Abstract
Extramedullary myeloma (EMM) is an infrequent but well-established manifestation of multiple myeloma (MM), defined as a soft tissue plasma cell neoplasm without bone marrow involvement. Gallbladder involvement in EMM, however, is a very rare occurrence, with only 8 cases found in the English medical literature. Here, we present a case of an older adult male with a gallbladder mass in the presence of increasing serum kappa light chains after a normal bone marrow biopsy confirmed the complete remission of a previous MM diagnosis. Histopathologic evaluation of a biopsied sample confirmed the mass as an atypical plasma cell neoplasm. Later in his treatment, he developed several firm, smooth, violaceous skin nodules on the torso, which histopathology confirmed as also being atypical plasma cell neoplasms. We aim to contribute to the medical literature by expanding the pool of information regarding EMM of the gallbladder to support future diagnostic and treatment recommendations.
Introduction
Extramedullary myeloma (EMM), also known as an extramedullary plasmacytoma (EMP), is defined as a plasma cell neoplasm of the soft tissue without bone marrow involvement. 1 Extramedullary myeloma is an uncommon manifestation observed in those patients having a previous diagnosis (primary EMM) or current diagnosis (secondary EMM) of multiple myeloma (MM).2,3 The incidence is between 7% and 18% for primary EMM and up to 20% at relapse. 2 It can involve the skin, visceral organs, central nervous system, or skeletal system, and the presentation is variable depending on the type and extent of organ involvement. Gallbladder involvement in EMM, however, is extremely rare, with only 8 cases found in the English medical literature.4-11
MM classically arises in those aged between 65 and 70 years. 12 Clinical manifestations consist of hypercalcemia, renal insufficiency, anemia, and/or bone lesions (CRAB symptoms). EMM will present with similar symptomatology in the presence of soft tissue lesions, which are the manifestations of disseminated disease. 2 EMM is considered more aggressive than MM and confers a poorer prognosis. 13 The prevalence, however, has improved as novel therapies continue to be developed for the treatment of MM, prolonging life expectancy. 13 EMM patients are often treated with high-risk myeloma regimens, such as bortezomib, melphalan, and prednisone (VMP) or lenalidomide, bortezomib, and dexamethasone (RVD) with or without the addition of daratumumab in transplant-ineligible patients. In patients who are eligible for transplant, however, intensive anti-myeloma or anti-lymphoma regimens, such as bortezomib, thalidomide, and dexamethasone (VTD) and/or cisplatin, doxorubicin, cyclophosphamide, and etoposide (PACE) combined with stem cell transplant (SCT) have been proposed. In cases of relapse, treatment proposals are similar to that of transplant-eligible patients. Patients with relapsed MM or EMM may be treated with regimens, such as PACE, dexamethasone, cyclophosphamide, etoposide, and cisplatin (DCEP), or dexamethasone, carmustine [BCNU], etoposide, cytarabine, and melphalan (Dexa-BEAM). 14
We report a rare case of secondary EMM of the gallbladder. Our literature search retrieved 8 other cases of EMM involving the gallbladder in the English medical literature.4-11 Our patient presented to us after a previously resolved case of MM before developing EMM initially manifesting as a gallbladder mass and then progressing with the formation of cutaneous skin lesions.
Case Description
A 76-year-old male with a past medical history of relapsed IgG kappa MM presented to the outpatient clinic with generalized fatigue and weakness. His condition was diagnosed in his home country of India and he came to our clinic to discuss the management of his condition. At that time, he was taking a chemotherapeutic regimen of daratumumab and dexamethasone (DD) with the completion of 1 cycle. He was unable to tolerate the addition of pomalidomide or carfilzomib in the past due to severe cytopenia and fever. The physical exam was significant for a tired appearance and trace bilateral pedal edema. Baseline laboratory data were notable for a hemoglobin of 9.70 g/dl (n = 13.1-17.0 g/dL), indicating anemia, creatinine of 1.30 mg/dl (n = 0.70-1.30 mg/dL), serum calcium of 8.00 mg/dL (n = 8.50-10.10 mg/dL), and free kappa light chains measured at 40.0 mg/dL (n = 3.30-19.40 mg/dL). A peripheral blood smear demonstrated pancytopenia, moderate macrocytic anemia with marked anisopoikilocytosis, and a mild rouleaux formation. His most recent bone marrow biopsy from 5 months prior demonstrated focal hypercellular areas of 30% to 40% cellularity with dense sheets of atypical large plasma cells with prominent nucleoli. The cytogenetics karyotype was 46, XY.
