Abstract
Lung cancer, a leading cause of cancer mortality, often involves epidermal growth factor receptor (EGFR) mutations, common in 17% of Caucasian and 40% of Asian non-small-cell lung cancer (NSCLC) patients. While the exon 19 deletion and L858R mutation are prevalent, rare variants like L833V/H835L are less understood. This case reports a 75-year-old female with NSCLC harboring L833V/H835L mutations. Initial imaging showed a right upper lobe mass and nodularity in the left upper lobe. Biopsy confirmed adenocarcinoma, and genomic analysis identified EGFR L833V/H835L mutations. Based on these findings, the patient was treated with osimertinib 160 mg daily, reduced to 80 mg due to side effects. After 3 months, positron emission tomography (PET) scans revealed significant tumor reduction, and brain metastasis remained stable. This case demonstrates the efficacy of osimertinib for rare EGFR mutations, aligning with literature suggesting its potential for managing such variants. Although large-scale trials are impractical due to the rarity of these mutations, this report adds valuable evidence supporting osimertinib’s use, highlighting the need for comprehensive genomic profiling in NSCLC.
Introduction
Lung cancer is among the most common cancers and is the leading cause of death related to cancer. Mutations in the epidermal growth factor receptor (EGFR) gene play a crucial role in the development of non-small-cell lung cancer (NSCLC), with an occurrence rate of approximately 17% among Caucasian and 40% among Asian patients.1,2 The exon 19 deletion and the exon 21 L858R mutation are the most frequent EGFR mutations found in lung cancer, together constituting about 90% of all EGFR mutations. 3 Numerous large-scale Phase III clinical trials have established that NSCLC patients with these typical mutations respond effectively to EGFR tyrosine kinase inhibitors (TKIs), which are typically administered as the first-line therapy.4,5
In contrast, there is relatively limited information on rare variants, which represent a diverse array of genomic alterations. 6 The increasing use of next-generation sequencing (NGS) has led to the detection of more rare variants. They may be categorized into relatively rare variants, such as G719X and L861Q, and exceptionally rare variants, including L833V/H835L and other compound mutations. 7
We report a case of a patient with NSCLC carrying a complex EGFR L833V/H835L mutation, which responded clinically to osimertinib treatment.
Case Presentation
A 75-year-old white female presented to the clinic with 6 weeks of nonproductive cough, unresponsive to supportive management. Chest X-ray in 7/2023 showed a mass-like area in the medial aspect of the right upper lobe with some nodularity in the left upper lobe (Figure 1). Follow-up computed tomography (CT) scan of the chest with intravenous (IV) contrast showed a right lower lobe mass measuring 1.7 cm × 4.1 cm occluding the right upper lobe bronchus with right hilar adenopathy (Figure 2). Positron emission tomography (PET) scan confirmed hypermetabolic activity within the right upper lobe, consistent with an underlying mass. There were areas of hypermetabolic activity in the left lung as well. EBUS/bronchoscopy with biopsy of the left upper and right upper lobes was consistent with non-small-cell carcinoma, adenocarcinoma (Figure 3). Genomic findings: EGFR, L833V, H835L, CDK4 amplification, and MDM2 amplification. The tumor cells stained positive for cytokeratin 7 and TTF-1 (Figure 4). Magnetic resonance imaging (MRI) brain with and without contrast revealed metastasis to the right parietal lobe (Figure 5). Radiation oncology recommended systemic treatment for the lung and brain lesions, as the brain lesion would likely respond well to systemic therapy. The patient was started on an EGFR inhibitor, osimertinib 160 mg daily until diarrhea and rash became an issue, at which time the dose was decreased to 80 mg daily for 1 week. Repeat PET scan after 3 months of therapy showed a positive response to therapy. The right upper lobe lesion had decreased in size and the right hilar lymphadenopathy had resolved completely. Repeat chest X-ray in 5/2024 also showed improvement of the right upper lobe lesion (Figure 6). Repeat brain MRIs × 2 showed no change in the size of the brain lesion. Ultimately, the patient was diagnosed with adenocarcinoma of the lung with metastasis to the contralateral lung and brain-T2bN1M1b-Stage IVc-PDL-1 50% and EGFR L833V H835L mutation. The EGFR L833V H835L mutation incidence is very rare in lung cancer; however, despite that, the patient exhibited a good treatment response to the EGFR inhibitor, osimertinib.

C-Xray from 7/2023 showing right upper lobe mass.

CT chest with IV contrast demonstrating a right lower lobe mass measuring 1.7 × 4.1 cm.
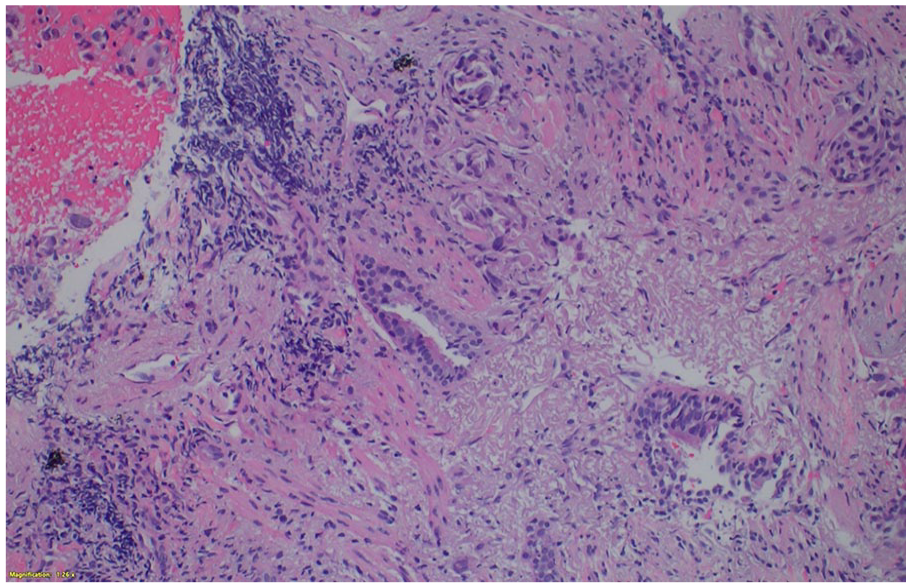
Histologic section of the lung mass biopsy ×200 demonstrates adenocarcinoma with an acinar growth pattern with significant background desmoplasia and necrosis.

