Abstract
Hepatic angiomyolipoma (HAML) is an uncommon primary liver tumor with limited reported cases in the literature. It is composed of varying amounts of fat, smooth muscle, and blood vessels, typically non-malignant proliferation. It usually presents diagnostic challenges due to diverse imaging characteristics. We present a case of a 50-year-old female with a small HAML causing mild abdominal pain. It displayed radiographic characteristics mimicking hepatocellular carcinoma (HCC), and subsequent diagnosis was established upon histopathological findings of liver biopsy. The patient was radiologically monitored, and then she underwent surgical resection later on because of the size increase. Follow-up showed no signs of recurrence at 3 months. This case highlights the importance of histologic diagnosis of HAML if there are suggestive imaging features, given the variety of differential diagnoses. Symptoms or tumor growth is to be considered as warning signs that should lead to surgical resection, which remains the primary treatment modality.
Introduction
Angiomyolipoma is a rare, usually benign, subtype of a mesenchymal neoplasm of the perivascular epithelioid cell (PEC) tumor group, also known as PEComas, according to the World Health Organization (WHO) classification. 1 It is frequently observed in the kidneys; however, hepatic angiomyolipoma (HAML) is a rare entity, first reported since around 50 years ago, with only approximately 600 cases documented in the literature to-date. 2
The HAML is predominantly described in adult females, often incidentally discovered on imaging. It is most commonly sporadic; however, it is associated with tuberous sclerosing complex in only 5% to 10% of cases, unlike renal angiomyolipoma (>50%). 2 This tumor is typically a mixture of 3 components in various amounts: smooth muscle, adipose tissue, and blood vessels. 3 Radiological features are thus also variable and not specific, potentially mimicking a myriad of differential entities. Therefore, diagnosis relies mainly on histopathological findings. Besides, no standard management guidelines exist regarding HAML.
In this report, we present a case of HAML, pointing to the challenging imaging-based initial diagnosis and emphasizing the role of pathology. We also highlight the progressive potential of the tumor and the place for surgery. With that in mind, we conducted a comprehensive literature review to present current scientific knowledge in relation to this rare tumor.
Case Presentation
A 50-year-old non-smoker Caucasian woman, with no medical history, presented with chronic mild abdominal pain in the upper right quadrant that started a year ago. Physical examination showed no abnormalities. Laboratory findings were normal, notably liver function tests and blood count. Abdominal ultrasound (US) showed a non-dysmorphic liver with a homogeneous hyperechoic nodule of 15 mm in the segment VI. Further investigation with hepatic magnetic resonance imaging (MRI) revealed a well-circumscribed nodule in segment VI characterized by moderate T2 hyperintensity, T1 hypointensity, and diffusion hyperintensity with apparent diffusion coefficient (ADC) restriction, measuring 17 mm. The nodule demonstrated enhancement during the arterial phase, followed by washout in the portal and delayed phases. In addition, there was capsule enhancement in both the portal and delayed phases (Figure 1).

Liver magnetic resonance imaging revealing a well-circumscribed nodule in segment 6 (white arrow), measuring 36 mm in diameter. The lesion is moderately hyperintense on the T2 sequence (A) and hypointense on the T1 sequence (B). It displays hyperintensity on the T1-weighted Dixon fat sequence (C) with a signal drop on the T1-weighted opposed-phase (D), highlighting the fat component of the lesion. The nodule also demonstrates arterial enhancement after gadolinium injection (E) and shows washout in the portal phase with an enhanced capsule (F).
This radiological presentation was consistent with a hypervascular steatotic nodule; therefore, fat-containing hepatocellular carcinoma (HCC) and hepatocellular adenoma were the main considered diagnoses. Alpha fetoprotein (AFP) level was normal, and viral hepatitis B and C were negative. However, HCC was deemed less likely given that the patient had no signs of portal hypertension or hepatocellular dysfunction. Subsequently, a liver biopsy was performed. Histopathological findings revealed a heterogeneous proliferation of 3 distinct components: predominantly adipocytic, a network of small blood vessels varying in abundance, and epithelioid or rhabdoid cells. On immunohistochemical analysis, the cells were positive for vimentin and showed focal positivity for HMB-45. The CD34 staining highlighted a rich vascular network. The cells were negative for anti-hepatocyte, desmin, S100, cytokeratin (CK), CK7, CK20, and CK19 antibodies. The Ki67 proliferation index was less than 10%. There was no sign of malignancy as well.