Given the patient’s tolerance to DD, lenalidomide 10 mg daily by mouth was added to the regimen (DLD) with a 3-to-1 week-on, week-off schedule. He continued this regimen for 4 months until he developed another increase in serum-free kappa light chains and worsening anemia. The patient also began to experience right upper quadrant abdominal pain without nausea, vomiting, or diarrhea. Over-the-counter nonsteroidal anti-inflammatory drugs (NSAIDs) were able to relieve his pain. A comprehensive metabolic panel revealed bilirubin and liver function tests (LFTs) within normal limits. Another bone marrow biopsy was performed at this visit, demonstrating normal cellularity and cytogenetics with no evidence of MM.
During the next follow-up visit, he continued to have complaints of intermittent abdominal pain. Laboratory values at this visit were significant for another elevation in serum-free kappa light chains to 7000 mg/dL. The 18-fluorodeoxyglucose positron emission tomography/computed tomography (FDG PET/CT) was recommended with the suspicion of EMP. The imaging revealed multiple bony metastases, with increased metabolic activity involving the sternum, left transverse process of T9, the vertebral body of L5, and the left iliac spine. There was also a 4 cm mass of hypermetabolic activity at the inferior edge of the liver, which was indistinguishable from the gallbladder (Figure 1A). Magnetic resonance imaging (MRI) of the liver with gadolinium contrast confirmed a 7 cm mass located at the gallbladder fossa (Figure 2). An ultrasound of the liver further demonstrated a hypoechoic mass at the gallbladder fossa that was most consistent with a complete filling of the gallbladder lumen with an obstructive soft tissue mass that may reflect gallbladder malignancy (Figure 3). A hepatobiliary iminodiacetic acid (HIDA) scan could not visualize the gallbladder (Figure 4).

(A) Initial positron emission tomography/computed tomography (PET/CT) image demonstrating a mass at the gallbladder fossa with intense fluorodeoxyglucose (FDG) uptake. (B) Subsequent PET/CT image demonstrating an increase in size and metabolic activity of the gallbladder mass approximately 4 months later.
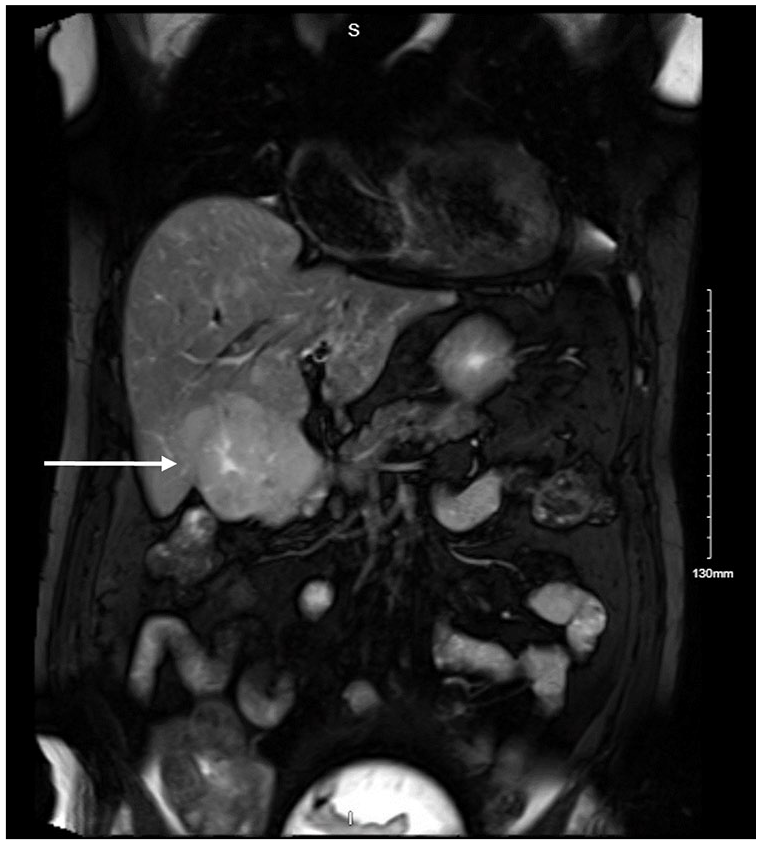
Magnetic resonance imaging (MRI) with gadolinium contrast demonstrating a 7-cm mass at the gallbladder fossa (white arrow).

An ultrasound of the liver demonstrating a hypoechoic mass at the gallbladder fossa that was most consistent with a complete filling of the gallbladder lumen secondary to an obstructive mass.
