Abstract
Pulmonary contusion (PC), defined as damage to the lung parenchyma with edema and hemorrhage, has classically been associated with acceleration-deceleration injuries. It is a frequent pathology in clinical practice. However, its clinical presentation and imaging findings are nonspecific. Patients with this entity can present with findings that can range from mild dyspnea to life-threatening respiratory failure and hemodynamic instability. We present the case of a 61-year-old man, a former smoker, who presented to the emergency department after suffering blunt chest trauma. On admission, he complained of only mild shortness of breath, and his vital signs were typical. Initial imaging identified asymmetric pulmonary infiltrates and mediastinal lymphadenopathy; this was suspicious for additional pathology in addition to PC. After an exhaustive evaluation, a neoplastic or infectious disease process was ruled out. Even though the patient presented with a clinical deterioration of respiratory function compatible with secondary acute respiratory distress syndrome, there was a complete recovery after supportive measures and supplemental oxygen. In conclusion, the nonspecific clinical and imaging findings in patients with pulmonary contusion warrant a complete evaluation of these cases. An early diagnosis is essential to establish adequate support and monitoring to prevent possible complications that could worsen the patient’s prognosis.
Introduction
At the end of the 18th century, the Italian anatomist Giovanni Battista was the first to describe an injury to the lung parenchyma without injury to the chest wall. 1 Pulmonary contusion (PC) occurs when there is direct or indirect damage to the lung parenchyma that produces edema and alveolar hemorrhage, resulting in lung function deterioration. 2 Pulmonary contusion has been most commonly reported secondary to a rapid deceleration when the moving chest hits a fixed object 3 and has been identified as a risk factor for the occurrence of acute respiratory distress syndrome (ARDS), 4 pneumonia, 5 and long-standing respiratory functional impartment. 1
The clinical presentation of PC varies according to the severity of the pulmonary injury, ranging from mild dyspnea to life-threatening respiratory failure. Its radiological examination is unspecific. Although it does not have an established therapy, adequate supportive management and monitoring can prevent unintentional lung injury, complications, or ventilatory failure. 2
We describe the case of a 61-year-old former smoker who arrived in the emergency department after suffering blunt chest trauma. He presented with mild shortness of breath and typical vital signs. His imaging showed asymmetric lung infiltrates that suggested a possible additional pathology associated with the PC, such as a lepidic growth pattern of adenocarcinoma. Given this, complementary studies were accomplished. Finally, other diseases were ruled out. The patient did have manifestations of mild secondary ARDS that improved with supportive measures, and he completely recovered.
Case Presentation
A 61-year-old Hispanic male came to the emergency room 3 days after sustaining closed chest wall trauma to the anterior chest. The patient was struck by a cabinet that fell from a height without loss of consciousness or a wall chest wound. Family history was noncontributory. The patient’s medical history was relevant for smoking 18 pack years; he denied previous chronic medical illnesses, and he was not taking any medications.
Upon admission, he was complaining of diffuse chest pain and mild shortness of breath. He denied palpitations or loss of consciousness. During the initial examination, his vital signs were normal, with an oxygen saturation (SpO2) of 95% without supplemental oxygen. No abnormalities were detected in his head and neck. There was an asymmetry to his thorax with decreased respiratory sounds on the right hemi thorax. No signs of penetrating trauma or unstable thorax were found. Heart auscultation and abdominal exam were normal. His extremities were symmetrical, without edema, and he had regular peripheral pulses. Analgesic treatment and supplemental oxygen by nasal cannula of 3 L/min were initiated.
Initial blood work revealed white blood cell counts of 6.3 × 103/mL, neutrophils 90%, hemoglobin 11.9 g/dL, platelets 135 × 103/mL, prothrombin time 13.1 seconds, partial thromboplastin time 26 seconds, international normalized ratio 1.1, and C-reactive protein (CRP) 4.2 mg/dL. The arterial blood gas revealed a pH of 7.37, partial pressure of carbon dioxide (PaCO2) 33.1 mm Hg, partial pressure of oxygen (PaO2) 74.7 mm Hg, bicarbonate 19 mmol/L, base excess −4.4, and SpO2 93%. The fraction of inspired oxygen (FiO2) was 32%. The PaO2/FiO2 ratio was 233.4. A reverse transcription-polymerase chain reaction assay of an upper respiratory tract specimen also ruled out SARS-CoV-2 infection.