These findings suggested a mesenchymal neoplasm, most likely an angiomyolipoma. Given the benign characteristic of the tumor and that the patient was asymptomatic at the time, the medical decision was clinical and radiological surveillance every 3 months.
However, the patient was then lost to follow-up for 12 months and then presented with a recurrence of right upper quadrant pain. A new MRI showed an increase in the size of the nodule, reaching 36 mm. Taking into account the presence of symptoms as well as the increase in size of the nodule, multidisciplinary team meeting recommended that patient undergo surgery. She had a laparotomy with hepatic wedge resection removing the nodule. Macroscopic examination of the surgical specimen revealed whitish yellow friable fragments of soft consistency (Figure 2). On microscopy, the coexistence of smooth muscle, mature adipose tissue, and abundant blood vessel proliferation was identified. The smooth muscle contingent was predominant (80%) with variable morphology: epithelioid, fusiform, or pleomorphic cells. These cells showed ample eosinophilic cytoplasm as well as large nucleolated nuclei and focally marked cytologic atypia (Figure 3). Immunohistochemistry staining exhibited positive Melan-A and HMB-45 and negative CD 117. Mitotic activity was assessed at 3 mitoses per 50 high-power field (HPF), with no detected necrosis. Overall, histological findings were consistent with a pleomorphic myomatous HAML.
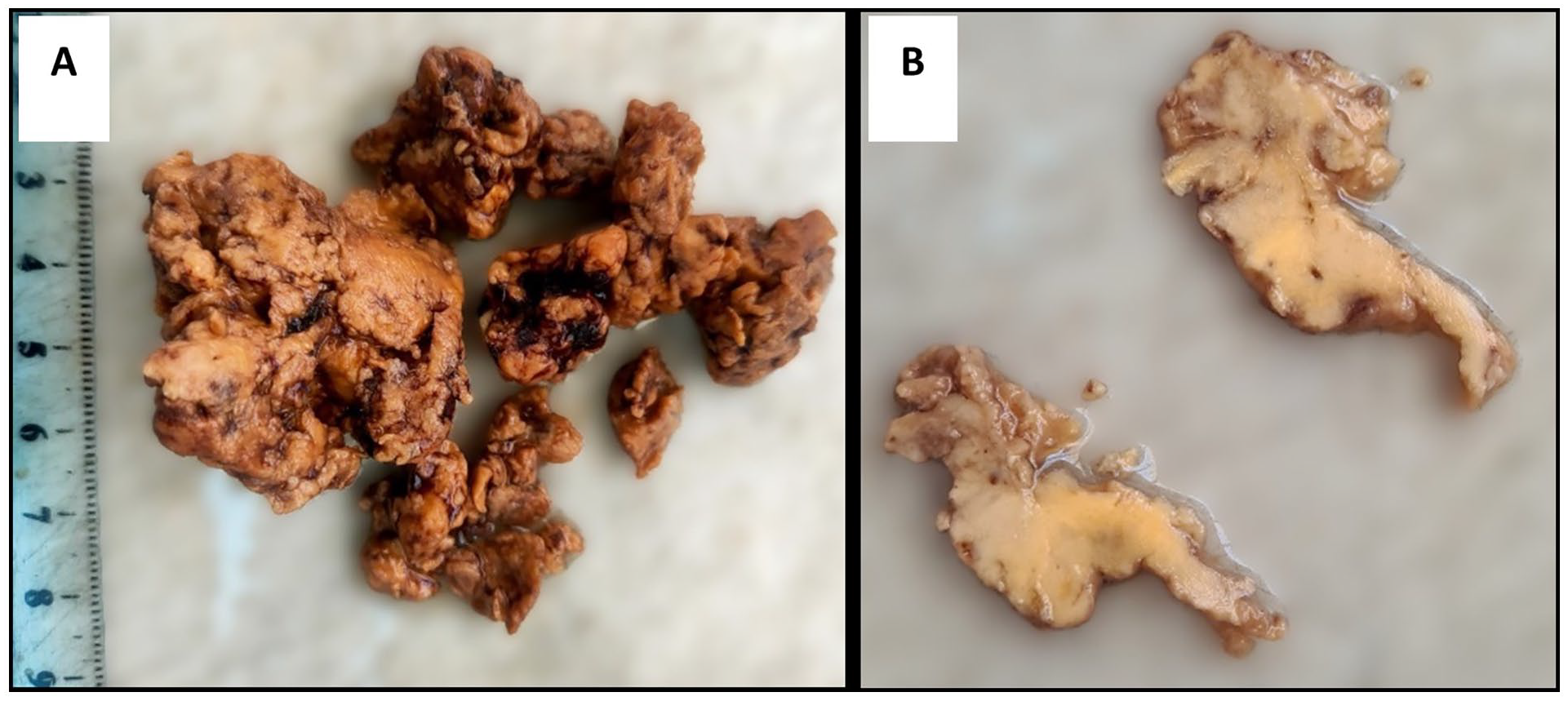
Surgical resection specimen, showing a friable and fragmented soft tissue (A) with yellowish color on cross-sectional examination (B).

Histopathological findings and immunohistochemistry staining of hepatic resection specimen. (A) Hematoxylin and eosin (H&E) findings on 100× magnification showing proliferation of different components: blood vessels (*) varying in size, mature adipocytes (#), and smooth muscle (§). (B) H&E on 200× magnification demonstrating prominent smooth muscle contingent, with variable morphology: fusiform (black arrows), epithelioid (white arrows), and pleomorphic cells. These cells displayed abundant eosinophilic cytoplasm as well as large nucleolated nuclei. On immunohistochemistry, compared with adjacent normal liver, the tumor proliferation exhibited positive staining with Melan-A (C) and HMB-45 (D).
A severe bilateral pulmonary embolism complicated the postoperative course at 3 weeks, requiring emergency fibrinolysis and a 4-day intensive care stay, with favorable subsequent outcomes. During a 3-month follow-up, no tumor recurrence was detected.