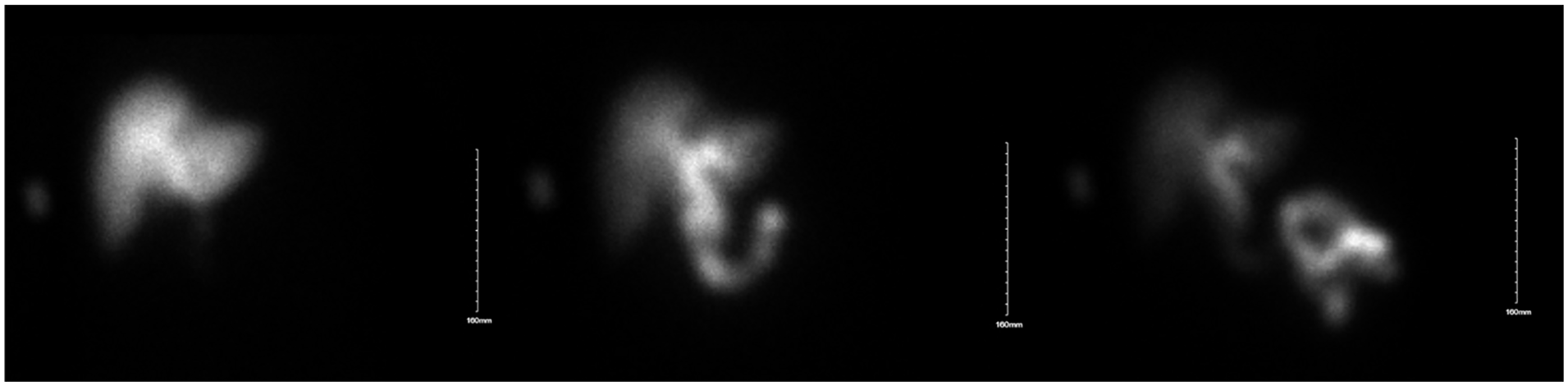
Hepatobiliary iminodiacetic acid (HIDA) scan demonstrating contrast emptying into the biliary duct system and into the duodenum without passing into the lumen of the gallbladder, indicating an obstruction of the cystic duct.
At this time, primary malignancy of the gallbladder was suspected. Interventional radiology felt a biopsy would be high risk for the development of biliary peritonitis. This risk was discussed with the patient, who ultimately chose to forego the procedure initially. At follow-up a few weeks later, the serum-free kappa light-chain value continued to increase to >11 000 mg/dL. At this time, the patient was presented with the option of a CT-guided biopsy for the second time. He was counseled regarding the risks, benefits, and alternative management options and he ultimately chose to pursue the procedure. Histopathologic evaluation of the CT-guided biopsy sample demonstrated diffuse proliferation of atypical plasma cells with no evidence of liver parenchyma (Figure 5A). Immunohistochemical analysis revealed positivity for MUM-1, CD56, Kappa in-situ hybridization, and CD138. CD3 was highlighted in a few scattered T cells (Figure 5B).

(A) Hematoxylin and eosin (H&E) staining demonstrating atypical plasma cells with an abundant cytoplasm and eccentric nuclei replacing the normal liver parenchyma. (B) Immunohistochemical (IHC) staining demonstrating plasma cells with positivity for CD138.
Based on the histological results from the biopsy, a diagnosis of EMM of the gallbladder was confirmed. On follow-up, the serum-free kappa light-chain value had increased again to 16 000 mg/dL, and creatinine had increased to 4 mg/dL. Given the rapid progression of light chains, a chemotherapeutic regimen consisting of bortezomib, dexamethasone, cyclophosphamide, and etoposide (V-DCE) was initiated. Cisplatin was not included in this therapy due to the compromised kidney function. The patient was admitted to the hospital and had a complicated hospital course following the initiation of chemotherapy with prolonged pancytopenia, gastrointestinal bleeding, respiratory failure, sepsis, encephalopathy, and acute kidney injury requiring hemodialysis. Computed tomography of the abdomen and serum-free light chains indicated an initial response 2 weeks post-induction, however, repeat 18-FDG PET/CT 6 weeks later demonstrated disease progression with increased size and hypermetabolic activity. There was also new metabolic activity within a new left perinephric mass and the posterior left ilium (Figure 1B).
On follow-up, the serum-free kappa light chains continued to elevate to >25 000 mg/dL. The patient noted new-onset, rapidly evolving varying sized, violaceous papules and nodules on chest and abdomen (Figure 6). These lesions were firm and non-tender with a smooth surface. Punch biopsy of the skin lesions established the presence of kappa-restricted plasma cells. Given the apparent resistance of his disease to V-DCE, he was counseled regarding the poor prognosis. Despite all treatments, the patient succumbed to the disease 2 1/2 years after the initial diagnosis of MM and 6 months from the diagnosis of EMM.

