Abstract
Citrobacter koseri (formerly classified as Citrobacter diversus) is a gram-negative bacillus (GNB) that occurs as an opportunistic pathogen in neonates and immunocompromised patients. Citrobacter species have been implicated in nosocomial settings leading to infections involving the urinary tract, respiratory tract, liver, biliary tract, meninges, and even in rarer conditions—blood stream infection and infective endocarditis (IE). Gram-negative bacilli are responsible for 3% to 4% of all IE cases and have been traditionally associated with intravenous drug users. Patients with non-HACEK (species other than Haemophilus species, Actinobacillus actinomycetemcomitans, Cardiobacterium hominis, Eikenella corrodens, or Kinglella species) GNB IE have poor clinical outcomes with higher rates of in-hospital mortality and complications. The American Heart Association (AHA) and Infectious Diseases Society of America (IDSA) both recommend the use of combination antibiotic therapy with a beta-lactam (penicillins, cephalosporins, or carbapenems) and either an aminoglycoside or fluoroquinolones for 6 weeks (about 1 and a half months) to treat IE due to non-HACEK GNB. Citrobacter koseri is becoming more recognized due to its inherent resistance to ampicillin and emerging drug resistance to beta lactams and aminoglycosides requiring carbapenem therapy. Our case is of a 75-year-old male with no previously reported history of primary or secondary immunodeficiency disorders who developed C koseri blood stream infection. His infectious work-up revealed mitral valve IE and septic cerebral emboli resulting in ischemic infarcts. This case illustrates the importance of recognizing GNB organisms as rising human pathogens in IE cases even without active injection drug use or nosocomial exposure.
Introduction
Citrobacter species are motile and anaerobic gram-negative bacilli (GNBs) that belong to the Enterobacteriaceae family. There are 3 species that have been known to be pathogenic in humans—C amalonaticus, C koseri (previously named C diversus), and C freundii.1,2 These pathogens are commonly found in water, food, soil, and human and animal intestinal tracts. Risk factors for Citrobacter infections include diabetes mellitus, congestive heart failure, hepatobiliary disease, malignancy, or structural abnormalities of the urinary tract. In adults, infections are usually reported in immunocompromised patients, but 11% of patients exhibit no underlying disease.2,3 In 2007, Mohanty et al 3 analyzed 205 patients with Citrobacter infections in a prospective study and discovered 94.6% of patients acquired their infection nosocomially. Microbiology data showed cultures yielded C koseri in 90.2% (185) of cases and C freundii in 9.8% (20) of patients. The distribution of isolates were identified in the following sources: urine (46.2%), respiratory tract (16.3%), blood (15.8%), pus (12.1%), and sterile body fluids (9.3%). 3 The study discovered that C koseri had a high degree of resistance to antimicrobials: third-generation cephalosporins (85.3%), piperacillin (80.5%), aminoglycosides (46.3%), ciprofloxacin (81.9%), and carbapenems (7.3%). Among the beta-lactam/beta-lactamase inhibitor combinations, piperacillin/tazobactam had the lowest level of resistance (11.7%) in case reports worldwide. Citrobacter freundii is typically at a higher risk compared to other Citrobacter species for harboring AmpC resistance. Citrobacter koseri is naturally resistant to ampicillin but can gain antibiotic resistance over time through chromosomal and plasmid-mediated gene transfers. 4 Based on literature review, C koseri infective endocarditis (IE) is significantly rare, and only a few cases are reported.
