Abstract
This case series explores the association between tirzepatide-assisted weight loss and the development of foot drop due to peroneal nerve neuropathy, a phenomenon known as slimmer’s paralysis. Two cases are presented of patients who experienced rapid weight loss after initiation of tirzepatide therapy and within 6 to 8 months developed bilateral foot drop. As providers, we have more medications than ever to assist patients in their weight loss journeys, but both of these cases are reminders of the risks of rapid weight loss and the need to monitor therapy closely for patients on tirzepatide and similar medications.
Introduction
There has been great advances in the last 10 years in the treatment of diabetes and obesity. Medications like liraglutide, semaglutide, and tirzepatide are being used for their benefits in controlling diabetes but also for their benefits for weight loss and cardiovascular risk reduction. With these new medications also comes risks. This case series discusses the association with tirzepatide treatment, weight loss, and the development of neurologic complications, specifically foot drop resulting from peroneal nerve neuropathy.
Case 1
A 77-year-old male with a history of obesity, prediabetes, coronary artery disease, hyperlipidemia, stage 3a chronic kidney disease, chronic compensated heart failure with reduced ejection fraction, history of lumbar decompression (2017), and left total knee arthroplasty (2012) presented in September 2023 with onset of left foot drop and again in October 2023 with development of foot drop on the right as well. Neurologic exam was notable for 5/5 strength in proximal lower extremities but weakness at the bilateral ankles with dorsiflexion (0/5 on right, 1/5 on left) and eversion (1/5 on right, 2/5 on left), diminished sensation to light touch in left lateral leg, and 1+ reflexes at knee and ankle bilaterally. Extensive laboratory testing, including complete blood count (CBC), comprehensive metabolic panel (CMP), thyroid-stimulating hormone (TSH), serum protein electrophoresis (SPEP), vitamin B1 and B6, heavy metals, copper, and treponema pallidium antibodies, was all unremarkable. Vitamin B12 level was 480 pg/mL. Patient does have known history of prediabetes but hemoglobin A1C at the time was 4.9% (highest previously was 6.4% in 2022). Patient had a past history of lumbar spinal stenosis and a new magnetic resonance imaging (MRI) of the lumbar spine was done which noted degenerative changes, L1-L2 and L3-L4 moderate to severe canal stenosis, and multilevel bilateral neural foraminal narrowing—including moderate to severe neuroforaminal stenosis at L4-L5 (see Figure 1).

Magnetic resonance imaging of lumbar spine showing multilevel spinal canal stenosis, most severe at L1-L2 level.
A neurosurgery evaluation was completed by two different specialists who did not feel symptoms were related to the spine but were a result of a peripheral neuropathy given the absence of any significant back pain, neurogenic claudication, radiculopathy, or other related symptoms. A nerve conduction study (NCS) and electromyography (EMG) were then completed for further evaluation. This study showed evidence of an acute on chronic bilateral peroneal nerve neuropathy at the fibular head, more severe on the right without any evidence of lumbar radiculopathy. There was, however, evidence of a length-dependent sensory polyneuropathy versus age-related changes based on neuromuscular specialist interpretation. In addition, the specialist noted that the findings consistent with fibular neuropathies at the fibular head can be seen with rapid weight loss or with compressive lesions (see Figures 2-3 for full nerve conduction and EMG tables). Further evaluation included ultrasounds of the peroneal nerves showing slightly increased neural vascularity of the nerves bilaterally which was of uncertain significance, but ultrasound was otherwise unremarkable. He is currently under evaluation for surgical release and is being managed with orthotics and physical therapy.

MNC and F wave summary for Case 1. The right ulnar and bilateral tibial nerves demonstrate normal distal motor latency, evoked response amplitude and conduction velocity. The bilateral fibular nerves show prolonged distal motor latency, low evoked response amplitude and conduction velocity at the EDB. The right fibular nerve at the TA shows low evoked response amplitude and conduction block at the fibular head while the left fibular nerve at the TA shows normal evoked response amplitude, conduction block at fibular head and slow conduction velocity.

