Abstract
The melanocortin (3 or 4) receptor (MC3/4R) is involved in regulating satiety and body weight. Therefore, pathogenic mutation in MC3/4R is associated with severe obesity, for which bariatric surgery is one of the treatment options. However, there is limited data on whether individuals with MC3/4R mutation will have differential weight response to surgery, especially among the Asian populations—the epi-center of the evolving global obesity epidemic. From our large prospective Obesity—Metabolism & Intervention Cohort Study (OMICS; N = 654, recruited between 2007 and 2022), 5 individuals with pathogenic MC3/4R mutations (“case”) were identified using candidate-genes panel next-generation sequencing (Illumina iSeq). These subjects were carefully propensity score–matched (baseline body mass index [BMI], age, sex, ethnicity, proportion with diabetes, type of bariatric surgery) in a 1:4 ratio to other controls. We performed linear mixed model analysis (for repeated measurements) to compare their longitudinal weight trajectories (percentage total weight loss, %TWL) over 12 months. The 5 cases with MC3/4R mutations were 48 ± 11 years, BMI 40.8 ± 11.2 kg/m2, 60% with diabetes, and all males. Their weight at baseline (pre-op), and 6 months and 12 months after surgery were 120 ± 38, 100 ± 31, and 101 ± 30 kg, respectively. Compared with propensity score–matched controls (N = 20), linear mixed model analysis suggested no difference in surgically induced %TWL (β coefficient = −5.8 ± 3.7, P = .13) over 12 months between the groups. Therefore, we conclude that rare pathogenic MC3/4R mutations do not significantly modify weight change (%TWL) in response to bariatric surgery.
Introduction
Obesity is a common complex condition driven by multiple genetic and environmental factors. The genetic architecture of obesity comprises both common variants (each with modest effect) and rare variants with moderate to large effect size. 1 Hypothalamic arcuate nucleus’ melanocortin (3 or 4) receptor (MC3/4R) is responsive to opposing regulatory signals from melanocyte-stimulating hormone (MSH) and agouti-related protein (AGRP). These peptides are derived from the pro-opiomelanocortin (POMC)-expressing neurons and AGRP-expressing neurons, respectively. Thus, MC3/4R is involved in energy homeostasis, satiety, and body weight control. 2 Rare pathogenic mutations (compound heterozygous or homozygous) in MC3/4R have been associated with severe obesity, for which bariatric surgery is one of the treatment options. However, there is limited data on whether individuals with less deleterious heterozygous pathogenic MC3/4R mutation will have differential weight response to surgery, especially among Asian population—the epi-center of the evolving global obesity epidemic. Hence, we investigated whether rare pathogenic MC3/4R mutations conferred any differential weight response to bariatric surgery in a large multi-ethnic Asian population with severe obesity.
Materials and Methods
The study population is nested within our large prospective Obesity—Metabolism & Intervention Cohort Study (OMICS), which has previously been described. 3 In brief, moderate to severely obese adults (mean body mass index [BMI] 42.6 ± 7.9 kg/m2) with or without diabetes scheduled for metabolic surgery were recruited between August 2007 and May 2022 (N = 654). This retrospective study included people who underwent laparoscopic adjustable gastric banding (LAGB), Roux-en-Y gastric bypass (RYGB), or sleeve gastrectomy (SG) and had at least 1-year follow-up data post-surgery. Written informed consent was obtained from all subjects.
Information on medical history and medication in use was recorded by questionnaire. Height and weight were measured by standard procedure using digital scale by Tanita, and BMI was computed as weight (in kg) divided by height (in m2). Body weight was captured at baseline (pre-op), and 6 and 12 months post-surgery as part of routine care. Percentage of total body weight loss (%TWL) was calculated as: 100 × (preoperative weight − postoperative weight)/preoperative weight.
