Abstract
Light-chain multiple myeloma (LCMM) is a rare subtype of plasma cell neoplasm, usually linked to kidney involvement and lytic bone lesions. However, case presents as osseus tumors are very uncommon. A 63-year-old male patient complained of persistent rib pain. Computed tomographic imaging showed an isolated bone tumor in the eighth rib. Further positron emission tomography–computed tomographic scan revealed multiple lytic bone lesions in other areas. Biopsy of the rib lesion confirmed the presence of plasma cells producing kappa light chains. The patient received 4 cycles of daratumumab, bortezomib, lenalidomide, and dexamethasone treatments, resulting in significant improvement. Reviewing literatures, osseus tumor with osteosclerotic lesions has been rarely described in LCMM, underlining the challenge in diagnosis and stressing the importance of considering LCMM in the differential diagnosis of bone tumors.
Introduction
Plasma cell neoplasms (PCNs) are disorders characterized by the abnormal growth of cloned plasma cells, which produce monoclonal immunoglobulin known as paraprotein or M-protein. These neoplasms can lead to excessive proliferation of plasma cells, the spread to bones, and rarely in soft tissues. 1 Multiple myeloma (MM) is more common in men, particularly in the African American population, with a median age of 65 years. 2 Clinical symptoms of MM include hypercalcemia, anemia, osteolytic bone lesions, and renal insufficiency. The myeloma protein, also known as M-protein, is typically composed of immunoglobulins IgG, IgA, or IgM. A special type of MM, most commonly known as light-chain multiple myeloma (LCMM), is less common than other forms of MM and is characterized by malignant plasma cells that cannot produce heavy chains, leading to the exclusive production of light chains. As a result, no M-spike is detectable in serum protein electrophoresis. This variant is associated with a poorer prognosis and often presents with acute renal failure and multiple lytic bone lesions. 3
We present a case of a patient with LCMM who initially presented with a bone tumor without renal failure, which is uncommon.
Case Presentation
A 63-year-old man was referred to hematology clinic for persistent chest pain and possible bone tumors. The computed tomographic (CT) scan of the thorax revealed a dominant bone tumor in left eighth rib (Figure 1) with multiple thoracic osseous tumor lesions including right upper lobe 4-mm lung nodule and left lower lobe 6-mm lung nodule. The left eighth rib lesion was suspicious for sarcoma. A positron emission tomography-computed tomography (PET/CT) scan was performed to evaluate the patient’s condition. The PET imaging is valuable in assessing whether the patient has bone metastasis and appears to be more sensitive than bone scan which can help to define the presence of CRAB features (Calcium elevation, Renal failure, Anemia, and Bone lesions) to guide diagnostic and treatment decisions in cases such as MM. 4

Low-dose chest CT without contrast reveals a single bone tumor–like lesions in the left eighth rib.
His PET/CT showed multiple hypermetabolic lesions, including a prominent destructive lytic lesion approximately 7.6 cm × 3.7 cm on the left eighth rib, consistent with findings from the standard CT scan. In addition, there are other lytic lesions observed throughout the thorax and other ribs (Figure 2). A biopsy was performed on the left eighth rib lesion, which showed fibromuscular tissue diffusely infiltrated by atypical plasma cells that express strong and diffuse MUM1 and kappa light-chain restriction by in situ hybridization. The “kappa light chain restriction” indicates that the malignant plasma cells are producing only kappa light chains, which is a key feature of myeloma and the “diffuse” pattern suggests that neoplastic cells are widespread throughout the tissue sample. The neoplastic cells were MUM1, CD138, CD20, and cyclin D1 positive, which are all markers typically associated with plasma cells involved in myeloma. Overall findings were consistent with extensive involvement by PCN (Figure 3). Laboratory tests showed a white blood cell count of 8.54 × 10³/µL, hemoglobin count of 11.7 g/dL, mean corpuscular volume of 94.9 fL, and a platelet count of 261 × 10³/µL. The alkaline phosphatase level was 142 U/L, IgM was 26 mg/dL (normal range: 35-242 