Abstract
Cutaneous squamous cell carcinoma (cSCC) comprises 20% of cases of nonmelanoma skin cancers in the United States. In total, 3% to 5% of squamous cell carcinoma (SCC) are metastatic at the time of presentation, associated with significant mortality due to a lack of standardized treatment options. In total, 95% of these tumors are amenable to the initial standard of treatment, which is surgical resection. However, a small percentage of them require systemic therapy as they are either locally advanced to regional lymph nodes or have distant metastasis. The common sites of presentation of cSCC are the scalp and the face with predictable spread to the intra-parotid, upper jugular, and perifacial lymph nodes. In our case report, however, our patient had a large lump lesion on the upper back, an unusual site of presentation of cSCC, with locally advanced metastasis to the left axillary lymph nodes. Subsequently, the tumor marker study revealed a positive SMARCA4 variant (the essential ATPase subunit of the Switch (SWI)/Sucrose Nonfermenting (SNF) chromatin-remodeling complex) that is even rarer in the context of cSCC. Furthermore, abnormalities in SWI/SNF chromatin-remodeling complex subunits have shown promising results as a target therapy for immune checkpoint inhibitor (ICI) therapy. We present an atypical presentation site of locally advanced rare variant SMARCA4-positive cSCC in a patient who received treatment with chemoradiation and systemic therapy with ICI after primary surgical resection. To date, only 2 cases of SMARCA4-positive cSCC were found in the literature with no details of the treatment received. Our case is unique in its atypical site of presentation as well as showing partial response to radiotherapy (RT) and systemic therapy with ICI.
Keywords
Introduction
Cutaneous squamous cell carcinoma (cSCC) accounts for 20% of cutaneous malignancies and ranks as the second most common nonmelanoma skin cancer after basal cell carcinoma. 1 In total, 2% to 5% of cSCC have locally advanced tumors with metastases to regional lymph nodes. 2 The development and progression of cSCC involves mutations of genes, epigenetic modifications, viral infections, and tumor microenvironment factors that regulate the molecular etiopathogenesis of the tumor. 3 Pooled analysis studies have shown that genes responsible for chromatin remodeling and histone modifications demonstrated a higher burden of mutations in metastatic tumors as compared to nonmetastatic tumors of the skin. 4 Several significant genes and their mutations play an important role in the genesis of head and neck squamous cell carcinoma (SCC) including TP53, CDKN2A, HRAS, PTEN, and PIK3CA. 5 SMARCA4 subgroup of genes, belonging to the SWI1/SNF1 family of chromatin repair, has recently gained attention as a substantial oncogenic molecule that can be potentially targeted in cancer therapies. 6 SMARCA4-deficient undifferentiated malignant neoplasms (SD-UMN) are a known entity of aggressive type of lung carcinoma; 7 however, SMARCA4-positive genomic variant in cSCC is not well known and is considered as a very rare presentation. To date, only 2 cases of primary cutaneous SD-UMN have been reported. 8 With regard to SMARCA4-positive cSCC, we were able to find 2 case reports, namely, a case involving a 74-year-old Caucasian male with cSCC of the head and neck who was treated with pembrolizumab 9 and the other was a research article based on genomic variants of cSCC showing 1 sample positive for SMARCA4. 10
Case Report
Our patient is a 50-year-old male who presented with a mass on the left upper back that was increasing in size associated with fluid oozing and ulceration. The mass grew to its present size of approximately 10 cm × 8 cm more than 2 to 3 months. He had no significant past medical history or family history of cancer. He worked as a porter in a building, and the occupation did not involve exposure to sun or outdoor activities. He denied any smoking or use of substances. On examination, around 10 cm × 8 cm mass was noted on the left upper back with central ulceration and serosanguinous fluid, non–foul-smelling oozing out of it (Figure 1). The mass was mildly tender, restrictive mobility, and soft in consistency. Computed tomography thoracic spine was done and showed 7.5 cm × 9 cm × 4.2 cm heterogeneous well-defined superficial soft tissue mass in the left upper back with infiltration to the adipose tissue (Figure 2). He underwent an incisional biopsy of the mass on the back, and the tissue was sent for pathology. Per operative findings approximately 10 cm × 10 cm ulcerated mass on the back located on the left scapula area. Preliminary pathology reported SCC of skin. The patient was planned for wide local excision of the mass (Figure 3) that was done with a pathology report (Figures 4 and 5) showing invasive high-grade SCC, poorly differentiated with marked cytological atypia and foci of tumor necrosis. The invasive margin reached 1 mm of the deep inked margins. Ki-67 was 60% on Immunohistochemical stain. Additional testing for human papillomavirus (HPV) RNA (6/11, 16/18, 31/33) was negative. The patient had left axillary lymphadenopathy palpable clinically with approximately 3 lymph nodes that showed associated perinodal soft tissue invasion seen in 1 of the 2 lymph nodes with soft tissue inflammation after resection.