Genetic Analysis
Genomic DNA was extracted from archived peripheral blood specimens using the QIAamp DNA Blood Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s specifications. The entire exonic regions of 21 genes related to non-syndromic obesity were amplified with a customized gene panel designed using the Illumina Design Studio Software (Illumina, San Diego, California). The polymerase chain reaction (PCR)-based library preparation was performed using the AmpliSeq Library PLUS for Illumina kit according to the manufacturer’s instructions, followed by sequencing on the iSeq 100 System. The demultiplexed FASTQ files were generated using the built-in iSeq Local Run Manager Generate FASTQ Analysis Module v2.0. The downstream analysis for streamlined mapping (human genome reference hg38) and variant detection was done using the DRAGEN DNA Amplicon App (Illumina). Reported variants were filtered based on their frequency and functional effects. Variants with minor allele frequency <1% according to the ExAC data were flagged as rare/novel and subjected to further analysis. Bi-directional Sanger nucleotide sequencing was performed to confirm the presence of variant.
The Alamut Visual Plus version 1.3 software (Sophia Genetics, Lausanne, Switzerland) was used to determine the pathogenicity of the variants. Functional classification was performed according to the American College of Medical Genetics and genomics (ACMG) 2015 guideline that classifies variants as pathogenic, likely pathogenic, variant of unknown significance (VUS), likely benign, and benign. Furthermore, online bioinformatics tools such as PolyPhen-2, Sorting Intolerant From Tolerant (SIFT), Mutation Taster, Align GVGD, The Human Gene Mutation Database (HGMD), dbSNP, NHLBI Exome Sequencing Project (Exome Variant Server), Exome Aggregation Consortium (ExAC), and 1000 Genomes were used to support the variant classification.
Statistical Analysis
Continuous variables were expressed as mean ± SD, and categorical variables were expressed as proportion (%). Subjects with rare pathogenic MC3/4R mutations were identified as “cases.” “Controls” were derived from other null-mutation participants of the OMICS cohort by careful propensity score matching with the “cases” at a ratio of 4:1. Age, sex, ethnicity, and BMI were included as covariates to derive the propensity scores, and matching was done in a 4:1 ratio with controls (null MC3/4R mutation group) to cases (MC3/4R mutation group). Propensity scores were calculated using logistic regression to create a multivariate composite of the covariates. 4 When creating propensity scores via logistic regression, the algorithm is simply computing the probability that a person is having the MC3/4R mutation, given the set of covariates included in the model. Subsequently, the balance between presence and absence of MC3/4R mutation was determined by using one-to-many matchings, in which the nearest neighbor approach was used to generate matches from propensity scores. 5 The quality of matching was assessed by comparing the balance between the cases (MC3/4R mutation group) and controls (null MC3/4R mutation group) (Figure 1) both numerically and visually.
