Abstract
We describe a 30-year-old Caribbean-Black woman with a clinical presentation suggestive of a transient ischemic attack (TIA) with no conventional cerebrovascular risk factors, albeit with a newly diagnosed quadricuspid aortic valve (QAV) with moderate aortic regurgitation (AR). Although QAV is a recognized congenital cardiac defect, its association with TIA remains elusive. This case highlights the importance of considering potential atypical etiologies, such as QAV, in the evaluation and management of young patients presenting with cerebrovascular events.
Introduction
The aortic valve (AV), an integral cardiac structure, ensures unidirectional blood flow from the left ventricle to the aorta. Its normal anatomy comprises 3 leaflets (tricuspid) that coapt during the cardiac cycle.1,2 However, a rare congenital anomaly known as the quadricuspid aortic valve (QAV) presents with an additional leaflet, deviating from this standard architecture. The estimated prevalence of QAV in the general population is exceedingly low, ranging from 0.0003% based on autopsy studies to <1% in individuals undergoing aortic valve replacement (AVR) for aortic regurgitation (AR).3,4
A transient ischemic attack (TIA) is a temporary interruption of cerebral perfusion, where the inciting thrombus usually undergoes autolysis or is dislodged, and the symptom duration is typically less than 5 minutes but can last up to 24 hours.5-8 However, emerging evidence suggests a possible link between QAV and an accentuated risk of stroke, particularly in individuals without conventional risk factors. 9 The mechanisms linking QAV to stroke are not well elucidated; however, the authors postulate that the additional leaflet results in turbulent hemodynamics, which can potentially dislodge atheroemboli from the aortic valvar complex, leading to cerebral ischemia as well as thoracic aortic blood flow reversal.9,10
Given the paucity of cases linking QAV and TIA, we describe a 30-year-old Caribbean-Black woman with a clinical presentation suggestive of a TIA with no conventional cerebrovascular risk factors, albeit with a newly diagnosed QAV with moderate AR.
Case Presentation
A 30-year-old Caribbean-Black woman with no prior medical history presented to the emergency department with suspected stroke symptomatology, which included sudden onset of left-sided weakness and slurred speech. The symptoms began abruptly 30 minutes before arrival and progressively worsened. She denied any history of headache, visual disturbances, presyncope, or syncope. She did not report any antecedent infection or recent ill contacts upon evaluation. The patient denied head trauma, current prescription or complementary and alternative medication use, or pertinent travel and pet history. She maintained her usual dietary intake and physical activity levels, with no lifestyle modifications preceding her symptom-complex onset. She denied any tobacco use, illicit drug use, or significant alcohol consumption. Family history was non-contributory for cardiovascular, cerebrovascular, or neurodegenerative disease.
On physical examination, she was alert and oriented but exhibited significant left-sided hemiparesis and dysarthria. Blood pressure was 143/88 millimeters of mercury (mm Hg), heart rate was regular and 87 beats per minute, and respiratory rate was tachypneic at 22 breaths per minute with pulse-oximetry saturation of 99% on room air. Neurological examination revealed left facial droop, upper and lower extremity weakness (3/5 on the Medical Research Council [MRC] muscle strength scale), and left-sided neglect. Cardiopulmonary auscultation revealed a grade 3/6 diastolic murmur, loudest at the left lower sternal border, with normal jugular venous waveform pulse and apical impulse. Her vesicular breath sounds were equal and bilateral. There was no sacral or peripheral edema.
She was subsequently admitted to the neurology ward, where she underwent a comprehensive diagnostic evaluation while her symptoms nearly completely normalized. Her routine blood investigations, including high-sensitivity cardiac troponin, d-dimer and NT-pro-brain natriuretic peptide, glycated hemoglobin, fasting lipid panel, and thyroid profile, were unremarkable. A 12-lead electrocardiogram did not reveal any acute, dynamic changes consistent with ischemia or infarction. A chest radiograph demonstrated a normal cardiac silhouette without any evidence of air-space disease or pulmonary edema. An initial, non-contrast computed tomography (CT) of the brain did not reveal any acute intracranial finding, which was swiftly followed by magnetic resonance imaging and angiography (MRI/MRA), which also revealed no significant abnormalities within the visualized intracranial vasculature with no evidence of arteriovenous malformation (AVM) or aneurysm (Figure 1). A 2-dimensional transthoracic echocardiogram (2D-TTE) demonstrated a characteristic appearance of the AV leaflets consistent with Hurwitz-Roberts class A and Nakamura type I QAV, preserved ejection fraction without diastolic dysfunction. The leaflets displayed an “X” configuration during diastole and a “square” configuration during systole.11,12 Color flow Doppler interrogation revealed a central regurgitant jet indicative of moderate AR, evidenced by a prolonged pressure half-time of 460 ms (Figures 2 and 3). A transesophageal echocardiogram also reflected similar valvular findings, with no patent foramen ovale or atrial septal defect observed (Figure 4). A 24-hour Holter monitor did not reveal any atrial fibrillation (AF) or advanced heart block.