On examination, around 10 cm × 8 cm mass was noted on the left upper back, soft in consistency, mildly tender with central ulceration and serosanguinous fluid, non–foul-smelling oozing out of it.

CT thoracic spine axial view showing 9.3 cm lesion on upper back marked by the red arrow.
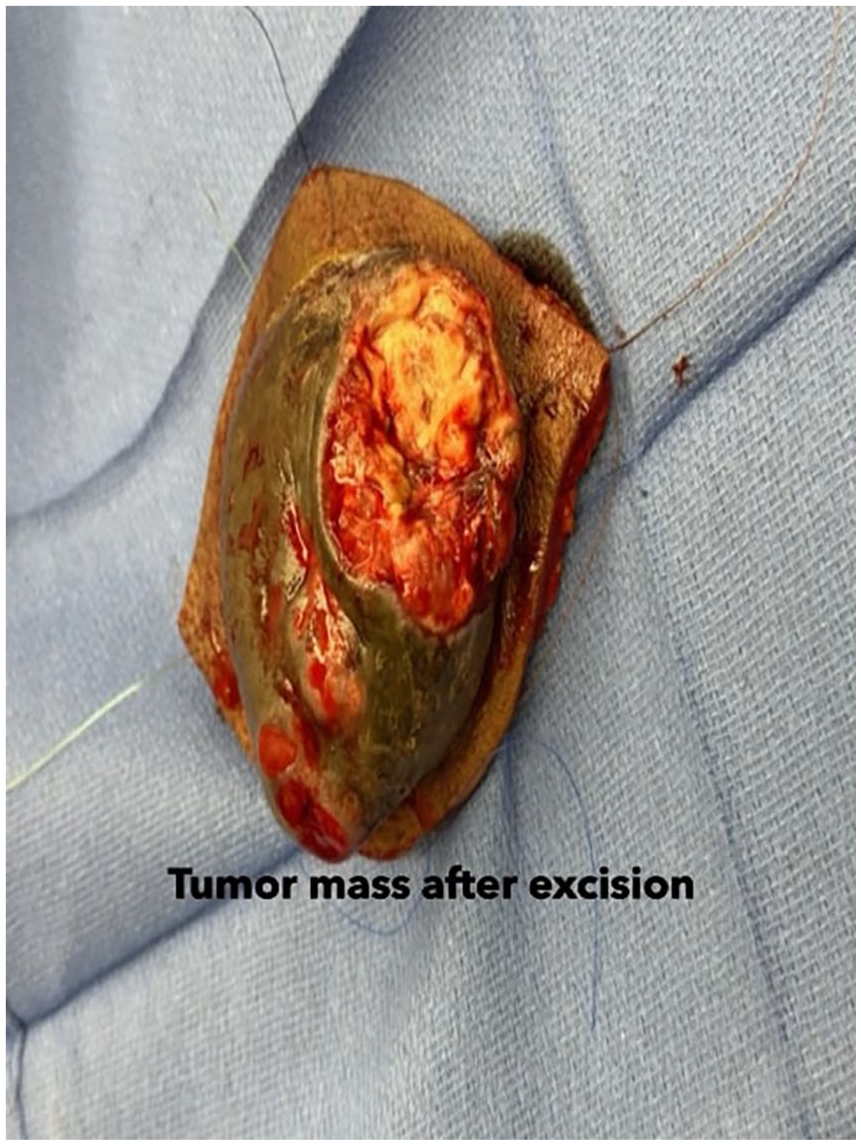
Wide local excision of the tumor mass on the upper back.