SNC and EMG summary for Case 1. SNC demonstrates normal ulnar sensory evoked response and conduction velocity while there is no sensory evoked response in the bilateral sural and superficial fibular nerves. The EMG shows 2+ t0 3+ fib and discrete or reduced recruitment in the left and right tibialis anterior and fibularis longus while the exam of the remaining muscles were within normal limits.
Only notable medication change prior to the development of foot drop was the initiation of tirzepatide therapy in February 2023. Prior to starting tirzepatide, he weighed 254 lb (body mass index [BMI] 37.51 kg/m2) and A1C was 6.4%. By September, he was down to 193 lb (BMI 27.98 kg/m2), a reduction of about 24% body weight, and A1C had improved to 5.2%.
Case 2
A 56-year-old female with a history of type 2 diabetes mellitus, depression, anxiety, systemic lupus erythematous, Sjogren’s syndrome, and attention-deficit hyperactivity disorder presented with new development of right foot drop in April of 2023. Her exam was notable for 1/5 strength in tibialis anterior/extensor hallucis longus (TA/EHL) but 5/5 throughout the rest of the lower extremities. She had a normal patellar reflex and 1+ ankle reflex. She had labs and an evaluation by neurosurgery, an MRI of the lumbar spine, and a NCS with EMG. Her labs indicated her diabetes was well controlled with an A1C of 4.7% (highest was 7.8% in 2022). Other labs, including CBC, CMP, vitamin B12, vitamin B6, TSH, copper, and SPEP, were all normal with exception of copper level being mildly elevated at 221 mcg/dL (reference range 77-206 mcg/dL) and vitamin B6 being slightly low at 4 mcg/L (reference range 5-50 mcg/L). Her MRI was unrevealing without evidence of spinal stenosis or neuroforaminal narrowing. Her NCS/EMG confirmed evidence of a moderately severe right peroneal neuropathy at the fibular head (see Figure 4). She ultimately underwent a right peroneal nerve release in May of 2023 and was managed with an ankle foot orthotic (AFO) and physical therapy.

Initial NCS and EMG summary for Case 2.