The patient’s brain magnetic resonance imaging (MRI) series. (A) This is an apparent diffusion coefficient (ADC) image of the patient’s brain, which did not reveal any acute intracranial lesion. (B) This is a diffusion-weighted image (DWI) image of the patient’s brain, which did not reveal any acute intracranial lesion.
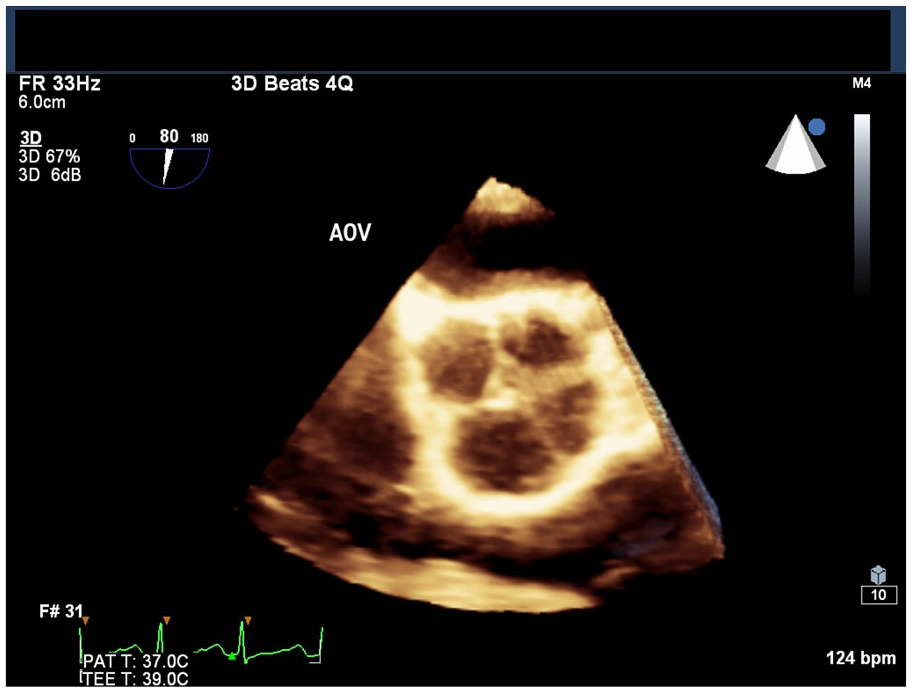
The patient’s 2- and 3-dimensional transesophageal echocardiogram displays the Hurwitz-Roberts class A and Nakamura type I quadricuspid aortic valve (QAV).

The patient’s 2-dimensional transthoracic (2D-TTE) echocardiogram. (A) The parasternal short-axis view (zoomed in on the aortic valve) displays the Hurwitz-Roberts class A and Nakamura type I quadricuspid aortic valve (QAV). The leaflets display an “X” configuration during diastole and a “square” configuration during systole. (B) The parasternal long axis (zoomed in on the aortic valve) view displays the Hurwitz-Roberts class A and Nakamura type I quadricuspid aortic valve (QAV). Color flow Doppler interrogation revealed a central regurgitant jet indicative of moderate aortic regurgitation, evidenced by a prolonged pressure half-time of 460 ms.

The patient’s transesophageal (TEE) echocardiogram. (A) A short-axis (zoomed in on the aortic valve) view displaying the Hurwitz-Roberts class A and Nakamura type I quadricuspid aortic valve (QAV) in an open position. (B) A short-axis (zoomed in on the aortic valve) view displaying the Hurwitz-Roberts class A and Nakamura type I quadricuspid aortic valve (QAV) in a closed position. There is evidence of mild sclerosis and calcification. (C) A view of the interatrial septum demonstrating no patent foramen ovale, atrial septal defect, or bubble contrast in both left and right atria soon after injection.