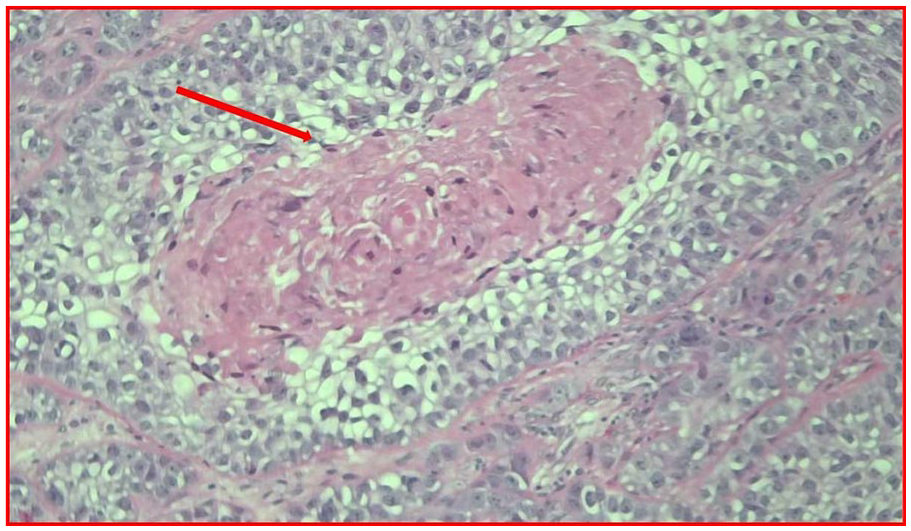
Showing 40× magnification area with dense pink cytoplasmic keratinization marked by the red arrow and H&E staining showing cells suggestive of features of SCC.
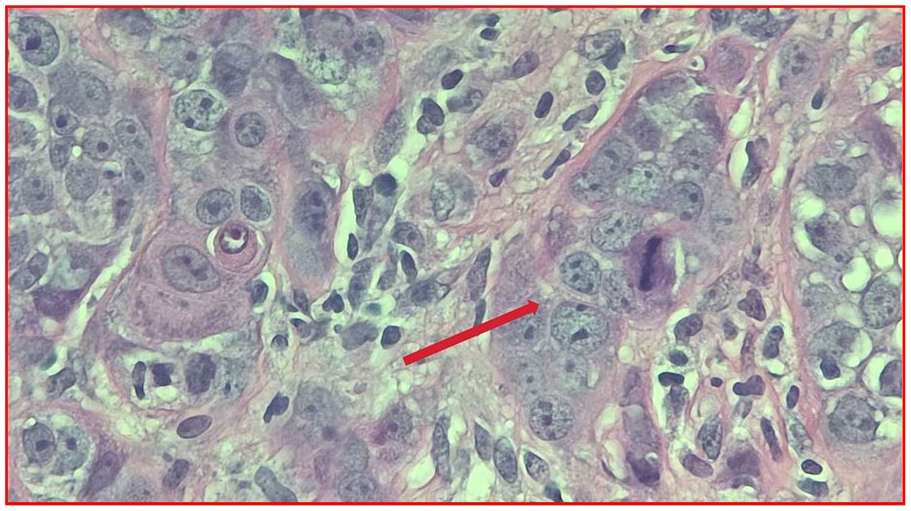
Showing high power field (HPF) 100× magnification of cells with features of invasive, high-grade, poorly differentiated with marked cytological atypia shown by the red arrow and foci of tumor necrosis of SCC.