A chest radiograph showed alveolar opacities in the right lung’s upper lobe and a reticular interstitial pattern in both lungs (Figure 1). A computed tomography (CT) scan of the lung was performed. This revealed consolidations in a central distribution in the upper lobes associated with ground-glass opacities and thickening of the intralobular and the anterior lobular septum of the periphery of upper lobes compatible with a “crazy-paving pattern” suggestive of a possible infiltrative process (Figure 2). Although these findings can be found in PCs, they are more consistent with neoplastic processes such as lepidic growth adenocarcinoma, which was felt to possibly be an incidental finding in this case possibly. Other findings were atelectasis of the lateral basal segment of the right lower lobe, adenopathy at station 2R and 4R in the mediastinum (Figure 3), and 2 undisplaced fractures of the left third and fourth ribs without lytic or blast lesions.

Chest X-ray showing consolidation in the apical segment of the right upper lobe with diffuse reticular interstitial infiltrate in both upper lobes, no rib lesions identified.
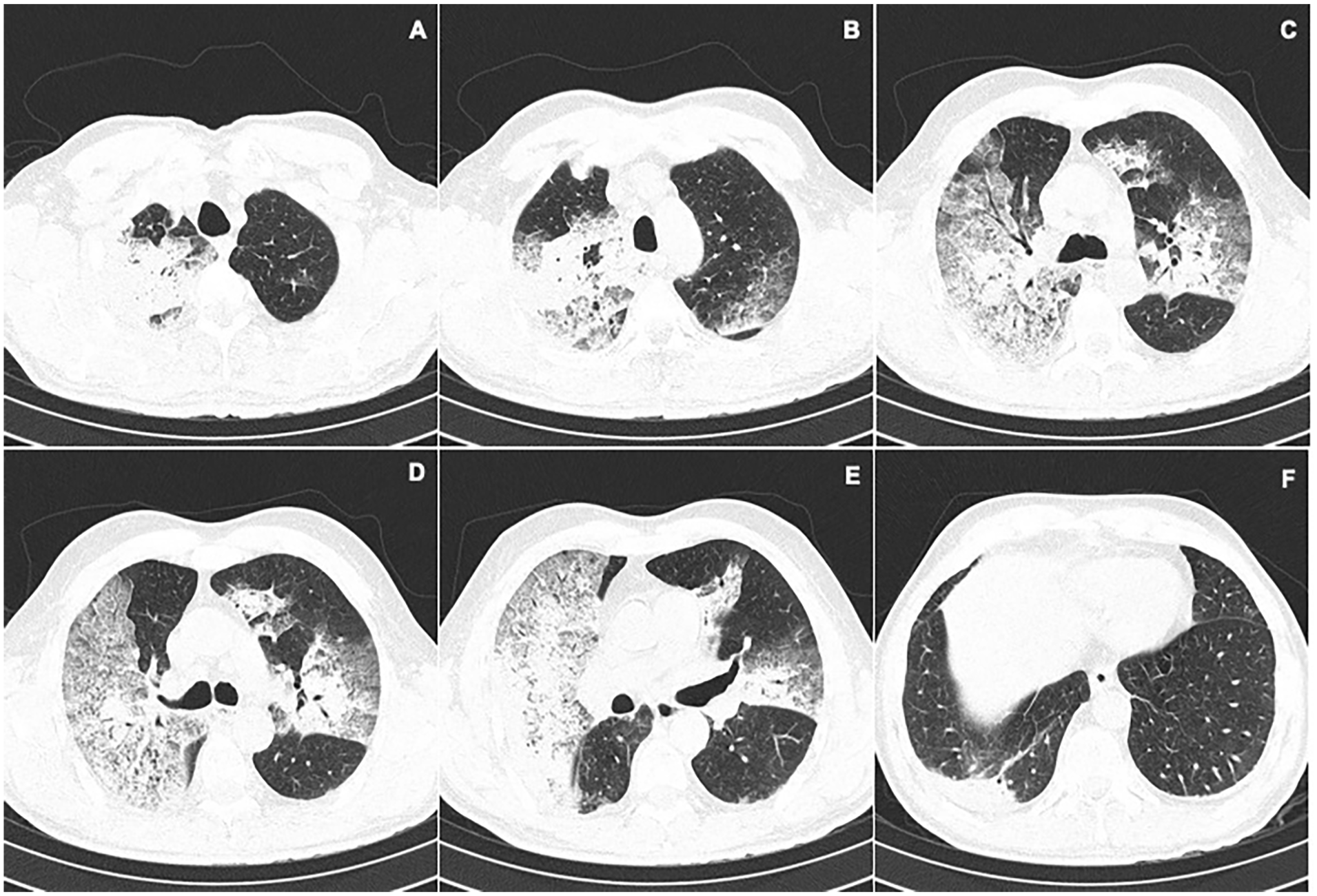
Computed tomography of the chest where an increase in attenuation is identified in A, B, C due to the presence of consolidations of central distribution in the upper lobes. In D, E, F, ground glass infiltrates and thickening of intralobular and interlobular septa are seen that form a “cobblestone” pattern of peripheral distribution.
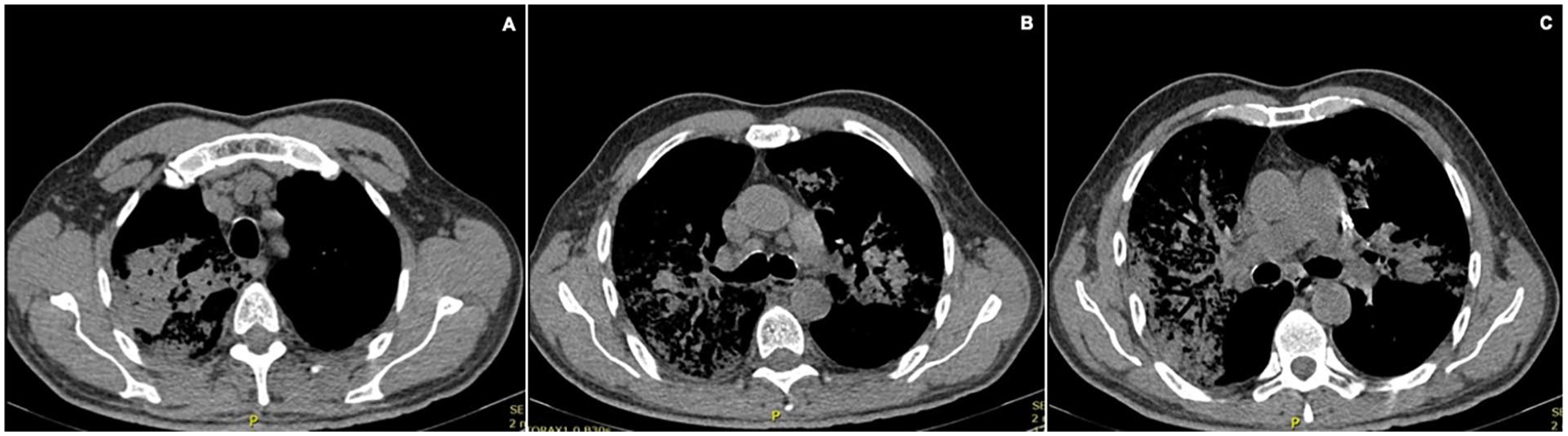
Computed tomography, window for mediastinum. Lymph nodes are found at station 2R (A) and 4R (B, C) measuring 12 mm in the short axis. Non-displaced fractures of the third and fourth left costal arches. No lytic or blastic lesions were observed in the bone structures studied.