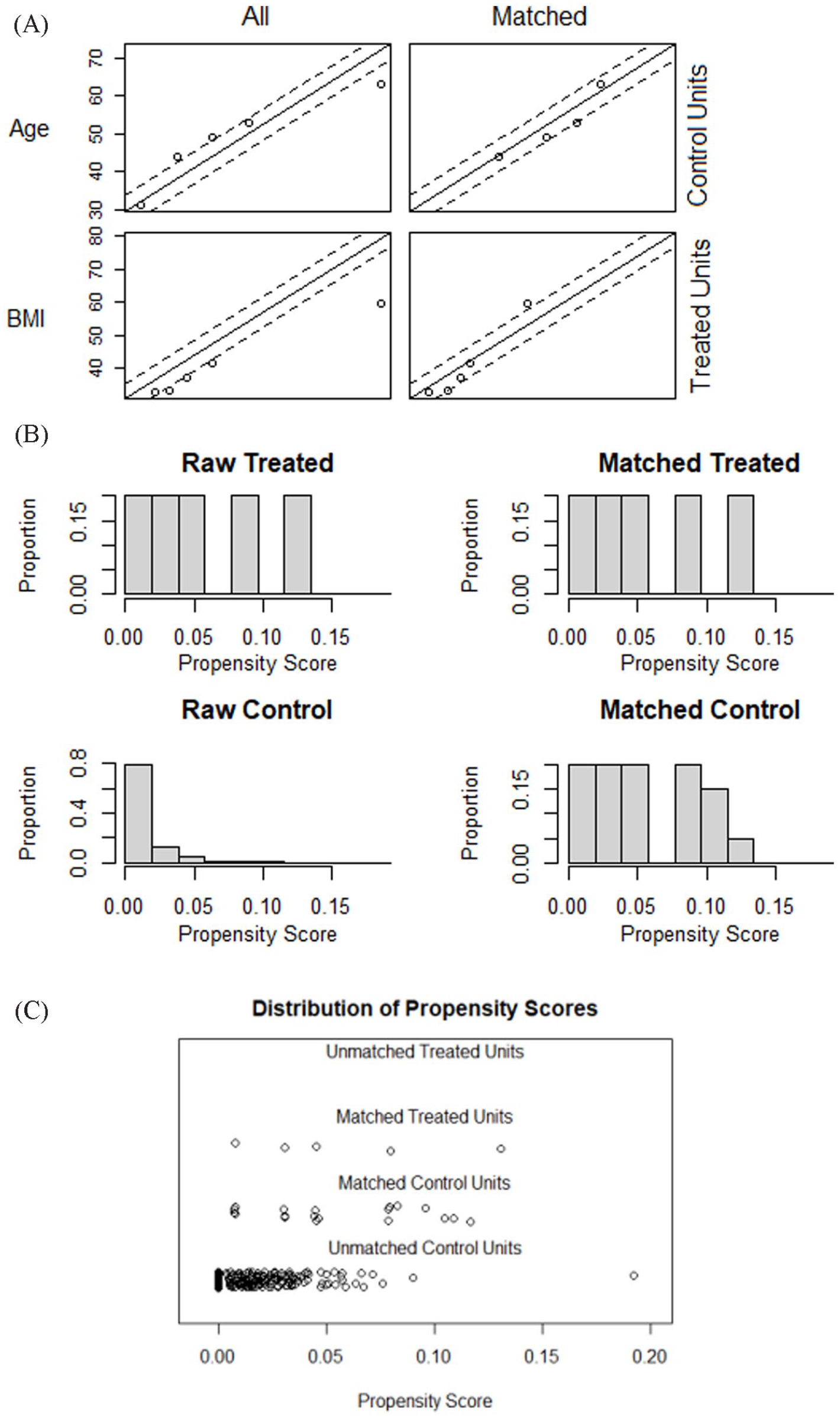
Propensity scores: (A) Quantile-quantile plots to show good correlation in age and body mass index; (B) bar chart to show improvement in scores between cases and controls after matching; and (C) distribution of scores well-matched between cases and controls.
Linear Mixed-Effects Model
We used mixed-effects models because of the dependency and correlation among repeated observations (over time) in the dataset. Body weight was measured at baseline, and 6 months and 12 months after surgery. Therefore, we used a random intercept model. A random intercept model allows the intercepts to vary, and therefore, the values on the dependent variable (%TWL) for each individual observations were predicted by the intercept that varied across groups.
Results
After careful annotation, 5 subjects with pathogenic MC3/4R mutations were identified (Table 1), suggesting a prevalence of 0.76% (5/654). They were all males, mean age 48 ± 11 years, BMI 40.8 ± 11.2 kg/m2, 60% had diabetes, and mixed ethnicity of Chinese:Indian:Malay:Caucasian 20:40:20:20%, respectively. Their average weight at baseline (pre-op), and 6 months and 12 months after surgery were 120 ± 38, 100 ± 31, and 101 ± 30 kg, respectively. Compared with propensity score–matched controls (N = 20), linear mixed model analysis suggested no difference in surgically induced %TWL (β coefficient: −5.8 ± 3.7, P = .13) over 12 months between the groups, with the presence of covariates age, BMI, and ethnicity (Figure 2).