Positive genomic variants noted on the tumor biopsy block using next-generation sequencing (NGS) were SMARCA4T910M, CDKN2A Y129fs, ERBB2 V777L, and TP53 C242fs. Positron emission tomography-computed tomography (PET-CT) scan done—hypermetabolic skin thickening in the left upper back (Figure 6A and B), hypermetabolic subcutaneous nodule/lymph node in the left posterolateral upper back with hypermetabolic left axillary lymphadenopathy noted (Figure 7), which is most consistent with metastatic nodal disease. No other distant metastasis was noted.

(A) Axial view PET scan showing heterogeneous hypermetabolic activity in skin thickening in the left upper back, marked by the red arrow, approximately 9.3 cm in length. The standard uptake values of this activity range from 3.5 to 4.4. (B) Sagittal view PET scan showing heterogeneous hypermetabolic activity in skin thickening in the left upper back, marked by the red arrow, approximately 9.3 cm in length. The standard uptake values of this activity range from 3.5 to 4.4.

Positron emission tomography (PET) scan image showing left axillary lymphadenopathy as marked by blue and red arrows with largest one approximately 1 cm that were resected and showed associated perinodal soft tissue invasion seen in 1 of the 2 lymph nodes.
The patient was admitted again for a planned procedure of split-thickness skin graft for the open wound on the left upper back. After healing of the wound, he was started on radiotherapy (RT) along with a radiation-sensitizing dose of cisplatin that continued for 8 weeks. He was regularly followed up in the clinic. During the last week of his treatment, he developed an acute onset of dysphagia with the inability to tolerate both solid and liquid food associated with severe retching, gag reflex, and vomiting which were likely adverse effects of chemo-RT. During his hospital stay, CT chest/abdomen/pelvis without contrast showed pleural-based pulmonary metastases that were questionable with scattered hypodense liver lesions the largest in the right hepatic lobe measuring 4 cm. Liver biopsy was done, and pathology was consistent with liver hemangioma and not metastasis. The pleural-based pulmonary findings were reviewed by the radiologist, and the decision was taken to observe them over a while with one of the differentials being RT-induced changes. Given the high risk of local progression of the disease, the patient started on systemic therapy with pembrolizumab.
After completion of RT and receiving 3 cycles of immune checkpoint inhibitor (ICI), the patient underwent his first surveillance PET scan (approximately 9 months after the diagnosis), which showed interval resolution of the hypermetabolic activity in the left axillary lymph nodes; however, a new hypermetabolic 2.2 cm nodule appeared in the skin and subcutaneous tissues inferior to the mildly hypermetabolic soft tissue changes in the left upper back with the standard uptake values (SUV) ranging from 5.8 to 11.2 (Figure 8). On examination, the site of the lesion appeared well-healed with no clinical palpable nodule or lymph node in the axilla (Figure 9). To summarize the response of the tumor after receiving appropriate chemo-RT treatment, the patient had resolution of the original lesion but developed new hypermetabolic skin findings adjacent to the original site pointing toward a locally progressive process.

Surveillance PET scan done 1 year later showed a new hypermetabolic 2.2 cm nodule (marked by the red arrow) in the skin and subcutaneous tissues inferior to the mildly hypermetabolic soft tissue changes in the left upper back with the standard uptake values (SUV) ranging from 5.8 to 11.2.

After 1 year of treatment on examination, the site of the lesion appeared well-healed with no clinical palpable nodule.
The case was discussed in the tumor board after the clinical course showed local disease progression after 9 months of treatment and a decision was taken by the multidisciplinary team to observe and switch systemic therapy from ICI to carboplatin, paclitaxel, and cetuximab (monoclonal antibody for EGFR) based on The National Comprehensive Cancer Network (NCCN) Guidelines. The patient is currently under close surveillance of oncology, radiation oncology, and surgery teams receiving systemic therapy and under watchful monitoring with regular PET scans.