She subsequently developed a left foot drop in August of 2023 with EMG again confirming a peroneal nerve neuropathy secondary to compression at the fibular head with similar findings on the left with some interval improvement on the right following decompressive surgery. Of note, the EMG also showed the left vastus lateralis had increased motor unit amplitude and slightly increased polyphasic potentials, the significance of which was not known (see Figure 5). Overall, the predominant finding was the peroneal nerve neuropathy. She underwent left peroneal release in September of 2023. She had clinical improvement in foot drop bilaterally following surgical release. Of note, she had not started taking any vitamin B6 supplements.
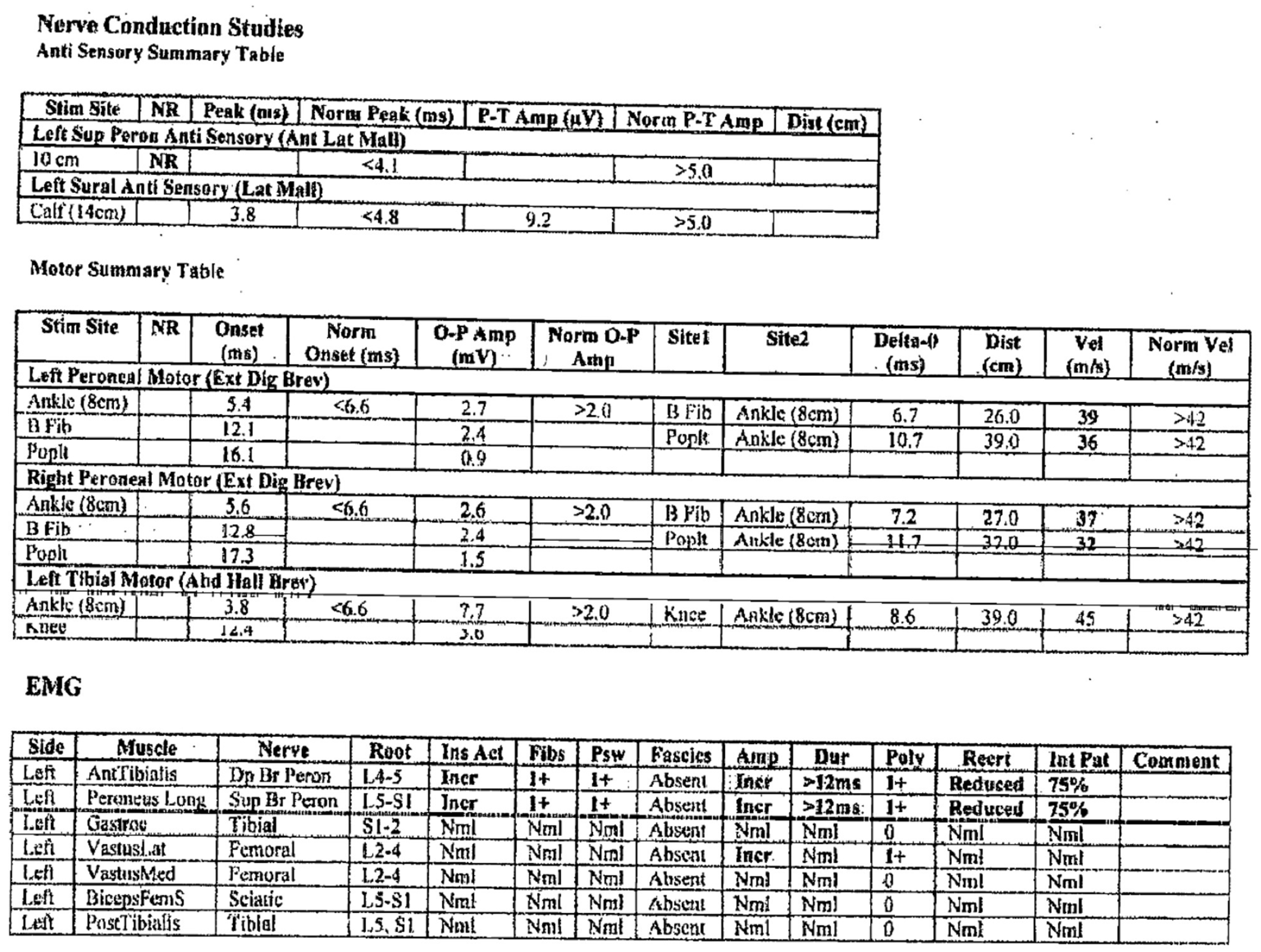
Second NCS and EMG for Case 2.
Reviewing her records, she was diagnosed with diabetes in February 2018 with an A1C of 7.4% and previously had been prediabetic since 2015. Prior to presentation for foot drop, she had not shown any evidence of end organ damage with normal microalbumin testing, normal foot exams, and normal retinal exams. At the time of her initial diagnosis, she was initially started on metformin with limited results and was then started on semaglutide. She remained on semaglutide from October 2019 through September 2022 at which time she was switched to tirzepatide. When she started on semaglutide, her starting weight was 187 lb (BMI 35.37 kg/m2) and A1C was 7.4%. Over the first several months of treatment, she had lost about 30 lb. Her weight loss then slowed significantly over the next 6 months and her weight remained stable over the next 2 years despite dose increases. She was switched from semaglutide 2 mg to tirzepatide in September 2022 due to A1C remaining above goal (7.8% in June 2022 and 6.8% in September 2022), titrating up from a starting dose of 2.5 mg weekly to 15 mg weekly (increasing by 2.5 mg each month). Her weight when she started tirzepatide was 146 lb (BMI 27.66kg/m2) and by April 2023 was down to 111 lb (BMI 21.09 kg/m2) and reached a low of 105 lb (BMI 19.84 kg/m2) in September 2023, a reduction of 28% body weight since initiation of tirzepatide. Her A1C normalized to 4.7% by August 2023.