The following day, there was an interval complete resolution of her symptoms. She was initiated on guideline-directed medical therapy (GDMT) of her AR, which comprised aspirin, high-intensity rosuvastatin, low-dose valsartan-sacubitril, bisoprolol, finerenone, and empagliflozin. She underwent 24 hours of neurological observation to ensure continued stability. During her hospitalization, the patient also underwent extensive rheumatologic and thrombophilia panel testing, both of which were unremarkable (Table 1). The remainder of her hospital course was uneventful, and she was eventually discharged with scheduled outpatient cardiology appointments for continued management of her moderate AR.
The Patient’s Rheumatologic and Thrombophilia Panel.

Discussion
Despite the well-established association between stroke and aging, there is an increasing incidence of stroke in young adults aged 18 to 45 years.13-18 Established stroke risk factors include diabetes mellitus (DM), hypertension (HTN), dyslipidemia, AF, and obesity, among others. 19 Cardioembolic phenomena and congenital heart defects contribute significantly to stroke presentations in young adults.20,21
The QAV, a rare congenital cardiac abnormality in young adults with a prevalence of 0.005%, presents unique challenges.22,23 Traditionally, the QAV embryological defect was attributed to anomalous septation of the conotruncus and the endocardial cushions, potentially triggered by an inflammatory episode.24,25 The Hurwitz and Roberts 11 classification system categorizes QAVs into 7 subtypes (A-G) based on the relative size and configuration of the 4 cusps. Clinical presentation is usually contingent on the AV functional status, myocardium, and associated cardiovascular conditions; however, its predominant functional abnormality is AR, compared with aortic stenosis in young adults.3,4,26 Transesophageal echocardiography is considered the gold standard for diagnosis and follow-up of a patient with QAV.27,28 Natural history data suggest that valve dysfunction typically manifests during the fourth decade of life, with progressive deterioration during the following 2 decades.3,4 The AVR or repair is usually warranted in the fifth or sixth decade, and approximately one-third of the patients require surgery.28,29 Multimodality imaging is imperative in assessing patients with QAV with significant AR and may potentially improve their management strategy. Recently, it has been demonstrated that CT regurgitant orifice area may be a potential ancillary prognostic marker.29,30 The AV surgery is the definitive treatment strategy in patients with AR secondary to QAV; however, the long-term sequelae of this condition remain largely unknown. 31 Surgical intervention remains the cornerstone therapeutic option before incipient heart failure. 9
The majority of international guidelines are concordant and harmonized with respect to the initial assessment of AR and surgical indications in patients with symptomatic severe AR. 32 Left ventricular ejection fraction (LVEF) remains a powerful prognosticator in patients with moderate AR, with LVEF ≤55% being a deleterious marker, and LVEF 35% to 55% may represent a subgroup that may benefit from early surgical correction. Our patient’s AR was considered moderate by echocardiographic criteria with a preserved LVEF, and although she experienced a TIA, she opted for interval surveillance with multimodality imaging, given her apprehension for an immediate surgical correction.33,34 Although she was young and did not display typical risk factors for TIAs, the QAV with resultant valve dysfunction appeared to be a potential culprit. The mechanisms linking QAV to stroke are not well elucidated; however, the authors’ pathophysiological hypothesis posits that the additional leaflet results in turbulent hemodynamics, which can potentially dislodge atheroemboli from the aortic valvar complex, leading to cerebral ischemia.9,10 The AR may also affect undetermined embolic stroke in patients by accentuating thoracic aorta flow reversal.35,36
Our patient was initiated with GDMT, which comprised aspirin, high-intensity rosuvastatin, low-dose valsartan-sacubitril, bisoprolol, finerenone, and empagliflozin for the management of her moderate AR. Notably, she tolerated this regimen without hemodynamic complications. Pharmacotherapeutic management is crucial in selected AR patients, especially in those detected early; however, it should be underscored that it is not an optimal substitute for AV replacement.37,38
Conclusion
We describe a 30-year-old Caribbean-Black woman with a clinical presentation suggestive of a TIA with no conventional cerebrovascular risk factors, albeit with a newly diagnosed QAV with moderate AR. Although a definitive causal link between QAV and TIA and stroke necessitates further exploration, this presentation underscores the importance of multimodality imaging and interim GDMT. Early diagnosis and management of potential TIA-related QAV is critical for optimizing patient outcomes and preventing future cerebrovascular events.
Footnotes
Author Contributions
All authors contributed equally to writing the manuscript, and all authors read and approved the final manuscript.
Data Availability Statement
All available data can be obtained by contacting the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