Discussion
SMARCA4 (SWI/SNF-related, matrix-associated, actin-dependent regulator of chromatin, subfamily A, member 4) plays an important role in organ development by regulating gene expression, DNA repair, and chromatin accessibility. 11 The SMARCA4 protein acts like a catalytic subunit of the SWI/SNF complex with ATPase activity. 12 There are several SMARCA4 variants identified in human malignancies as evidenced by a recent study done by Fernando et al. 13 Alteration of SMARCA4, leading to mutation in chromatin-remodeling SWI/SNF complex genes and tumor immunity, is found in 3% to 7% of all malignant neoplasms. 14 In medical literature, SMARCA4-deficient undifferentiated tumor (SMARCA4-dUT) of the lung/thorax, small cell carcinoma of the ovary with hypercalcemia type (SCCOHT), and SMARCA4-deficient non–small-cell lung carcinoma (SMARCA4-dNSCLC) are well-known entities. 15 However, in head and neck squamous cell cancers (HNSCC), common genes implicated in the malignancy are TP53, CDKN2A, PTEN, PIK3CA, HRAS, NOTCH1, IRF6, and TP63, whereas SMARCA4 dysregulation is found in only small subset of the cases. 5
There are approximately 1.8 million cases of cSCC annually in the United States. 16 The clinical presentation of cSCC can vary from low-risk SCC in situ to high-risk locally advanced or metastatic cancers with the regional lymph node being the most common site. 17 In the United States, there are 2 staging systems used for cSCC that help predict the risk of metastasis, namely those by Brigham and Women’s Hospital (BWH) and the American Joint Committee on Cancer (AJCC) eighth edition.18,19 The NCCN guidelines, proposed in 2022, categorically divide cSCC into high-risk and very high-risk cSCC based on the presence of risk factors for local recurrence, metastasis, or death to aid in the direction of patient management.16,20
There are multifactorial mechanisms involved in the etiopathogenesis of cSCC including environmental, genetic, and immunological factors. Major predisposing factors include ultraviolet rays, cumulative sun exposure, psoralen and ultraviolet A (PUVA) treatment for skin diseases, HPV subtypes infection, smoking, immunosuppression, and being organ transplantation recipients.21-23 The risk of metastasis in primary cSCC is usually assessed by features including location (lip, ear), immunosuppression, tumor thickness, poor differentiation, previous scar tissue, perineural infiltration, and lymphovascular invasion (LVI). 24 Invasive cSCC is usually seen in sun-exposed areas with 55% developing in the head and neck area and 18% on the dorsum of hand and forearms. 25 The upper back is an extremely rare site for cSCC as it only accounts for 4% of the reported incidence. 25 Poorly differentiated cSCC presents as a soft, ulcerated, rapidly growing lesion with distant metastasis as seen in 27% of the cases. 26 In our case, the patient developed a rapidly enlarging mass more than 2 to 3 months on the upper back with ulceration with metastasis in 2 out of 14 lymph nodes that were resected. At the time of presentation to the clinic, the patient had stage IV invasive cSCC (T3N2bM0) based on the AJCC Staging for cSCC (eighth edition, 2017) (Tables 1 and 2). 19 The characteristics of a high-risk lesion are clinical and notable pathological variables include size, location, poorly defined borders, rapidly growing tumor, perineural invasion (PNI), LVI, poorly differentiated pathology, modified Breslow thickness >4 mm, previous history of RT, and neurological symptoms. 21
TNM definitions for cutaneous carcinoma of the head and neck based on American Joint Committee on Cancer (AJCC) Staging for cSCC (eighth edition, 2017).

Abbreviations: LN, lymph node; ENE, extra nodal extension.
Showing AJCC staging of cSCC (eighth edition, 2017).