Discussion
The HAML is a rare primary tumor of the liver that has been reported in approximately 600 cases, including 105 in the past decade.2,4 Liver is though considered the second site affected by this tumor after the kidneys. 2 Similar to our case, it predominantly affects female patients in 74% to 84% of cases, with a median age of 56 years old.5,6 The HAML is solitary in 95% of cases, located mostly in the right lobe (64%) followed by the left lobe (30%) and rarely in the caudate lobe (6%), 3 with a median size ranging from 2 cm to 12.7 cm.5,6 Our patient presented with episodic abdominal pain, diverging from typical presentations where it is usually an incidental finding on imaging, with possible symptoms in only less than half of the cases, like abdominal discomfort, pain, bloating, nausea, anorexia, or weight loss.2,5 Routine laboratory results, including liver function tests, as well as AFP levels, are usually normal, also in our case. Nonetheless, acute presentation as severe abdominal pain or hemorrhagic shock due to spontaneous rupture of HAML has been reported in a few cases (around 12 cases), mainly in tumors measuring more than 5 cm.2,7,8
Owing to the varying proportions of fat, smooth muscle, and vascular components, the radiographic features of HAML can be quite diverse, rendering diagnosis based on imaging modalities challenging. The lesion is usually well circumscribed. On US, it is often hyperechogenic and sometimes heterogeneous with hypoechoic spots due to fat content. The HAML manifests as a heterogeneous mass on cross-sectional imaging, hypodense in computed tomography (CT), and hyperintense on MRI like as fatty tissue on both T1 and T2 with loss of signal on fat-suppressed sequences, hence making MRI the best modality for detecting fat component. 9 The tumor also displays arterial enhancement owing to its hypervascular characteristic, with common washout in the portal and delayed phases. In a series of 23 resection cases, Jung et al 10 postulated that lesions rich in central vessels on histology were more likely to exhibit washout in venous phases, whereas those with few or no vessels showed persistent enhancement. In addition, the lesion presents hypointensity in the hepatobiliary phase when using a hepatocyte-specific agent, 9 evidently due to the absence of hepatocytes within the tumor.