Considering that the lesion in the right lobe was highly suggestive of a possible neoplastic process and the patient’s history of smoking, a bronchoscopy with endobronchial ultrasound–guided transbronchial needle aspiration (EBUS-TBNA) was performed. It revealed normal respiratory mucosa and mediastinal lymphadenopathy in stations 2R and 4R.
In the following days, the patient deteriorated clinically with increasing respiratory distress associated with a nonproductive cough. On physical examination, he was afebrile and was found to have a decrease in blood pressure (95/64 mm Hg). His SaO2 remained poor even with nasal oxygen cannula support at 3 L/min. Given the risk of ventilatory failure, he was transferred to the intensive care unit (ICU), where a chest X-ray was taken, evidencing the progression of infiltrates compared with previous images (Figure 4A). He slowly improved his respiratory function after increasing his supplemental oxygen via a high-flow nasal cannula. There was a subsequent increase in the white blood cell count with neutrophilia (white blood cells 21.7 × 103/mL, neutrophils 96%) and an elevated CRP (2.1 mg/dL). This raised the possibility of bacterial pneumonia as a complication, and empirical antimicrobial management was started with piperacillin-tazobactam. The antibiotics were later discontinued due to the absence of fever and an average procalcitonin assay. An additional chest X-ray was performed, evidencing decreased infiltrates (Figure 4B).
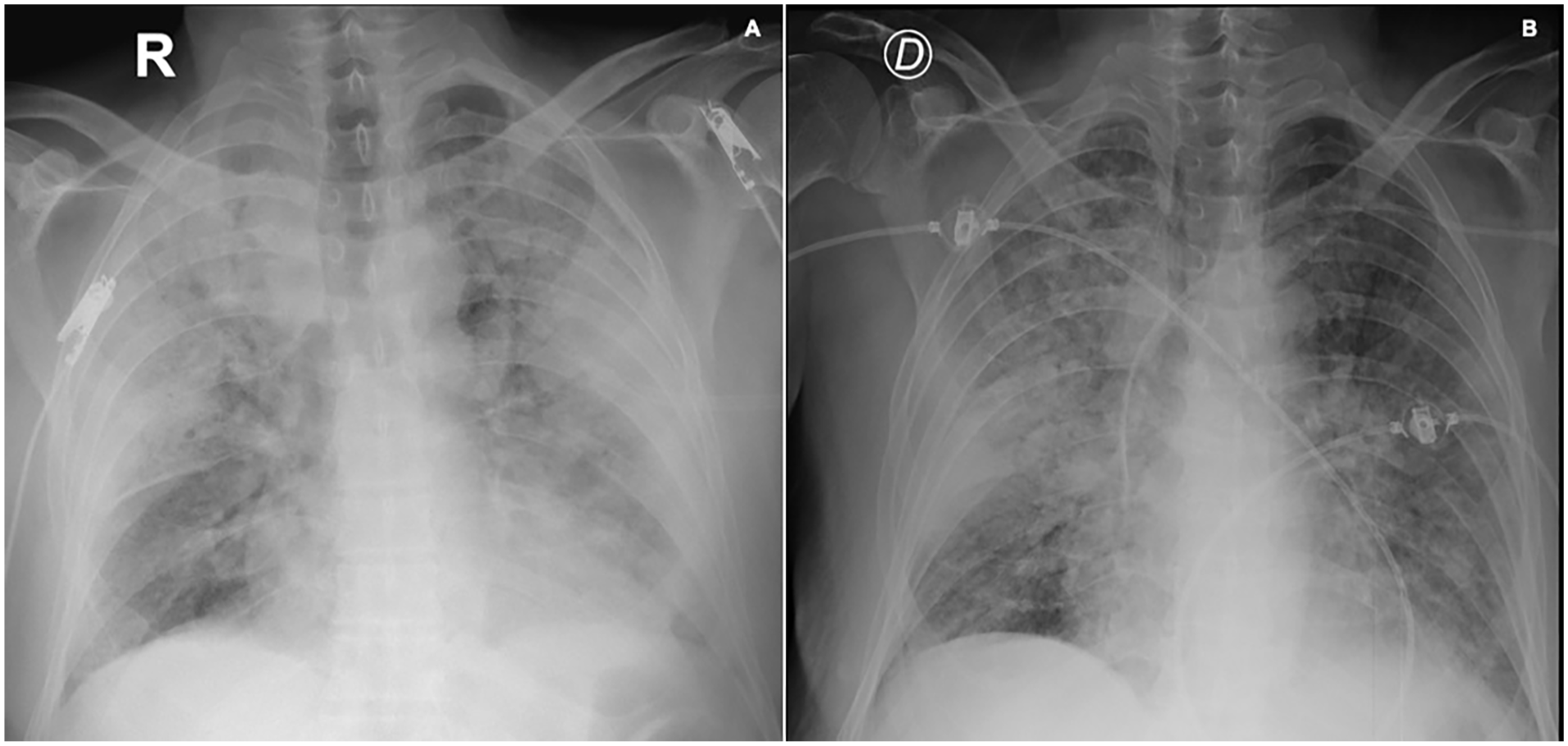
(A) Chest X-ray showing diffuse alveolar compromise pattern with condensation of both upper lobes, with progression of infiltrates compared with previous images. (B) Decreased infiltrates in both upper lobes, mild diffuse interstitial infiltrate persists, no pleural effusion, monitoring elements, and subclavian catheter can be seen.
Finally, the bronchoalveolar lavage results, including smear microscopy, cytology, potassium hydroxide (KOH) test, Romanowsky-Giemsa staining, and GeneXpert test for Mycobacterium tuberculosis, were reported as negative for microorganisms or malignant cells. The lymph node biopsies were negative for malignancy, and the transbronchial biopsy showed only inflammatory infiltrate in the alveoli with a predominance of neutrophils, eosinophils, and lymphocytes and the presence of hyaline membrane. It was negative for malignancy, granulomas, or vasculitis (Figure 5).