Discussion
Slimmer’s paralysis was a term initially coined by Soteniemi in 1984 1 documenting a case of peroneal nerve neuropathy after rapid weight loss. This effect has continued to be well documented over the years. Most notably, there are multiple studies looking at this effect following bariatric surgery.2 -4 The mechanism seems to be loss of a fat pad that protects the peroneal nerve. 5 With this loss of protection, even leg crossing can cause injury to the nerve.6 -8 Interestingly, in the first case above, the patient reported that before his weight loss he could not physically cross his legs, but after he lost significant weight he started crossing his legs frequently. There are a wide range of potential causes for foot drop, including spinal pathology, metabolic disorders, autoimmune diseases, compressive lesions, surgical complications, and so on. In each case above, there are certainly multiple possible causes of foot drop but given the evidence of a specific peroneal nerve neuropathy and the timing of the onset with the weight loss, there is definite concern for slimmer’s paralysis in these cases.
In the first case, there was initially a suspicion that the foot drop was secondary to his known spinal disease. His MRI was concerning, with evidence of canal stenosis and multilevel neuroforaminal stenosis; however, his EMG did not support lumbar radiculopathy but rather pointed to an acute on chronic neuropathy of the peroneal nerves at the fibular heads with evidence of active denervation and reinnervation in fibular-innervated muscles and absence of EMG findings in other tested muscles. Imaging was unable to find a mass in the area that could be causing compression of the nerve and laboratory evaluation was equally unrevealing. Given his evaluation and timing of onset, this case suggests compressive peroneal neuropathy due to his rapid weight loss. Of note, his sensory nerve conduction did demonstrate absent responses of the sural and peroneal nerves bilaterally. This could be representative of an underlying polyneuropathy but also may be a normal finding based on his age. Studies have shown that sensory responses decline with age and a recent study by Taams et al demonstrated that as many as 30% of males over the age of 80 will have absent sural nerve amplitude potentials.9,10 Thus, it is possible that this is a normal finding for his age as noted by the neuromuscular specialist who interpreted the results.
The second case also had multiple other possibilities on the differential, with the most suspicious being her history of autoimmune disease which can be associated with polyneuropathy and mononeuritis multiplex. There is even suggestion of an abnormality in the vastus lateralis on her EMG for which the significance was unknown. However, this abnormality was only noted on the second EMG and her clinical course suggests that compressive peroneal neuropathy is the primary etiology as she clinically improved following surgical release.
With the introduction of more medications that cause significant amounts of weight loss, we are going to see more complications of rapid weight loss like what was seen in these cases. Tirzepatide is a medication that has been shown to cause unprecedented levels of weight loss, that even rival weight loss seen following bariatric surgery. 11 In the case of bariatric surgery, faster weight loss was associated with a higher risk of peroneal nerve neuropathy and the incidence occurred mostly 5 to 11 months following surgery—as was seen in these cases as well.2,12 The lessons learned in managing these post-bariatric surgery patients can also be applied to patients who have rapid weight loss with medication assistance. Close monitoring with dose adjustments to help slow the rate of weight loss should be considered to help reduce the risk of weight loss complications like peroneal nerve neuropathy and foot drop.
For treatment, we can also turn to the lessons learned in management of bariatric surgery patients. Studies have seen that peroneal nerve decompression is effective and can lead to recovery. The sooner we recognize and diagnose, the quicker we can get these patients to surgery to speed their road to recovery.7,12 In the second case, the patient showed clinical improvement in foot drop following surgical release which was done within a couple of months of symptom presentation.
Slimmer’s paralysis is not a new phenomenon, but its incidence is certain to increase as more effective weight loss medications become available. It is important for us as providers to be aware of these dangers so we can adjust medication titration, counsel regarding these risks, and efficiently manage these complications when they occur. We need to be prepared.
Conclusion
Rapid weight loss is associated with complications, including peroneal nerve neuropathy. These complications will be more common with the influx of effective weight loss medications on the market, like tirzepatide. Peroneal nerve neuropathy, resulting in foot drop, is one of these complications that we need to be aware of and try to prevent, but quickly recognize and treat when it occurs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
