Abstract
Papillary muscle rupture is commonly linked to ischemic events, occurring typically postmyocardial infarction. However, iatrogenic and nonischemic etiologies can play a role in papillary muscle rupture, including infective endocarditis. The case presented herein is an 85-year-old female with a history of heart failure with preserved ejection fraction (HFpEF), who presented with progressive dyspnea despite furosemide therapy, and was discovered to have a flail posterior mitral valve leaflet secondary to papillary muscle rupture associated with Streptococcus lutetiensis endocarditis. Transthoracic echocardiography revealed flail posterior mitral valve leaflet with newly appreciated severe wide-open mitral regurgitation. Emergent mitral valve repair was performed with a successful MitraClip placement. Patient had an atypical presentation without ischemic symptomology or electrocardiogram (EKG) changes indicative of ischemia. Despite transesophageal echocardiography, not revealing vegetations, infective endocarditis was presumed due to presence of Streptococcus lutetiensis bacteremia, osteomyelitis, elevated inflammatory markers, and clinical presentation. Papillary muscle rupture results in severe mitral regurgitation that is a medical emergency that requires prompt diagnosis and management from a multidisciplinary team. Therefore, it is imperative to ensure clinical insight into the multiple etiologies of papillary muscle rupture to provide timely treatment.
Introduction
Papillary muscle rupture represents a recognized complication linked to ischemic events, predominantly myocardial infarctions, manifesting in approximately 0.07-0.26% of patients postmyocardial infarction. 1 Infrequently, iatrogenic and nonischemic factors can precipitate papillary muscle rupture, with nonischemic etiologies encompassing Ehlers-Danlos syndrome, blunt chest trauma, myxomatous disease, spontaneous rupture, and rarely, infective endocarditis (IE). Unlike ischemic causes, nonischemic etiologies may preserve ejection fraction (EF) and ventricular function, resulting in a decreased burden of cardiogenic shock.2,3 Herein, we present a case involving an 85-year-old female with a history of heart failure with preserved ejection fraction (HFpEF), who presented with deteriorating dyspnea and was diagnosed with a flail posterior mitral valve leaflet secondary to papillary muscle rupture associated with Streptococcus lutetiensis endocarditis.
Case Presentation
An 85-year-old female with a medical history notable for heart failure with preserved ejection fraction (HFpEF) at LVEF 60-65%, 5% PVC burden, hypertension, hyperlipidemia, and moderate carotid artery stenosis, presented with progressive dyspnea over the past week, unresponsive to furosemide therapy. On presentation, she exhibited hypoxia necessitating supplemental oxygen via nasal cannula and hypotension with mean arterial pressures (MAPs) in the high 60s. Elevated troponin at 180 and B-type natriuretic peptide (BNP) of 663. Electrocardiogram (EKG) revealed sinus rhythm with premature ventricular contractions (PVCs) with no ischemic changes. Chest x-ray was completed on admission due to dyspnea, which illustrated bilateral haziness [Figure 1]. Prompting CT (computed tomography) scan of the chest, which showed bilateral pleural effusions alongside lower lobe atelectatic consolidations [Figure 2]. Despite initial intravenous furosemide administration and thoracentesis yielding 900 mL of transudative output, oxygenation did not significantly improve. Transthoracic echocardiogram (TTE) revealed an EF of 60% to 65% with a flail posterior mitral valve leaflet attributed to calcified chordae rupture, accompanied by severe wide-open mitral regurgitation and moderate left atrial enlargement [Figure 3], a deviation from a prior TTE 5 months earlier which depicted similar EF but with mildly thickened mitral valve leaflets and mild mitral regurgitation. Following cardiology consultation, initiation of a nitroprusside drip was advised for afterload reduction, prompting transfer to the intensive care unit (ICU) for closer monitoring.