Considering these radiographic features, potentially overlapping with other entities, several differential diagnoses are to be considered such as HCC, hepatocellular adenoma, metastases from gastrointestinal stromal tumors, and focal steatosis. In fact, the preoperative diagnosis of HAML based on imaging was correct in only less than 30% in previous studies.3,5,10,11 Among differential diagnosis, HAML is most commonly misdiagnosed as HCC given the frequent hypervascularity, the washout, and the possibility of fat-containing HCC. Likewise, our case exemplifies the diagnostic difficulty relying solely on radiological results that showed a hypervascular lesion with washout in the delayed phase, raising suspicion mainly of HCC. A pseudocapsule was also observed in our case, although it has been rarely reported in 3% to 20%.11,12 Despite the lack of specific radiological discriminative signs, the absence of a pseudocapsule and the presence of intratumoral vessel connected to a draining hepatic vein are useful features that can help distinguish HAML from HCC.9,12
Therefore, histopathologic assessment is key for establishing diagnosis. Unlike HCC, HAML typically develops in non-cirrhotic liver, presenting on gross examination as well-circumscribed non-encapsulated (or partially encapsulated) yellow soft mass with possible red-brown patches.2,3 In our case, the resected specimen was also soft tissue and of yellow color, yet friable and fragmented, probably due to the extensive network of thin-walled blood vessels. The HAML can be classified according to the relative proportions of the 3 tissue types, namely: mixed or conventional type (> 20% of each of the 3 components), lipomatous (> 80% fat), myomatous (> 80% smooth muscle) like in this case, or angiomatous (> 80% blood vessels). 3 The mixed and the myomatous types are the most common, observed in 36.4% and 42.4% in a series of 151 cases. 5 An inflammatory subtype, characterized by inflammatory infiltrate surpassing 50% in proportion, has more recently been reported in 15 cases to-date. 13 Moreover, extramedullary hematopoiesis foci can also be observed in 33% to 64% of cases. 3
The smooth muscle cells are usually predominant and also the most important for histologic diagnosis. They can be epithelioid, spindled, intermediate, or pleomorphic. Immunochemistry analysis reveals positive staining for melanocytic markers, with HMB-45 and Melan-A being the most sensitive, as well as for smooth muscle markers, such as actin and vimentin.2,3,14 Conversely, the absence of actin staining does not exclude the diagnosis, and HMB-45 is considered the most specific marker found in 96% of cases. 14 Classically, these tumors do not exhibit epithelial markers, such as CK, nor do they express S100 protein or alpha-fetoprotein. 2
However, histologic diagnosis of HAML can sometimes be also challenging. Given their uniform expression of melanocytic markers, they might be confused with melanoma, but this latter typically expresses diffusely S100 protein compared with possible weak staining in 44% of HAML cases 14 and does not stain with smooth muscle actin. Besides, gastrointestinal stromal tumors can sometimes be considered due to the presence of fusiform cells and the occasional expression of KIT (CD 117) by HAML. Yet, the presence of adipocytic contingent and the low mitotic rate could help distinguish HAML from these differential diagnoses. 2 In our study, pathology findings were consistent with a typical HAML profile showing a myomatous subtype with positive Melan-A and HMB-45, and no staining with CD 117 or S100, making histologic diagnosis less problematic.