mg/dL), IgG was 803 mg/dL (normal range: 610-1660 mg/dL). The Ig kappa free light chains (FLCs) were 739 mg/dL (normal range: 0.33-1.94 mg/dL), and the Ig lambda chains were 1.29 mg/dL (normal range: 0.57-2.63). The FLC ratio was 573 (normal range: 0.26-1.65). Serum beta-2 microglobulin was 2.5 mg/L (normal range: 0.8-2.2 mg/L), and albumin was normal. Peripheral blood smear showed no plasma cells. Bone marrow also revealed a 39% infiltration of plasma cells, consistent with the pattern observed in the rib biopsy. Bone marrow cytogenetic studies detected complex cytogenetic abnormalities (46,XY,+5[5p15.33;q35.3], +7[7p22.1;q36.3], +9[9p24.3;q34.3], −13[13q12.11;q34], +19[19p13.3;q13.43]), indicating a hyper diploid. The detection of abnormalities such as hyper diploid clones indicates a more aggressive disease and is often associated with a higher risk of progression. In the context of the specific findings (46, XY, +5, +7, +9, −13, +19), these chromosomal alterations may suggest a poor prognosis and guide treatment decisions, including the need for more intensive therapy or closer monitoring. The Congo red stain for amyloidosis was negative, and the 24-hour urine protein was within the normal range, making the diagnosis of MM with systemic amyloidosis unlikely. Patient received daratumumab, bortezomib, lenalidomide, and dexamethasone (D-VRd), which is a well-established regimen for treating high-risk plasma cell disorders, particularly MM. While D-VRd is a powerful regimen, alternatives exist such as VRd regimen (bortezomib, lenalidomide, dexamethasone), KRD (carfilzomib, lenalidomide, dexamethasone). Clinical studies have shown that D-VRd improves overall response rates and progression-free survival compared with other regimens, making it a preferred option for high-risk cases. 5 After 4 cycles of this treatment, the patient achieved clinical remission and his FLC ratio returned to normal (FLC ratio:1.54). After 5 cycles of therapy, he was referred to Mount Sinai Hospital for a bone marrow transplantation, which offers state-of-the-art facilities equipped with the latest technologies for both the transplantation process and post-transplant care.

Whole-body PET/CT scan: The image shows left eighth destructive lytic lesion approximately 7.6 cm × 3.7 cm. Right scapula 7.5-cm lytic lesion, and multiple other lytic lesions throughout the thorax and other ribs.

Paraclinical features of the patient.
Discussion
Light-chain multiple myeloma is an aggressive subtype of MM, accounting for approximately 15% of cases and is associated with lower survival rates compared with other types.2,3,6 Unlike classical MM, LCMM is characterized by the exclusive production of light chains. Around two-thirds of LCMM cases produce kappa light chains, whereas the remaining third produce lambda light chains.3,6 The exact mechanisms behind the abnormal production of light chains are not yet fully understood. However, Magrangeas et al 6 have shown using fluorescence in situ hybridization that patients with LCMM do not have the typical IgH gene rearrangement seen in classical MM.
Patients with LCMM often experience a higher incidence of kidney failure, bone disease, and systemic light-chain AL amyloidosis. 2 Studies have indicated that kidney disease occurs in approximately 55% of LCMM cases, with bone lesions present in about 53% and systemic amyloid light chain amyloidosis in 5% to 10% of cases. Kidney involvement is especially common in LCMM and is more prevalent compared with other subtypes of MM.2,3,7 This condition, known as “myeloma kidney,” is caused by the deposition of monoclonal FLCs in the renal system. The severity of renal injury in patients with LCMM is closely related to the concentration of FLCs in the urine, with higher levels associated with more severe damage.
The pathophysiology of myeloma kidney involves the buildup of high concentrations of FLCs in the proximal tubules of the kidneys, which impairs reabsorption and leads to the deposition of FLCs and cast formation in the loop of Henle. This process ultimately contributes to renal injury.
Multiple lytic bone lesions are often associated with advanced stages of MM but may not always be present at the initial diagnosis. 2
Myeloma bone disease can manifest as discrete lytic lesions, widespread osteopenia, or multiple lytic lesions affecting various skeletal areas.
