Abstract
We present a case of a 42-year-old male with sarcoidosis manifesting as endobronchial mass-like lesions, a rare and atypical presentation of the disease. Sarcoidosis typically involves the respiratory system, but its occurrence as endobronchial polyps mimicking malignancy is uncommon. The diagnosis was confirmed through bronchoscopy and biopsy, revealing non-caseating granulomas. Treatment with corticosteroids led to significant clinical improvement. This case underscores the importance of considering sarcoidosis in the differential diagnosis of endobronchial masses and the role of biopsy in confirming the diagnosis.
Introduction
Sarcoidosis is a granulomatous disease that affects multiple organ systems, with an unknown cause. It is defined by the formation of non-caseating granulomas in the organs involved. 1 The typical radiologic appearance of pulmonary sarcoidosis is bilateral hilar lymphadenopathy with peribronchovascular nodules. Rarely, it can manifest with pulmonary cavitation or pleural involvement, but reports of sarcoidosis presenting as an endobronchial lung mass are rare. 2 Our report highlights a rare presentation of sarcoidosis characterized by multiple partially obstructing airway polyps, clinically resembling malignancy.
Case Presentation
A 42-year-old male with no significant medical history presented with progressively worsening dyspnea and exertional cough, which began 4 months prior following a respiratory infection. The patient also reported a 33-pound unintentional weight loss due to decreased appetite, accompanied by nocturnal sweating over the past several months. He denied recent exposure to illness or any travel within the last 2 years. On admission, his vital signs were stable, with oxygen saturation at 98% on room air, and no signs of respiratory distress. Lung auscultation was normal. Initial chest radiography revealed diffuse infiltrates in the left lung, a 2-cm nodular opacity in the left lower lobe, and patchy atelectasis at the right lung base (Figure 1). A computed tomography (CT) of the thorax with contrast demonstrated extensive interstitial reticular thickening, raising concern for lymphangitic carcinomatosis, along with mediastinal and hilar lymphadenopathy (Figure 2). Given the suspicion of malignancy, a CT of the abdomen and pelvis with contrast was performed, which showed no significant lymphadenopathy in the retroperitoneal, mesenteric, or inguinal regions. Laboratory results indicated leukopenia on a complete blood count (CBC). Interferon-gamma release assay, HIV antibodies, blood and sputum cultures, hepatitis panel, antinuclear antibodies (ANA) rheumatoid factor (RF), and peripheral smear were unremarkable.
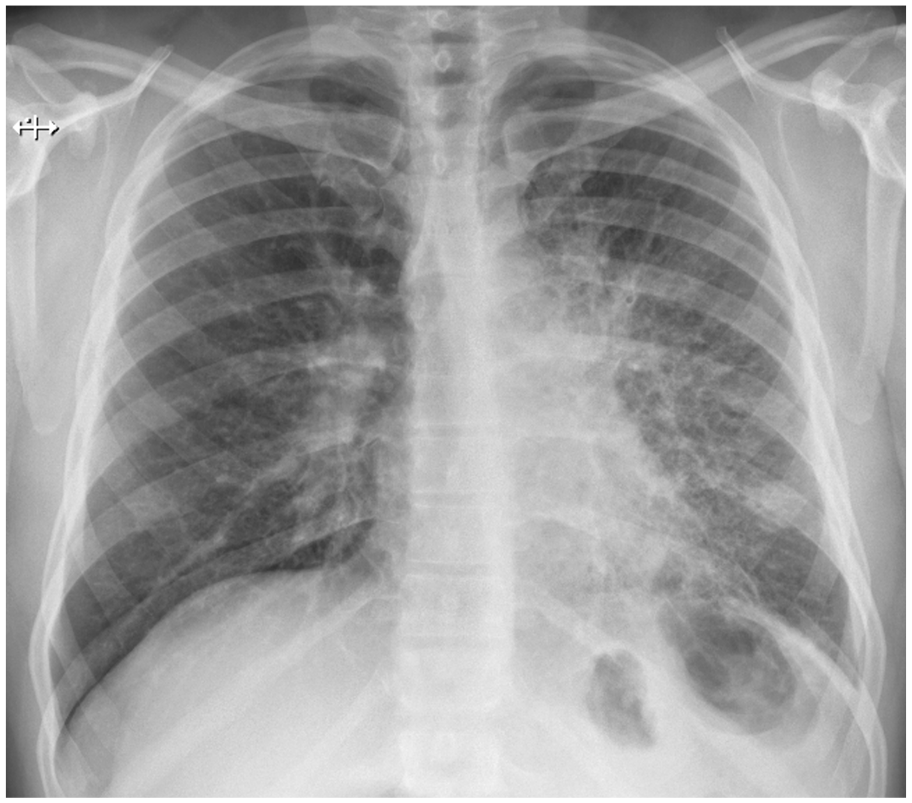
Diffuse infiltrates throughout the left lung along with patchy atelectasis at the right lung base. There is a 2-cm nodular focus overlying the left lower lobe suspicious for a mass.