Clinical Characteristics of the Five Patients With Pathogenic Melanocortin (3 or 4) Receptor Mutations.
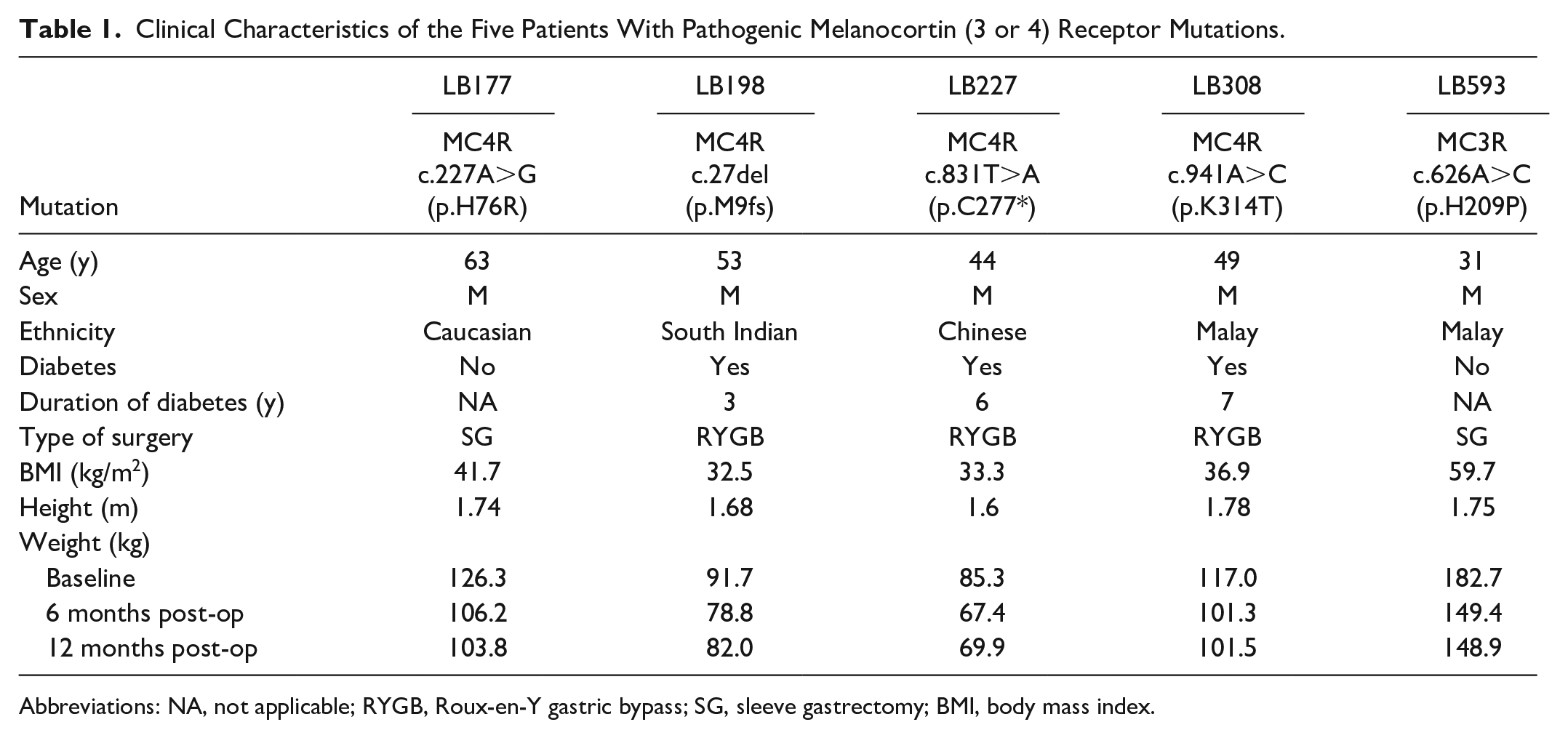
Abbreviations: NA, not applicable; RYGB, Roux-en-Y gastric bypass; SG, sleeve gastrectomy; BMI, body mass index.