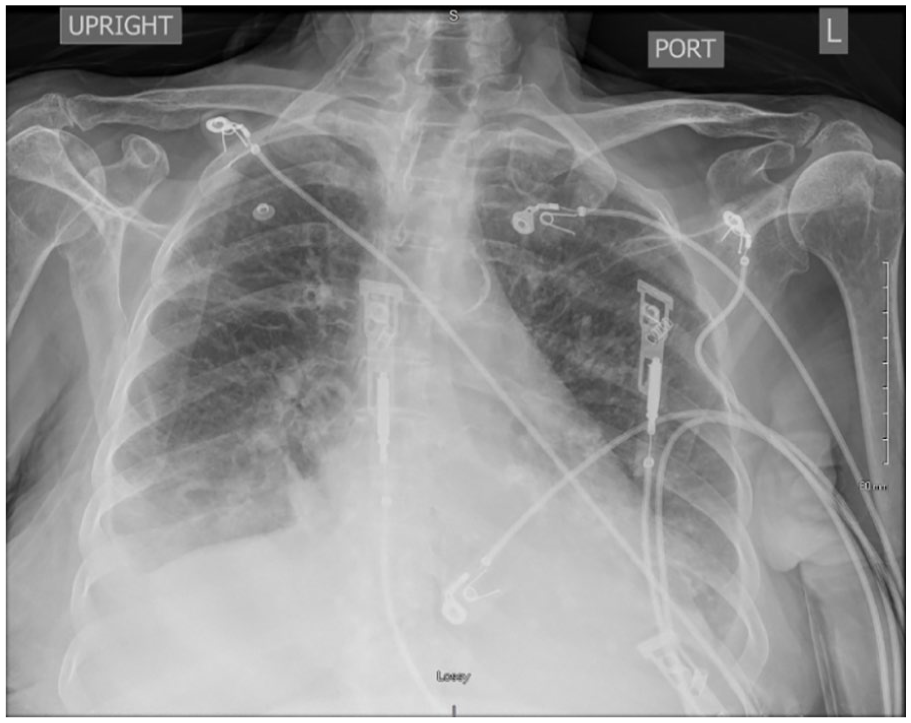
Chest X-ray of patient upon admission due to shortness of breath with decreased oxygen saturation. The image illustrates bilateral haziness in lower lung fields. This prompted further investigative imaging with a CT of the chest.

CT Chest with contrast illustrating bilateral pleural effusions with lower lobe atelectasis consolidations. Diuretics were started to decrease the pleural effusions, but due to the patient’s persistent symptoms and large consolidations a thoracentesis was performed. However, oxygen saturation and symptoms of dyspnea did not improve.

Transthoracic echocardiogram (TTE) was completed which revealed a preserved ejection fraction of 60% to 65% with flail posterior mitral valve leaflet, severe mitral valve regurgitation, and moderate left atrial enlargement. This differed from previous TTE 5 months prior. Red Arrow highlighting the flail mitral valve leaflet in the parasternal (Figure 3A) and apical 4 chamber (Figure 3B) views. Figure 3B illustrating the flow through the mitral valve with Doppler.
Subsequent right and left heart catheterization and coronary angiography unveiled subtotal occlusion of the mid-left anterior descending (LAD) artery without any further significant obstructive disease within the LAD, alongside low right-sided filling pressures, severe pulmonary artery hypertension, and elevated pulmonary capillary wedge pressures. Intravenous diuresis was withheld due to low right-sided pressures, while aspirin and rosuvastatin were introduced for secondary prevention of atherosclerosis. Due to the cardiac catheterization findings, cardiothoracic surgery considered the case for possible surgical mitral valve replacement and coronary artery bypass surgery with the left internal mammary artery (LIMA) to LAD; however, the patient’s high surgical risk status precluded immediate intervention. Elevated inflammatory markers prompted suspicion of endocarditis, supported by positive blood cultures for pan-susceptible S lutetiensis. Consequently, broad-spectrum vancomycin was transitioned to ceftriaxone. Transesophageal echocardiogram (TEE) delineated sclerotic mitral leaflets with a moderately calcified annulus, a ruptured lateral papillary muscle leading to flail posterior leaflet along P1-P2 scallops, and severe mitral regurgitation.