Extensive interstitial reticular thickening, concerning for lymphangitic carcinomatosis along with mediastinal and hilar lymphadenopathy.
Consequently, the patient underwent bronchoscopy, which revealed friable mucosa throughout the airway with small, non-obstructing endobronchial polyps at the left upper and lower lobe orifices, as well as a partially obstructing polypoid lesion at the right lower lobe orifice (Figure 3). Cytology from brush samples demonstrated benign reactive respiratory epithelial cells with a background of lymphocytes and histiocytes. Bronchoalveolar lavage (BAL) fluid showed lymphocytosis, negative for malignant cells, acid-fast bacilli, and gram stains. The CD4/CD8 ratio in BAL fluid was 2.06. Multiple transbronchial and endobronchial biopsy specimens showed confluent non-caseating granulomas consistent with sarcoidosis (Figure 4). Corticosteroid treatment with prednisone 50 mg was started, which resulted in significant clinical improvement within 1 week during his hospital stay. The patient was discharged on a regimen of prednisone 50 mg daily for 1 month with a gradual taper and close pulmonology follow-up. Trimethoprim and sulfamethoxazole prophylaxis for Pneumocystis jiroveci was also initiated. During follow-up visits, the patient reported a complete resolution of respiratory symptoms. A repeat high-resolution CT of the chest showed notable improvement in mediastinal and hilar lymphadenopathy, as well as resolution of areas of consolidation and diffuse peribronchovascular interstitial changes (Figure 5).

Small, non-obstructing endobronchial polyps at the left upper and lower lobe orifices, as well as a partially obstructing polypoid lesion at the right lower lobe orifice.

Confluent non-caseating granulomas consistent with sarcoidosis.

Resolution of lymphadenopathy and remarkable improvement of the confluent areas of consolidation and diffuse peribronchovascular interstitial changes.