Although almost all HAML are benign, 1 malignant behavior has been described in rare instances. 2 According to the WHO, marked nuclear atypia along with high mitotic rate and diffuse pleomorphism are predictors of malignant potential of PEComas. 1 However, histological atypia is not considered a reliable criterion for malignancy in HAML, 1 with marked atypia seen in nearly 25%, 3 plus there is no consensus definition of invasive features for these tumors. Still, a mitotic activity of more than 1 per 50 HPF has been suggested as an invasive feature. 2 In a systematic review 5 published in 2017, including 292 patients with HAML, malignant behavior was observed in 4.1%, defined according to the authors as progression, recurrence, or tumor-related death. Besides, the epithelioid subtype is believed to be associated with a higher risk of aggressive behavior.2,5
Given the lack of standard of care for the management of HAML, practices diverge from conservative treatment to surgery. Klompenhouwer et al 6 suggested biannual surveillance for histologically confirmed HAML with no suspected malignant patterns. Surgical resection is otherwise advocated, as well as in case of symptoms or invasive growth. Initially, we opted for a wait-and-watch approach for our patient, with regular radiological monitoring. That strategy seemed reasonable considering the small size of the nodule, with histologically confirmed diagnosis of HAML and the absence of symptoms at the time. As a matter of fact, progression was observed in 6 of 29 patients (20.1%) with biopsy-proven HAML who underwent conservative treatment, as reported by Klompenhouwer et al, 5 showing an estimated cumulative growth rate of 0.77 cm per year. In our case, the nodule increased faster at a rate of 1 cm per year, thus raising concern about an aggressive behavior, which mainly drove surgery decision. This decision was further advocated by the detrimental features demonstrated by the tumor in our case, aside from the rapid growth rate, in particular the high mitotic index (>1 mitosis/50 HPF) and the considerable epithelioid contingent. However, our observation still raises the question of whether resection should have been opted for earlier based only on the presence of occasional mild symptoms.
In the aforementioned systematic review, 5 248 patients (84.2%) underwent surgical resection but only 70 had a confirmed preoperative diagnosis of HAML. The authors reported postoperative complications in 2.2%, death in 0.9%, and post-resection recurrence in 2.6%. A more recent single-center retrospective observational study of 40 resected HAML cases, including 9 laparoscopy interventions, indicated no major surgical complications, with negative margins in all cases and no tumor recurrence in a 76-month median follow-up. 15 Besides, in the same series, 5 patients with confirmed HAML were followed-up from 1 to 7 years but ended up being resected due to the tumor’s progressive growth. In another retrospective study including 92 cases of HAML, 68 patients (74%) underwent surgery with 6.2% reported rate of postoperative complications, such as low serum albumin, pleural effusion, pulmonary infection, short-term intestinal obstruction, and bile leakage; all classified as non-severe, 11 in contrast to our patient, who experienced a severe thromboembolic complication. Two cases (2.9%) of tumor recurrence following radical hepatectomy were also recorded after 4 and 4.5 years. The short follow-up duration in our study limits the ability to draw conclusions regarding recurrence.
Other therapeutic alternatives, mainly for cases unsuitable for surgery, comprise radiofrequency ablation (RFA), transarterial chemoembolization (TACE), transcatheter arterial embolization (TAE), and targeted therapy. The latter comprises the use of sirolimus, an antiproliferative drug that has proven its efficacy in size reduction of renal angiomyolipoma. 2 In fact, it has been administered in few cases of HAML (n = 4), with an 80% rate of tumor regression 5 ; yet, no conclusions could be drawn. The TAE has been used as an emergency treatment, and a bridge to surgery, in ruptured HAML. 7 However, RFA seems to be a promising and efficient therapeutic option. It has been applied in 22 patients, all with tumor size less than 5 cm, with no reported recurrence. 11
Conclusion
Our case depicts the clinical, radiological, and histological features of HAML, along with its progressive course and therapeutic aspects. It contributes to the enrichment of limited actual knowledge regarding this rare entity. In fact, HAML is an uncommon primary hepatic mesenchymal tumor, usually asymptomatic, with a challenging diagnosis on imaging modalities due to its variable characteristics. Although it can sometimes be suggested based on radiographic findings, histopathological confirmation seems mandatory, especially when confronted with differential diagnoses, namely HCC, and when a conservatory strategy is proposed. Despite being usually benign, an aggressive potential has been increasingly recognized, prompting radical surgical resection, which remains the cornerstone of treatment in HAML. Subsequently, regular follow-up should be ensured even though recurrence is uncommon.
Footnotes
Authors’ Note
This manuscript has not been previously presented at a meeting or published elsewhere or under consideration by any other journal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for her anonymized information to be published in this article.
