts were also shown to be associated with the formation of pneumatoceles, and patients experiencing recurrent pneumonia secondary to anatomic abnormalities or aspiration are at greater risk of developing pneumatoceles.29,36 Other preexisting medical conditions, including immunosuppressive states, chronic granulomatous disease, hyper-IgE syndrome, cystic fibrosis, desquamative interstitial pneumonia, granulomatosis with polyangiitis, Langerhans cell histiocytosis, metastatic neoplasms, lung cancer, and pulmonary emphysema, can also increase susceptibility to pneumatoceles. 31 We believe that in both of our patients, recurrent SPE stemming from the tricuspid valve and the presence of multiple cavitary lesions might have played a significant role in the development of severe pulmonary parenchymal cystic changes. Lung cavities arise from necrosis and liquefaction, a process distinct from the check-valve mechanism that leads to pneumatocele formation. 31 However, pulmonary cavitations may contribute to pneumatocele development by causing irritation and inflammation of small draining bronchioles, leading to airway obstruction, which, in turn, creates a check-valve effect and results in air trapping. 28 Moreover, inflammation and necrosis of an airway segment can create direct communication between the airway and the bronchovascular interstitium, allowing air to travel through the bronchovascular connective tissue and form a pneumatocele. 37
Pneumatoceles can lead to a variety of complications, either due to their nature or as a result of diagnostic and interventional errors. The most common complication is infection, which is more frequently observed in posttraumatic or postinfectious pneumatoceles and may progress to empyema or lung abscess. 31 Infected pneumatoceles are associated with higher mortality rates compared to lung abscesses and, therefore, require prompt antibiotic therapy and, when indicated, drainage. 38 Pneumatoceles pose a significant risk of rupture, which can lead to various life-threatening complications. The accumulation of trapped air exerts pressure on the lung and mediastinal structures, potentially resulting in severe respiratory distress and cardiovascular instability. Rupture-related complications include pneumomediastinum, pyopneumothorax, hemothorax, and hemopneumothorax, all of which may necessitate urgent medical intervention.31,39,40 In their review of 37 patients with post-Covid-19 pneumo‑hematoceles, Chavez et al suggested that lesions measuring 3 cm or more are associated with a heightened risk of rupture, with each additional centimeter in lesion diameter correlating with a 1.5-fold increase in rupture risk. 41 As pneumatoceles enlarge, they can cause complications by compressing adjacent lung tissue and bronchial structures. This compression may contribute to progressive hypoxemia, atelectasis, recurrent infections, cardiopulmonary instability, and airway obstruction, all of which can severely affect respiratory function and overall clinical status. 31 Cardiopulmonary collapse may occur due to the rapid enlargement of a pneumatocele, a condition known as tension pneumatocele. 42 Patients receiving mechanical ventilation are particularly susceptible to pneumatocele enlargement; therefore, careful monitoring of pneumatocele size and appropriate adjustments to ventilator settings are crucial to prevent complications and optimize patient outcomes. 31 Chronic pneumatoceles, particularly those resulting from recurrent infections or trauma, can lead to fibrosis and scarring of the lung tissue, which reduces lung compliance and capacity, if left untreated, progressing to restrictive lung disease. Although extremely rare, extrathoracic extension of a pneumatocele has been reported in a patient undergoing anti-tuberculosis treatment.43,44 Complications tend to occur more frequently in cases of larger or infected pneumatoceles, in older patients, in those with predisposing conditions such as ciliary dysfunction, immunosuppression, or structural lung abnormalities, as well as in individuals with severe lung injury (such as Acute Respiratory Distress Syndrome) or those requiring positive pressure ventilation. 31
Most pneumatoceles resolve spontaneously with treating underlying pneumonia. 32 There are no established guidelines for the management of pneumatoceles; therefore, individualized management strategies are warranted. In several cases, video-assisted thoracoscopic surgery, percutaneous drainage, lobectomy, pneumonectomy, and intrapleural fibrin sealant injection were suggested as successful treatment methods for pneumatoceles.30,41,45 However, none of these interventions is risk-free but is associated with complications such as PTX and bronchopleural fistula, as well as increased mortality; therefore, a holistic approach should be taken during clinical decision-making.
Conclusion
Overall, we suggest that patients with SPE in the setting of IE might be more susceptible to developing lung injury due to recurrent showering emboli; therefore, they should be meticulously monitored with frequent imaging for early detection of pneumatocele formation or parenchymal cystic changes and risk to avoid life-threatening complications.
Footnotes
Acknowledgements
We sincerely thank the patient for their consent to use clinical information and imaging, which made this work possible. Their willingness to contribute to medical research is greatly appreciated.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for his anonymized information to be published in this article.