Case Presentation
A 75-year-old male was admitted to the hospital due to complaints of nausea, decreased oral intake, progressively worsening weakness, and fevers for 3 days. His past medical history was significant for hypertension, hyperlipidemia, gastroesophageal reflux disease (GERD), chronic obstructive pulmonary disease (COPD), and benign prostatic hyperplasia. One week prior to presentation, the patient had undergone a cystoscopy for urinary retention with normal findings reported per his wife. He disclosed a longstanding history of alcohol use, consuming 1 pint of vodka daily for over 20 years, but denied prior intravenous drug use. He denied any primary genetic disorders affecting T-cell, B-cell, or combined immunodeficiency or secondary immunodeficiency disorders (HIV, diabetes mellitus, corticosteroids, etc.). Vital signs on admission were a blood pressure of 106/56 mm Hg, pulse of 125 bpm, respiratory rate of 25 breaths/min, and a temperature of 100.5°F. Physical examination revealed a nontender, distended abdomen with hypoactive bowel sounds, scleral icterus, and visible jaundice. Cardiac auscultation revealed tachycardia with a holosystolic murmur. Laboratory results showed a white blood cell count of 13 660/mm3, hemoglobin of 11.8 mg/dL, platelet count of 90 000/mm3, potassium of 2.9 mEq/L, creatinine of 1.70 mg/dL, magnesium 1.4 mg/dL, aspartate aminotransferase (AST) 54 U/L, alanine transaminase (ALT) 41 U/L, alkaline phosphatase (ALP) 131 U/L, total bilirubin 4.13 mg/dL, lactic acid 3.3 mmol/L, and urinalysis positive for pyuria and nitrites. The patient was resuscitated with intravenous fluids and empirically treated with vancomycin and cefepime. Computed tomography (CT) of the chest, abdomen, and pelvis showed right lower lobe pulmonary consolidation, mild esophageal wall thickening, cirrhotic liver, and mild urinary bladder wall thickening. The patient developed acute hypoxia necessitating noninvasive ventilation and closer monitoring in the intensive care unit. Transthoracic echocardiogram (TTE) suggested a probable mitral valve vegetation (Image 1). Blood cultures returned positive for C koseri; the blood isolate was resistant to ampicillin and indeterminate to fluoroquinolones, but susceptible to other beta lactams (ceftriaxone, cefepime, piperacillin/tazobactam, ertapenem, and meropenem) and aminoglycosides (tobramycin and gentamicin). His broad-spectrum antibiotics were de-escalated to ceftriaxone 2 g IV daily based on susceptibilities. An esophagogastroduodenoscopy (EGD) was requested by the cardiology service to rule out esophageal varices prior to a transesophageal echocardiogram (TEE) for further mitral valve mass evaluation. Esophagogastroduodenoscopy revealed a nonobstructive esophageal ring, Los Angeles (LA) grade A esophagitis, and gastritis. Urine culture, gastrointestinal panel, and endoscopy were unable to determine the primary source of infection. Transesophageal echocardiogram confirmed the presence of a large mobile vegetation measuring 2.3 cm × 1.6 cm attached to the anterior mitral valve leaflet. Therefore, the patient was diagnosed with native mitral valve endocarditis due to C koseri.

Transthoracic echocardiogram (TTE) showing large mitral valve vegetation.
He exhibited worsening mentation and right-sided facial droop, prompting an urgent workup for stroke. A CT head did not find any acute pathology, but magnetic resonance imaging (MRI) of the brain confirmed multiple punctate foci of acute infarction within the left frontal lobe, right lateral ventricle, and left occipital lobe (Image 2). These findings strongly suggested a septic embolic phenomenon involving the central nervous system (CNS). For better CNS penetration, ceftriaxone frequency was increased to 2 g IV twice daily dosing. Additional sets of blood cultures beginning 36 hours after the initial positive blood cultures were obtained for surveillance. These serial sets of blood cultures remained negative; however, the patient continued to clinically deteriorate. He developed increasing oxygen requirements, and chest x-ray suggested concerns for aspiration pneumonia in the right lower lobe. Ceftriaxone was switched to piperacillin/tazobactam for 5 days to treat his aspiration pneumonia. Owing to persistence of fever and worsening leukocytosis, in the setting of large valvular IE and likely stroke from septic thromboembolism, cardiothoracic surgery was requested to evaluate the patient. They deemed him not a surgical candidate for valvular surgery due to his critical clinical condition and multiple co-morbidities and recommended conservative management with medical therapy. Owing to prolonged hospitalization and clinical deterioration, his regimen was escalated to IV meropenem with plans to complete a 6-week course to treat his C koseri mitral valve IE. The patient later developed multiorgan failure requiring intubation with mechanical ventilation, non-ST-elevation myocardial infarction (NSTEMI), acute renal failure requiring continuous renal replacement therapy. On hospital day 36, his family requested comfort care measures and the patient passed away peacefully.
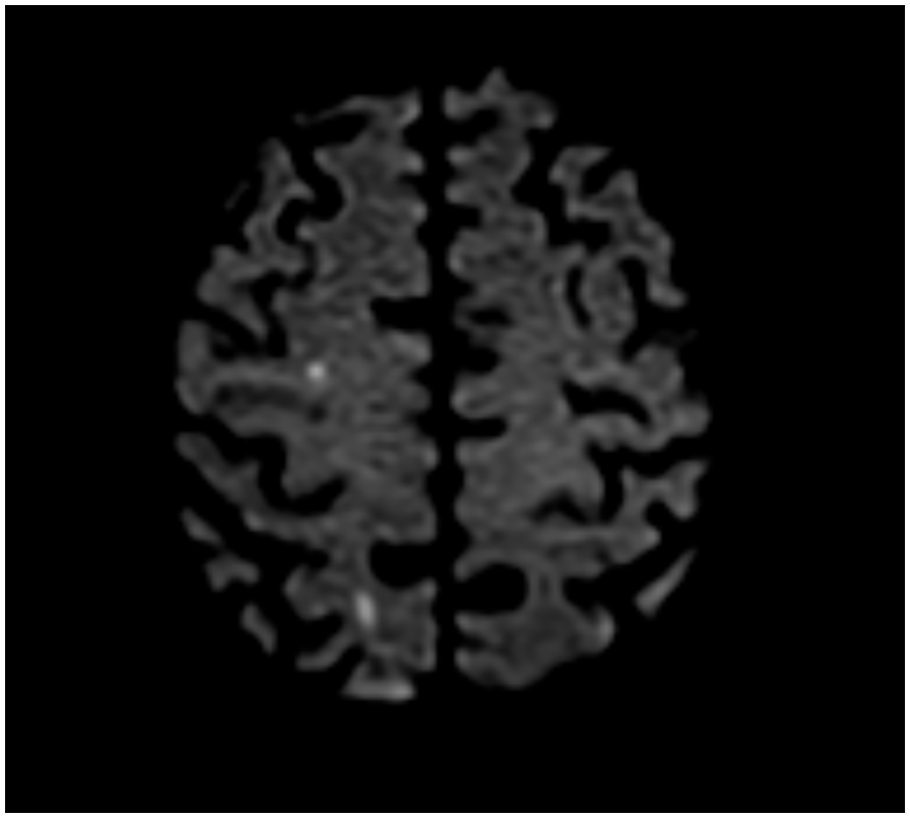
Magnetic resonance imaging (MRI) brain showing multiple punctate foci, suggestive of septic embolism and acute infarction.