(A and B) H&E staining (4×) shows respiratory mucosa and lung parenchyma with alteration of its architecture; acute inflammatory process, hyaline membrane, and fibrin are recognized. (C and D) H&E staining (40×) shows inflammation predominantly polymorphonuclear cellularity with occasional lymphocytes and eosinophils within alveolar lumens with the presence of accompanying fibrin. (E and F) Movat staining (10× and 4×) shows altered lung architecture, with alveolar lumens filled with fibrin, hyaline membrane, and acute inflammation with a predominance of polymorphonuclear cells.
Given the clinical, laboratory, imaging, and histopathological findings of this patient, who presented with PC and mild ARDS, the patient had complete improvement in dyspnea, did not require ongoing supplemental oxygen, and was discharged from the hospital.
Discussion
Pulmonary contusion is the most common lung injury in patients sustaining blunt chest trauma. 1 Our case is peculiar, given that most PCs have been associated with trauma to the chest from sudden deceleration injuries of the body when colliding with stationary objects (eg, in traffic accidents, falls from heights, or sports injuries) or in explosions. 6 The symptoms and signs associated with PC are often not apparent or present at the time of the patient’s arrival and evolve within the first 24 to 48 hours post-injury.7,8
The pathophysiology of PC involves direct trauma to the lung parenchyma and the associated inflammatory response. Direct trauma refers to the damage to pulmonary structures, such as alveoli and capillaries, that produces bleeding into the interstitium and the alveolar space. This may result in a decrease in surfactant production, leading to an increase in alveolar collapse with associated atelectasis. This can be associated with a reduction in lung compliance and an increase in intrapulmonary shunting 9 and explains the appearance of acute symptoms and signs of dyspnea, chest pain, and diminished breath sounds associated with the PC. 10
Lung injury can also activate the immune system, causing the activation of local cells such as macrophages, recruitment of leukocytes, and an increase in the production of pro-inflammatory substances such as cytokines, chemokines, and generation of oxygen radicals. This inflammatory cascade can induce damage to the lung parenchyma and can compromise both lung fields, leading to ARDS. 11 Acute respiratory distress syndrome is characterized by progressive dyspnea, radiology appearance of diffuse alveolar infiltrates, and increased oxygen requirements resulting in a clinical deterioration that this patient progressed to over several days after his arrival.