Histologically invasive cSCC is recognized by atypical keratinocytes with hyperchromatic, pleomorphic nuclei with mitoses, in addition to locally destructive growth with an increased risk of metastasis. The Broder staging is a predictor of prognosis that includes grading of the lesion: grade I includes tumors composed of <25% undifferentiated cells, grade II lesions with <50% undifferentiated cells, grade III lesions with <75% undifferentiated cells, and grade IV lesions with >75% undifferentiated cells. 27 Surgical resection is the primary modality of treatment for the majority of cSCC however 5% metastasize to nearby lymph nodes leading to a 3-year disease-free survival rate of 56% and a 5-year survival rate of 25% to 35%.28-30
To understand the molecular mechanism of etiopathogenesis of cSCC, we need to gain knowledge about the several genomic variants implicated in cSCC. SMARCA4 gene is located on chromosome 19p and encodes BRG1 (Brahma-related gene 1) protein, forming part of SWI/SNF chromatin-remodeling complex. 31 Approximately 20% of malignancies are found to have abnormalities in one of the genes encoding for the subunits, which thus confers promising results for the gene mutations in terms of acting as a marker for ICI responsiveness. 14 There were 2 kinds of mutations observed in the SMARCA4 category namely loss of function class 1 mutations and missense class 2 mutations. 32 A comprehensive analysis of genomic mutation in cSCC revealed several functionally significant somatic alterations belonging to cancer signaling pathways mainly the RAS/RTK/PI3K pathway, cell cycle pathway, squamous cell differentiation pathway, and chromatin-remodeling genes. 33 Yilmaz et al 4 analyzed exome and targeted sequencing studies to identify potential driver mutations on cSCC; most were found to have mutations of tumor suppressor tumor protein p53 (TP53), NOTCH pathway genes, CDKN2A, KMT2D, and KAT6A. Among the DNA repair genes, however, SMARCA4 mutation contributed around 33%. Frequent somatic alteration of the SMARCA4 gene, known for tumor suppressor function, was estimated to be approximately 5% in the HPV-negative viral subgroup. 5 Treatment modalities for patients with metastatic cSCCs are lacking and have been limited by a lack of knowledge of the genomic alterations that drive metastatic cSCCs. In addition, there are no validated molecular biomarkers predictive of disease behavior or treatment response. Currently, numerous trials are underway to investigate a combination of targeted therapies and immunotherapies in conjunction with chemotherapy or RT in an adjuvant and neoadjuvant setting. 34 After searching the literature, we found only couple of case reports of SMARCA4-positive cSCC including study done by Yvonne Li et al 10 on the genomic analysis of metastatic cSCC identifying 1 sample with SMARCA4 mutation; however, no details of the case were given in the article.
Surgical excision with a 4 to 6 mm margin of uninvolved skin around the tumor is the standard care of treatment for low-risk cSCC, with the American Academy of Dermatology (AAD) recommending excision to the depth of the subcutaneous adipose tissue. 35 However, there are limited data available for defining peripheral and deep margins for excision of high-risk tumors with standard excision as our case belonged to that category. A linear repair, skin graft, or healing by secondary intention is considered a surgical treatment modality for high-risk cSCC. 36
The role of adjuvant RT coupled with systemic therapy has been well-studied for high-risk cSCC. 37 The AAD recommends adjuvant RT for cSCC with concerning high-risk features including PNI and regional or distant metastasis after surgical excision. However, as per guidelines, there is no high-level evidence to support this treatment approach. 35 Similarly, adjuvant chemotherapy with platinum-based agents (cisplatin and carboplatin) and an anti-metabolite with 5-FU have been used for locally advanced/metastatic tumors but need more future prospective data to support the regimen. 38 The use of systemic chemotherapy has been able to achieve partial response in 34% to 44% of the cases with median progression-free survival (PFS) and overall survival (OS) of about 5 and 11 months, respectively, as evident in nonrandomized trials. 39 Targeted therapy has been used for advanced cSCC expressing epidermal growth factor receptor (EGFR) which is inversely correlated with clinical outcome and the degree of expression does not correlate with EGFR inhibitor’s efficacy. 40 Cetuximab (monoclonal antibody for EGFR) obtained an objective response of 28% with a mean OS of 8.1 months in a phase II study in which it was used as a first-line monotherapy in patients with unresectable SCC of the skin. 41