Discussion
Current literature suggests that non-HACEK GNB IE is a very uncommon phenomenon with C koseri as the causative pathogen being even rarer. The American Heart Association (AHA) and Infectious Diseases Society of America (IDSA) 2015 management guidelines for gram-negative endocarditis give consideration for beta-lactam and aminoglycoside or fluoroquinolone combination therapy for 6 weeks as a Class IIa; Level of Evidence C recommendation based on one observational study.5,6 Although with notable limitations, Morepeth et al noted no statistically significant survival benefit in those with combination therapy in their cohort of non-HACEK GNB IE. Repeated blood cultures indicated quick clearance of bacteremia with empiric cefepime and was clinically stable prior to de-escalation. In addition, concurrent complicating factors (i.e. AKI) precluded use of various other adjunctive therapies such as aminoglycosides.
In addition, previous literature has suggested that anterior mitral valve vegetations have the highest embolic risk at 37% compared to general mitral vegetations (25%) and aortic vegetations (10%). Other studies suggest that embolic risk may be dependent on the infecting organism as well. Clinical guidance suggests early surgical evaluation in those with mobile vegetations measuring greater than 10 mm, especially in those with anterior leaflet mitral valve involvement. 6 More recent literature continues to highlight concerns of vegetation size with poor outcomes. In 2019, Fosbol et al noted that left-sided IE with large vegetations (greater than 10 mm) compared to smaller vegetations were associated with higher inpatient mortality (20.8% vs 15.2%, respectively) and 6-month mortality (25.1% vs 19.4%); however, when surgical intervention was performed, outcomes were similar regardless of the vegetation size. Guidelines also recommend consultation with an infectious disease expert given possible concern for resistance. Finalized in vitro susceptibilities indicated limited beta-lactam resistance. Multiplex polymerase chain reaction (PCR) panel did not indicate any significant genotypic resistance (i.e. CTX-M, KPC, NDM, OXA-48, VIM, IMP). Although various gram-negative organisms have the potential to express inducible resistance (i.e. AmpC B-lactamases), recent literature indicate E cloacae complex, K aerogenes, and C freundii as the highest risk for clinically significant AmpC production in the Enterobacterales family. 7 Although various enteric organisms, including some Citrobacter species, may possess the ability to produce AmpC, clinical relevance may be insignificant due to low overall B-lactamase production, even in the presence of inducing agents; C koseri does not appear to have chromosomally encoded AmpC beta-lactamase.8,9 In Polsfuss et al’s 9 prospective study, 74 of the 2129 bacterial isolates were C koseri; none of the isolates were ampC positive.
We report a patient with no documented primary or secondary immunodeficiency disorder who was de-escalated to ceftriaxone based on susceptibilities with data to suggest limited to no risk of ampC beta-lactamase production.7,9,10 He clinically decompensated while on appropriate antibiotics, but numerous surveillance repeated blood cultures remained negative. Ultimately, antimicrobial therapy switched to IV meropenem due to prolonged hospitalization and risk of nosocomial infections. Based on available literature at the time and clinical situation, ceftriaxone monotherapy appeared to be a reasonable therapeutic option with limited risk for treatment failure due to resistance. Without surgical intervention, the patient carried a high risk for mortality and embolic phenomena, due to the size and location of vegetation. There still remains a paucity of data and guidance in the management of non-HACEK GNB IE, especially without surgical intervention. Additional case reporting and investigation is needed to add to the limited literature available.
Footnotes
Acknowledgements
The content of this publication are the sole responsibility of the authors and also certify all information presented is their own opinion and not that of their employers.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article.
Prior Presentation of Abstract Statement
This case or abstract has not previously been submitted or presented.