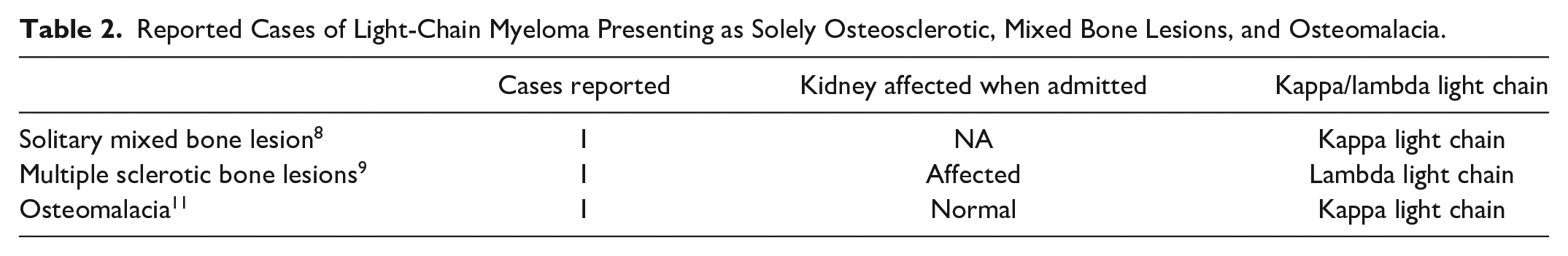
A study by Zhang et al 3 examining the clinical characteristics of patients with LCMM in China found that bone pain was the primary symptom in 77.1% of 96 patients, with 81.3% exhibiting more than 3 areas of bone destruction during disease progression. Another review by Cong et al 8 also demonstrated similar results, showing most patients presented with multiple lytic lesions, whereas only one showed osteoporosis. Despite a high prevalence of bone abnormalities, skeletal surveys revealed positive findings in only 17% of patients with LCMM at the initial presentation. 9 The Cosmos database also reflected a similar scenario, as only 12% of patients showed lytic bone lesions on x-ray when admitted between 2018 and 2024. Typically, LCMM presents with lytic bone lesions, and initial manifestations of osteosclerotic bone lesions are very rare. Table 1 gives a comprehensive summary of patient characteristics from various studies on LCMM. Table 2 summarizes case reports of LCMM presenting as bone lesions.11-13 Divakar et al 12 reported a case of LCMM with multiple osteosclerotic lesions, in which a 60-year-old female patient presented with low back pain for 2 months. The PET/CT showed multiple skeletal lesions in various areas. Biopsy and immunohistochemistry confirmed the diagnosis of Lambda light-chain myeloma. Fossaluzza et al also described a case revealing a solitary mixed lytic and sclerotic rib lesion with kappa light-chain myeloma. Our case not only exhibited multiple lytic bone lesions but also presented with a solitary osseus tumor with sclerotic lesions on the rib. 11 This atypical presentation may cause physicians to overlook considering MM as a diagnosis. In MM, primary sclerotic presentation occurs in only 3% of cases, indicating the rarity of such findings in both light chain and classical myeloma. 14 Multiple myeloma commonly forms osteolytic lesions, although in 1% to 3% of patients, it may be associated with osteosclerotic lesions. These osteosclerotic lesions have been reported in 2 groups: (1) focal osteosclerotic lesions associated with POEMS (polyneuropathy, organomegaly, endocrinopathy, M-protein, skin changes) syndrome and (2) myeloma with diffuse osteosclerosis. Another manifestation of bone lesions in light-chain myeloma is osteomalacia. 13 All the cases presented with lytic, sclerotic, and mixed bone lesions, but none of them experienced a solid bone tumor as an initial presentation. Our case report highlights a unique presentation of light-chain myeloma with a bone tumor with osteosclerotic lesions, and multiple lytic lesions, which has not been previously documented to our knowledge.
Presents a Comprehensive Summary of Patient Characteristics From Various Studies on Light-Chain Multiple Myeloma.

The Cosmos database is a large-scale, cloud-based data repository designed to manage and analyze health care–related data, often using standardized codes such as ICD-10 for categorizing and documenting medical diagnoses.
Of 31 patients examined, 27 manifested osteolytic lesions, 1 osteoporosis.
During the disease progression, there were 78 (81.3%) patients had more than 3 areas of bone destruction.
Reported Cases of Light-Chain Myeloma Presenting as Solely Osteosclerotic, Mixed Bone Lesions, and Osteomalacia.
Another rare complication of lambda LCMM is AL light-chain systemic amyloidosis, which is observed in only 5% to 10% of patients.2,15 Other rare clinical presentations include skin involvement, liver plasmacytomas, epidural plasmacytoid tumors, subglottic plasmacytoma, and mediastinal lymph node involvement.16 -19 Studies have shown higher response rates in patients with LCMM treated with bortezomib and bendamustine.3,20 The combination of bortezomib with bendamustine and prednisone has rapidly reduced light-chain production, preventing irreversible renal failure.21 -24 Recently, the anti-CD38 monoclonal antibody daratumumab in combination with lenalidomide and dexamethasone has been introduced for treating relapsed/refractory MM and light-chain amyloidosis, significantly improving progression-free survival. Studies have shown that the daratumumab group had a significantly greater overall response rate compared with the control group, with a higher complete response rate. 25
Conclusion
We demonstrated that light-chain myeloma can present as osteosclerotic bone lesions or osseus tumors. Recognizing this atypical presentation is crucial for accurate diagnosis and treatment. Therefore, thorough imaging and the use of specific immunohistochemical markers are essential to ensure timely and effective management of this condition.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JCW received research grant from Kartos Therapeutics Incorporation; others have no conflict interests with the pharmaceutical company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Brookdale Research Fund.
Ethics Approval
Our institution doesnot require ethical approval for case report publication.
Informed Consent
Written informed consent for patient information to be published in this article was obtained from the patient.