There has been a strong link between the immune system, innate immunosurveillance, mutational burden, and pathogenesis of cSCC. Studies have proven that immunodeficient states along with several mechanisms emphasizing the role of immune-tolerant microenvironment promote a higher risk of developing cSCC.42,43 Inflammatory cytokines, including IL-6, IL-10, IL-2, and TGF-beta, promote infiltration of Treg cells and myeloid-derived suppressor cells (MDSCs) that actively suppress CD4/CD8 T lymphocytes thus creating a tumor microenvironment that stimulates proliferation of tumor cells. 44 The expression of an immune checkpoint molecule such as programmed-death ligand 1 (PD-L1) is upregulated in cSCC showing a positive correlation between PD-L1 expression and risk of metastasis. 45 A study conducted by Hanna et al 34 evaluated the tumor mutational landscape in squamous cell carcinoma of the head and neck (SCCHN) based on anti-PD-1/L1 response and found that nonexclusive somatic alterations in NOTCH1, TP53, KMT2D, and SMARCA4 were the most commonly reported mutations among the 12 sequenced responders out of 81 cohort of patients. Currently, cemiplimab and pembrolizumab, monoclonal antibodies that bind to PD-1 and block the interaction with its ligands PD-L1/L2 have been studied and recommended for locoregional advanced or metastatic cSCC. 46
A phase 1 multicohort study 47 (n = 26 patients) and phase II EMPOWER-CSCC study48,49 (n = 193 patients) investigated and demonstrated the safety and efficacy of cemiplimab in cSCC. In total, 33.7% of patients had prior systemic treatment while 90.2% had prior surgery and 67.9% had received prior RT. 50 Independent central review showed that the response rate was 50% with a duration of response that exceeded 6 months in 7 of the 13 responding patients. 47 In the phase II study, 3 groups were present, namely group 1 (metastatic cSCC), group 2 (locally advanced cSCC) received 3 mg/kg once every 2 weeks, and group 3 (metastatic cSCC) received 350 mg once every 3 weeks. An objective response was noted in 49.2%, 43.6%, and 41.1% of patients in groups 1, 2, and 3, respectively, with a median response time of 1.9 months in groups 1 and 2 and 2.1 months in group 3. 48 Median OS and PFS were not reached by any treatment group.
In addition, KEYNOTE 629 (n = 105 patients) and CARSKIN (n = 57 patients), both phase II multicentric trials, investigated the use of pembrolizumab in cSCC. 51 In both groups, patients had received prior therapies either systemic or RT and showed the objective response rate of 34% with 69% experiencing durable responses longer than 6 months. On follow-up for approximately 10 months, the 1-year OS was 60% and the median PFS was 7 months. In the future, innovative regimens combining RT with immunotherapy are being proposed and studied under the UNSCARRed trial, which acts to combine avelumab (PD-L1 antibody) with radical RT 52 (clinicaltrials.gov: NCT03737721).
Our patient underwent wide surgical excision of the mass along with lymph node dissection of the left axillary region. He received concomitant chemoradiation with cisplatin and RT for 8 weeks. Given his tumor being high risk (LVI, bulky, positive lymph nodes), he was started on pembrolizumab after chemo-RT. He demonstrated partial response by resolution of hypermetabolic lymph nodes in the left axilla and the primary site at the upper back; however, a new lesion appeared adjacent to the primary site. Nevertheless, the time gap of progression of the disease in our case was approximately 1 year which by itself is a rare finding keeping in mind the highly aggressive nature of SMARCA4-positive variant in any tumor with median survival being approximately 6 to 8 months. Currently, we are awaiting the response of the tumor to the changed treatment plan from ICI to chemotherapy (carboplatin, paclitaxel, and cetuximab) based on NCCN guidelines.
Conclusion
Therapies for patients with metastatic cSCCs are lacking and have been limited by a dearth of knowledge of the genomic alterations that drive metastatic cSCCs. In addition, there are no validated molecular biomarkers predictive of disease behavior or treatment response. SMARCA4 is involved in developmental processes, transcriptional regulation, DNA repair, cell cycle control, and cancer genesis. Our case study encompasses the prognostic landscape of SMARCA4 expression and mutation across cancers as well as its role in regulating tumor microenvironment. This article elucidates the consequential need to conduct high-level research to identify several drug targets/ligands that can help with the development of new systemic therapies, including immunotherapy resulting in improved OS and clinical outcome of cSCC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