Photograph of the extensive cutaneous lesions manifesting as firm, smooth, violaceous papules and nodules.
Discussion
Multiple myeloma is an uncommon disease of clonal plasma cell proliferation with end organ damage. 1 Multiple myeloma can manifest as its own entity, with EMM involvement, or solitary plasmacytoma (SP). Extramedullary myeloma may be primary, which is observed in 68% of patients at the time of MM diagnosis, or secondary, which is observed in 10% to 13% of these patients. 15 The true incidence of EMM, however, may be underestimated due to the lack of prospective studies in this area. Solitary plasmacytoma is a discrete clinical entity from EMM, which is defined as a clonal proliferation of plasma cells outside the bone marrow with no concurrent diagnosis of myeloma. 16 It is important to differentiate between EMM and SP in the clinical setting because prognosis and treatment differ. In this case, the patient had a preexisting diagnosis of MM, which eliminated SP from the differential.
The pathogenesis of EMM is thought to be derived from the escape of the malignant myeloma cells from the bone marrow. 1 The interaction between these cells and the bone marrow microenvironment activates signaling cascades that mediate chemotaxis and adhesion of the myeloma cells to the bone marrow. 1 Extramedullary dissemination of the myeloma cells occurs through a variety of proposed mechanisms, such as decreased expression of adhesion molecules, the downregulation of tetraspanins and chemokine receptors, increased heparinase-1 expression, angiogenesis secondary to vascular endothelial growth factor (VEGF) secretion, and mutations in the NF-kB pathway. 2 Small cohort studies have established a 13q deletion, t(4;14), and a (17p) deletion as the most common cytogenetic abnormalities associated with the development of EMM. 17 Sheth et al 17 also discovered that p53 nuclear expression paired with no CD56 expression and an increased proliferative index was associated with the progression of disease from intramedullary to extramedullary sites in patients with MM. 18
Extramedullary myeloma involvement of gallbladder is rare. Our literature search retrieved 8 cases of EMM in gallbladder.4-11 Extramedullary myeloma of gallbladder can present with right upper quadrant pain, nausea, jaundice, cholecystitis, or pancreatitis. Cutaneous involvement can occur in less than 5% of EMM patients. Skin lesions can range from macules, papules, and nodules with a smooth surface and violaceous appearance. 19 In this case, the patient developed right upper quadrant abdominal pain and nausea, which resolved with NSAIDs. An ultrasound and HIDA scan confirmed the presence of a mass-like structure, which prevented filling of the gallbladder. This occurrence coincided with a continuous increase in serum-kappa light chains in spite of a normal bone marrow biopsy. Later on, the patient developed several violaceous lesions on the torso and abdomen, which corresponded to the dermal manifestations of EMM on both gross examination and histopathologic evaluation.
Treatment of EMM can be challenging as most of the recommendations are derived from retrospective studies. Alsouqi et al. 20 performed a retrospective, multicenter study, which evaluated the toxicities, outcomes, and potential use of a regimen consisting of carfilzomib, dexamethasone, thalidomide, cisplatin, doxorubicin, cyclophosphamide, and etoposide (KD-PACE) as bridging therapy to autologous or allogenic hematopoietic SCT or clinical trial for patients with relapsed or refractory MM. Patients in this study demonstrated a superior progression-free survival (8.3 months vs 2.3 months in the non-bridged group) and overall survival (median, 16.7 months vs 4.3 months in the non-bridged group). 20 Neutropenia (87%), leukopenia (96%), thrombocytopenia (90%), and anemia (90%) were the most common toxicities observed in patients on the KD-PACE regimen. 20 The patient in this case was treated with several different chemotherapeutic regimens over the course of his diagnosis. Upon arriving to our clinic in the United States, the patient began a regimen of DRD that led to the resolution of his condition based on the histologic analysis of a bone marrow specimen. Serum-kappa light chains began increasing again and a hypermetabolic mass was discovered in the gallbladder on PET/CT scan. The patient was then switched to a regimen of V-DCE before he succumbed to his condition.
Conclusion
In conclusion, we describe a case of an older adult male who developed secondary EMM of the gallbladder and cutaneous manifestations of the disease. We aim to contribute to the medical literature by expanding the pool of information regarding EMM of the gallbladder. Extramedullary myeloma should be suspected in those patients with a previous or existing diagnosis of MM who develop new-onset symptoms that coincide with an abrupt increase in serum-kappa light chains. Treatment guidelines are currently primarily supported by a culmination of retrospective or cohort studies. Future large-scale prospective studies should be pursued to further guide treatment recommendations, if possible.
Footnotes
Authors’ Note
Prior presentation of abstract statement: This abstract has not been presented at a meeting prior to submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