Discussion
Worldwide, sarcoidosis is most common in young and middle-aged people of all racial and gender identities; it is more common in Scandinavian nations and among African Americans, and less common in populations of Hispanic and Asian descent. 3 Sarcoidosis, a multisystemic granulomatous disease of unknown etiology, presents with a wide range of clinical manifestations, although nearly all patients experience respiratory tract involvement at some stage of the disease. 4 Sarcoidosis has a mortality rate of approximately 7% within a 5-year period, with over 10% of patients with pulmonary sarcoidosis developing progressive disease, and more than 60% of deaths attributed to advanced pulmonary involvement. 5 In the 1960s, Guy Scadding developed a “staging system” for pulmonary sarcoidosis using chest radiography: stage 1 is defined by the presence of bilateral hilar lymphadenopathy (BHL); stage 2 consists of BHL and parenchymal infiltrates; stage 3 is characterized by parenchymal abnormalities without BHL; stage 4 consists of upper-lobe predominant fibrotic changes with volume loss. 6 The staging system cannot reliably predict outcomes due to significant overlap between stages, and radiographic resolution is possible at nearly every stage, except for stage 4. 4 Chest X-ray is the initial imaging and is around 90% abnormal in sarcoidosis, even in asymptomatic patients, with the most common findings being bilateral hilar lymphadenopathy and parenchymal changes. 7 High-resolution CT chest (HRCT) can be done to evaluate the thoracic lymph nodes and lung parenchyma mainly. Typical features of sarcoidosis on HRCT are micronodules spread along the bronchovascular bundles in a distinctive lymphatic distribution, predominantly affecting mid-to-upper zones. 4 Sometimes, micronodules can conglomerate and lead to mass-like lesions along with distortion of parenchyma, creating the galaxy sign, which is considered typical for pulmonary sarcoidosis. 4 The HRCT may show reduced airway luminal diameter from endobronchial granulomas, extrinsic compression of the airways due to lymphadenopathy, bronchial distortion, and mural thickening; nevertheless, its utility in evaluating airway patency is limited.4,7 Bronchoscopy has the highest diagnostic yield and is employed to direct airway visualization as well as confirm the diagnosis when more accessible biopsy sites are unavailable. Airway abnormalities are observed in up to 60% of sarcoidosis patients, with bronchoscopic findings ranging from non-hypertrophic lesions such as erythema, edema, cobblestone mucosa, mucosal plaques, and nodules, to hypertrophic, tumor-like lesions, necessitating a biopsy to confirm the presence of non-caseating granulomas. 7 Non-caseating granulomas are the hallmarks of the disease; however, accurate diagnosis can be achieved by clinicopathologic correlation to exclude other non-sarcoid granulomatous diseases causing endobronchial abnormalities, including but not limited to microscopic polyangiitis, histoplasmosis, blastomycosis, coccidioidomycosis, mycobacterial infections, chronic berillyum disease, syphilis, amyloidosis, radiation-induced mucositis, and malignant neoplasms. 7 Generally, BAL analysis is performed to rule out other possible diagnosis. However, when non-caseating granulomas in a transbronchial biopsy were combined with lymphocytosis greater than 15%, a CD4+/CD8+ ratio greater than 3.5, and a combined CD103+CD4+/CD4+ ratio less than 0.2, it is considered highly suggestive of sarcoidosis.8,9 However, because it might be elevated, normal, or even decreased, the CD4+:CD8+ ratio has been shown to be less significant. 10
Polyps and endobronchial mass-like lesions arising from bronchial granulomas are rarely reported presentations of pulmonary sarcoidosis, and they can mimic malignant masses; therefore, they warrant biopsies from non-contiguous sites for diagnostic accuracy. 2 ,2,11-14 The first-line treatment for pulmonary sarcoidosis is systemic glucocorticoids; however, the optimal dose and length of therapy are unknown. Approximately 30% of the patients experience relapse following corticosteroid tapering or cessation; in these cases, disease-modifying antirheumatic drugs (methotrexate, azathioprine) are recommended in addition to corticosteroids as an alternative to corticosteroid dose escalation or in patients at risk of steroid toxicity. 15 Although in some case reports, complete clearance of airway obstruction with conventional treatment was reported, current literature provides insufficient data on how steroids have an effect on endobronchial lesions.7,16,17
Conclusion
Clinicians should remain vigilant for airway involvement in patients with sarcoidosis, as it can pose diagnostic challenges. This condition may present with persistent symptoms, such as unexplained cough, shortness of breath, or wheezing, despite symptomatic treatment, complicating the clinical management. Bronchoscopy followed by endobronchial and transbronchial biopsies from non-contagious sites are crucial diagnostic tools in differentiating sarcoidosis from malignancy.
Footnotes
Acknowledgements
The authors sincerely thank the patient for their consent to use clinical information and imaging, which made this work possible. Their willingness to contribute to medical research is greatly appreciated.
Authors’ Note
The Case Presentation and Images sections of this article have been published as an abstract and presented as a poster with the same title at the Chest Annual Meeting on October, 2024.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