Boxplot (A) absolute weight at baseline, and 6 and 12 months; (B) total percentage weight loss (%TWL) at 6 and 12 months post-surgery. Pink box: mutation-negative individuals (controls); turquoise box: mutation-positive individuals (cases).
Discussion
There are 2 key findings in this retrospective study. First, the prevalence of heterozygous pathogenic MC3/4R mutation is rare in our adult bariatric surgery population, at approximately 0.76%. Second, subjects with rare pathogenic MC3/4R mutations experienced 1-year weight-reduction trajectory similar to propensity score–matched individuals with null MC3/4R mutation.
The low prevalence of adults with severe obesity and heterozygous pathogenic MC3/4R mutation in our bariatric surgery cohort (0.76%) is broadly concordant with other similar cohorts, which mostly reported a prevalence of 1% to 2%.6-8 Therefore, pathogenic MC3/4R mutations only account for a small fraction of the genetic attributable risk for severe obesity in such populations. Hence, routine unselected screening for such mutation may not be warranted in the bariatric surgery weight-management clinics.
We observed non-differential 1-year weight-loss response to bariatric surgery by MC3/4R pathogenic mutation status. Our result is in agreement with that from other similar cohorts, cited above. This is perhaps not a surprise, given the evolving mechanistic insights of how bariatric surgery may mediate weight reduction and improvement in metabolic health. Some of these mechanisms include favorable change in the secretion of gut hormones (eg, glucagon-like-peptide 1), microbiome, bile acids (eg, cholic acid and chenodeoxycholic acid), and central neuronal-circuitry appetite control. 9 Hence, dysregulation of the latter by MC3/4R mutation may not be sufficient to negate the weight-reduction benefit of surgery. In corollary, bariatric surgery remains a viable therapeutic option to achieve meaningful weight reduction among Asians with severe obesity and MCR3/4R mutation.
The strengths of this report are, first, the large sampling frame (N = 645) of a decade-long multi-ethnic Asian bariatric surgery cohort with rich bio-repository and longitudinal data; and second, the extensive candidate gene sequencing for all the cohort participants. To the best of our knowledge, there has not been such a report in the literature. This has allowed us to accurately ascertain individuals with MC3/4R pathogenic mutations. Third, 4 controls to 1 case propensity score matching was performed to maximize the study’s statistical power. A weakness is the small number of subjects with rare pathogenic MC3/4R mutation. However, given the rarity of monogenic obesity and anticipated large effect size (if any) of such mutations, our study is at least on-par with previous publications to observe any clinically meaningful results. In addition, we do not have long-term data (eg, 5-year follow-up) to observe their susceptibility for weight regain.
Conclusion
Among Asians with severe obesity, rare pathogenic MC3/4R mutations do not significantly modify weight change (%TWL) in response to bariatric surgery. Our data may inform precision obesity management for individuals with such mutations in the clinics.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the Alexandra Health Fund Ltd through the Small Innovative Grant SIG/14034 and Alexandra Health Science Translational and Applied Research Grant STAR20202. SCL is supported by the Singapore Ministry of Health’s National Medical Research Council NMRC MOH-000714-01, MOH-000066, and MOH-0000714.
Ethical Approval
Clinical data and biospecimens were obtained with informed consent under a protocol approved by the National Healthcare Group Domain Specific Review Board (2014/01234 and 2020/00245).
Informed Consent
Informed consent for patient information to be published in this article was not obtained because the patient data and samples were de-identified.