Further management involved gastroenterology recommendation for colonoscopy to assess for colonic malignancy given S lutetiensis bacteremia, pending stabilization of cardiopulmonary status. Abdominal and pelvic CT imaging ruled out malignancy but revealed severe L2-L3 disk disease suggestive of disk-osteomyelitis, reinforced on subsequent magnetic resonance imaging (MRI). Bilateral thoracentesis was performed to optimize respiratory function before mitral valve intervention. Successful transcatheter edge-to-edge valve repair via MitraClip placement yielded residual mild to moderate mitral valve regurgitation. The patient was slated to receive intravenous ceftriaxone as per Infectious Disease guidelines for 6 weeks to manage endocarditis and osteomyelitis concurrently.
Discussion
Prompt diagnosis and thorough assessment of mitral regurgitation etiology are crucial to avoid complications such as cardiogenic shock and fatal arrhythmias that can contribute to mortality as high as 75% within the first 24 hours. 1 When patients present with ruptured papillary muscle secondary to IE, clinical presentation should determine management. 2 This case underscores the severe sequelae of IE, leading to papillary muscle rupture and severe, acute mitral regurgitation. 4 Although transesophageal echocardiography (TEE) did not reveal vegetations, the presence of bacteremia with osteomyelitis of L1-L2 alongside acute mitral regurgitation establishes the diagnosis of possible IE. Interestingly, although vegetations were not evident on TEE, the burden of micro vegetations was significant enough to cause severe mitral regurgitation. Notably, the substantial burden of micro vegetations, although not visualized on TEE, likely played a pivotal role in inducing severe mitral regurgitation. TEE is 96% sensitive for native valves; however, may be false-negative due to multiple etiologies: embolization of vegetation, inadequate views, or, such as in this case, micro vegetations smaller than limits of resolution. In cases where TEE does not show evidence of IE and clinical suspicion remains high, clinical judgment should continue to guide management. 5
Despite catheterization noting subtotal occlusion of the LAD artery and potential ischemic contribution to the patient’s symptoms, it is less likely the primary etiology given the absence of features indicative of a large myocardial infarction, such as no ischemic symptoms or EKG changes indicative of ischemia. Alongside the preservation of EF and right ventricular function, which is more typical of a nonischemic etiology of papillary muscle rupture. Furthermore, the dual blood supply from the LAD artery and left circumflex artery to the anterolateral papillary muscle further suggests that ischemia was likely not a contributing factor to the patient’s presentation.
It is also essential to highlight the multidisciplinary care team involved in this patient’s medical care. Early surgical intervention of mitral valve repair remains the standard of care with increased improvement in mortality, particularly with those with acute heart failure secondary to acute mitral regurgitation.6-8 After surgical repair, culture-guided prolonged antibiotic therapy is necessary for eradication of infection, especially those with prosthetic valves placed.2,4 In cases such as the one presented above, the bacteria present may indicate underlying pathology. 25% to 80% of patients with S. lutetiensis (previously known as S. bovis) bacteremia have concomitant colorectal tumors, with the neoplasm arising even years after the presentation of bacteremia or IE. 9 Therefore, all those who present with bacteremia of S. lutetiensis should have a colonoscopy for surveillance of underlying colorectal neoplasia.
Conclusion
Papillary muscle rupture regardless of etiology is a medical emergency that requires prompt treatment. All papillary muscle rupture results in the development of acute severe mitral regurgitation in which treatment is always operative. The timing of surgical intervention and the treatment of underlying etiology of the papillary muscle rupture varies with pathology. This underlines the importance of maintaining a thorough clinical assessment and swift consultation of specialists.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