Positive nuclear TTF-1 immunohistochemical stain expression confirms lung adenocarcinoma.

A 3-mm nodular enhancing focus in the right parietal lobe.

C-Xray from 5/2024 showing improvement of right upper lobe mass.
Discussion
In this case, we demonstrated the clinical effectiveness of Osimertinib in a patient with NSCLC carrying EGFR L833V/H835L mutations and accompanied by brain metastasis.
A compound EGFR variant involves 2 or more distinct mutations within the EGFR tyrosine kinase domain. Typically, this includes a combination of a common EGFR mutation and a rare variant. The occurrence of 2 or more rare EGFR mutations simultaneously in lung cancer is extremely rare. 7 A study comparing various EGFR detection methods identified only 3 cases of L833V/H835L mutations among 157 patients with NSCLC. 8 Not many studies have explored this mutation’s clinical significance. Based on published case reports, we have identified that the L833V/H835L variant appears to have a higher prevalence in Asian males despite our patient being White. In addition, there is no observed correlation between the presence of L833V/H835L mutations and the smoking history of the individuals. 9
Published reports suggest that a thorough assessment for EGFR mutations should be performed prior to initiating treatment in clinical practice. The use of NGS was crucial for identifying the L833V/H835L mutations in this patient, as traditional Amplification Refractory Mutation System polymerase chain reaction (PCR) methods typically do not detect such rare mutations. 10 Cao et al 11 demonstrated that NGS can effectively identify potential oncogenic driver mutations, guide mutation-targeted therapy decisions, and predict clinical outcomes in NSCLC patients. Previous studies have primarily detected L833V/H835L mutations in tumor tissue. One case suggested that liquid biopsy could be a viable substitute for mutation analysis as mutations were identified using circulating tumor DNA (ctDNA) from plasma and pleural effusion. It has been reported that ctDNA clearance during treatment serves as both a predictive and prognostic biomarker in advanced NSCLC. 12
A cohort study reported a patient with the L833V/H835L variant achieving over 15 months of progression-free survival (PFS) with gefitinib treatment 13 and another reported a case of L833V/H835L EGFR variant NSCLC and skin metastasis that showed nearly complete resolution of skin symptoms within 1 month of treatment with gefitinib. 9 On the contrary, a case of a patient with triple EGFR mutation (L833V/H835L/R670W) demonstrated resistance to gefitinib yet responsiveness to afatinib. This indicates that the R670W mutation might confer resistance to first-generation EGFR TKIs. 14 Li et al 15 found that aumolertinib could be a promising treatment option for patients with brain metastasis.
Luo et al reported an NSCLC patient with a complex EGFR L833V/H835L mutation in exon 21 who achieved complete response to first-line osimertinib monotherapy. No metastasis was detected, serum carcinoembryonic antigen levels returned to normal, and NGS monitoring of circulating tumor DNA mutations remained negative. Their case provided the first clinical evidence of the effectiveness of first-line osimertinib therapy in lung cancer patients with the rare L833V/H835L EGFR mutation. 16 In addition, Qin et al 14 reported a case with H833V/H835L/T790M triple mutation who responded favorably to osimertinib.
Our patient had stable yet persistent brain metastasis with osimertinib therapy. Adua et al explained this phenomenon, noting that despite widespread distribution of osimertinib in vivo, the brain microvascular tumor microenvironment (TME) allows malignant cell subpopulations to persist and eventually form osimertinib-resistant lesions. This persistence is regulated by the Ras homolog family member A (RhoA) and serum responsive factor (SRF) gene expression program, which promotes tumor cell growth in response to extracellular laminin, particularly in the brain. 17 This may necessitate further exploration of combination therapies. Jänne et al 18 reported that combining osimertinib with platinum-pemetrexed improves CNS efficacy and delays progression compared to osimertinib alone, supporting this combination as a first-line treatment for EGFR-mutated advanced NSCLC, including CNS metastases.
Because of the limited number of small-sample observational studies and case reports, conducting relevant clinical trials is nearly impossible. Consequently, there has not been a standardized treatment approach for patients with these extremely rare mutations.
Conclusion
This case emphasizes that EGFR L833V/H835L mutations, despite being uncommon, may respond well to osimertinib in NSCLC. Although designing a clinical trial is impractical due to the limited number of patients with these mutations, this case provides additional evidence supporting osimertinib’s clinical use. Further in vitro and in silico studies are needed to elucidate the precise mechanisms of osimertinib’s effectiveness against these mutations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