The most used diagnostic tests in patients with suspected PC include chest radiography and CT scans. The characteristic findings on chest X-rays consist of areas of consolidation with poorly defined borders; these changes generally present 24 to 48 hours after the trauma. 12 On the contrary, CT is the technique with the best sensitivity. The acute changes that have been described by this modality include areas of consolidation or nodular opacity and areas with nonsegmental ground-glass appearance. 2 However, these changes are nonspecific and similar to other disease processes such as pneumonia, pulmonary edema, or neoplasia.
In our patient’s case, he presented classic imaging characteristics of PC. However, his additional findings—such as mediastinal lymphadenopathy on CT scan, interlobular septal thickening, and intralobular lines described as “crazy-paving pattern” combined with the low specificity of the images—presented a diagnostic challenge due to the large number of pathologies that may also manifest these radiological findings. The “crazy-paving pattern” is described as diffuse ground-glass opacities with intralobular lines and interlobular septal thickening. Evidence has shown that it is a radiological pattern that may occur in multiple inflammatory or neoplastic disorders. It is a pattern also found in patients with ARDS, which is a known potential complication of PC. The finding of lymphadenopathy increases the likelihood of other pathologies, such as adenocarcinoma with a lepidic pattern, a well-differentiated type of adenocarcinoma. 13
Mediastinal lymph nodes that present with an abnormality of the lymph node size, density, and/or number are described as “lymphadenopathy.” 14 The most frequent causes of mediastinal lymphadenopathy include inflammatory, neoplastic, infectious conditions and other pathologies such as heart failure, amyloidosis, and even chronic obstructive pulmonary disease (COPD). 15 Given the presence of mediastinal lymphadenopathy in this patient, it required comprehensive evaluation. In this case, due to the patient’s smoking history and the imaging findings, it was necessary to rule out a neoplasm. 15 Without a malignant cause, other causes of these findings, such as COPD, an unidentified infectious pathogen, or an inflammatory response secondary to PC, needed to be explored.
Tissue samples from animal models with PC identified hemorrhage followed by edema and infiltration by inflammatory cells such as lymphocytes, neutrophils, and granulocytes in the alveolar space.2,16 Given these histopathological findings in our patient, this supported the diagnosis of PC.
There is no specific therapy for PC. The central management consists of supportive care while the contusion heals. Early recognition is essential, as adequate monitoring and measures such as avoiding fluid overload, giving mechanical ventilation in severe cases, and pulmonary rehabilitation can prevent additional occurrences, complications, and ventilatory failure.2,17
Conclusions
Pulmonary contusion is frequently encountered. However, its clinical and imaging characteristics are nonspecific and can be similar to other infectious and neoplastic pathologies. A high index of suspicion must always be maintained in patients with high-energy chest trauma, as it may not always present with classic deceleration trauma mechanisms. Therefore, the evaluation of these patients must be comprehensive. Although it does not have a specific therapy, quick recognition is essential to establish and provide support and monitoring. In this way, potential complications are prevented, and the prognosis of these patients is improved.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
All authors have read and approved the manuscript, and significantly contributed to this paper. AIC contributed to literature review, manuscript writing and correction, and final approval of manuscript. LFS contributed to conception and design, literature review, manuscript writing and correction, and final approval of manuscript. AS contributed to literature review, manuscript writing and correction, and final approval of manuscript. LF-T contributed to conception and design, literature review, manuscript writing and correction, and final approval of manuscript.
Availability of Data and Materials
All data and material are available for sharing if needed.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This manuscript was written in compliance with the ethical standards of the institutional ethics committee and with the 1964 Helsinki Declaration. We have approval of the Ethics Committee in Biomedical Research from Fundación Valle del Lili. This is supported in letter No. 223 of 2021. Act No. 13 of 2021 which is available if needed with the Corresponding Author.
Informed Consent
Written informed consent was obtained from the patients for publication of this case series and any accompanying images. A copy of the written consents is available for review by the Editor-in-Chief of this journal.
