Abstract
Bartonella species are gram-negative coccobacilli that are globally recognized as significant pathogens causing zoonotic infections. Among Bartonella species, B. henselae, B. quintana, and B. bacilliformis are prominent pathogens causing infections in humans, often manifesting as infective endocarditis. Bartonella endocarditis can pose diagnostic challenges due to its indolent presentation and limitations of standard microbiological culture techniques to identify the organism. We report a case of a 23-year-old male, who initially presented with the manifestations of hepatosplenic T-cell lymphoma, later diagnosed with blood culture–negative endocarditis caused by B. henselae. The patient had a complicated clinical course including pancytopenia, hepatosplenomegaly, and severe aortic valve regurgitation. Despite negative blood cultures, diagnostic clues included persistent fevers and bicuspid aortic valve with abscess. High Bartonella IgG titers (>1:800) supported the diagnosis. This case underscores the importance of considering Bartonella species in patients with suspected endocarditis, particularly in those with predisposing factors and negative blood cultures. Diagnosis relies heavily on serologic assays due to low sensitivity of conventional culture methods. Treatment involves a multidisciplinary approach with antibiotics and surgical intervention for optimal outcomes. Timely recognition and management are crucial to mitigate the high mortality associated with Bartonella endocarditis, and we hope this article offers insight for clinicians.
Introduction
Bartonella species are fastidious, gram-negative coccobacilli that cause zoonotic infections worldwide. It is typically transmitted to humans via cat fleas, human body lice, and animal scratches or bites (cats and dogs). Of the 30 different reported species of Bartonella, 12 species have been linked with human infection, among which three are commonly encountered clinically.1,2 These include the causative agents of cat scratch disease (Bartonella henselae), trench fever (Bartonella quintana), and Oroya fever (Bartonella bacilliformis). B. henselae causes cat scratch disease, meningoencephalitis, infective endocarditis (IE), prolonged fever in immunocompetent patients, as well as peliosis hepatis and bacillary angiomatosis in HIV-infected individuals. 3 B. henselae was first isolated in 1992 from an HIV-infected patient who presented with prolonged fever. 4
Bartonella species are responsible for 3% of all cases of IE and are the second most common cause of blood culture–negative endocarditis (BCNE) after Coxiella burnetti. 1 B. quintana has been associated with 75% of all IE cases, followed by B. henselae in 25% of all cases.5,6 IE due to B. quintana has been associated with homelessness or chronic alcoholism and occurs in individuals with no history of valvular disease; meanwhile, IE due to B. henselae has been seen in patients with a previous valvulopathy and contact with cats. 7
Bartonella endocarditis usually has a subacute presentation with patients developing nonspecific symptoms including fever, fatigue, and weight loss. The diagnosis can be challenging to make as the organism is difficult to isolate using standard microbiological culture techniques with sensitivity as low as 20%. 5 For primary isolation of Bartonella, several different agar base formulas (heart infusion, trypticase soy, brucella agar, and Columbia agar supplemented with 5% rabbit blood or 5% hemoglobin) have been used successfully, and extended incubation times of up to 21 days may be required. 5
Bartonella IgG and IgM serologies are the single most informative, noninvasive tool to aid in diagnosis. Immunofluorescence assays (IFAs) and enzyme-linked immunosorbent assays (ELISAs) are both available for B. quintana and B. henselae; however, IFA is preferred due to its better reliability in providing semi-quantitative titers. 6 In 2015, Edouard et al 7 compiled 106 cases of Bartonella endocarditis in the PubMed database and demonstrated IgG levels ≥1:800 correlate with endocarditis in 94% of cases.
Case Presentation
A 23-year-old male presented to the emergency department (ED) with complaints of unintentional weight loss of 70 pounds, night sweats, and fevers for the past 6 months. A computed tomography (CT) scan of the abdomen and pelvis with intravenous (IV) contrast revealed hepatosplenomegaly suggesting lymphoma/leukemia, with wedge-shaped infarcts favoring splenic infarcts (Figure 1A and 1B). A bone marrow biopsy showed lymphoproliferative T-cell lymphoma. He was discharged home, and his case was discussed at the multidisciplinary hematopathology conference. He was readmitted with plans to undergo induction chemotherapy and liver biopsy while admitted as inpatient.
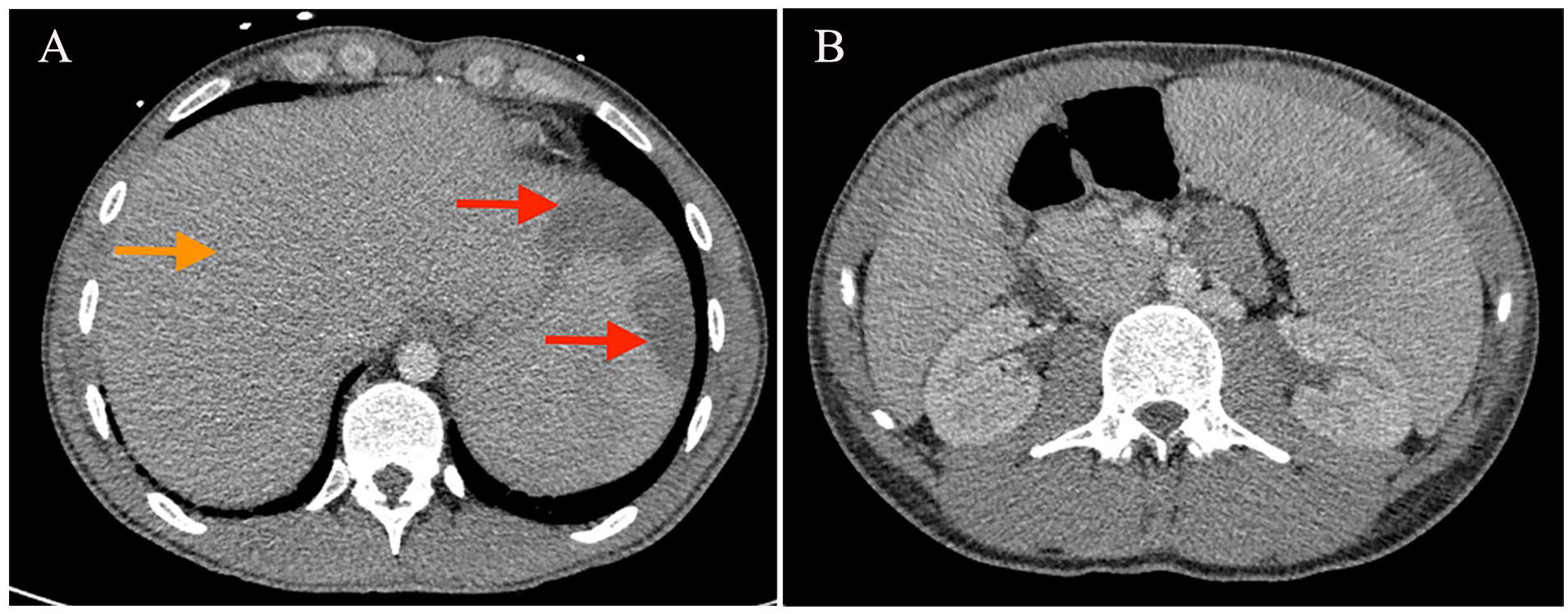
(A) CT abdomen and pelvis with IV contrast showing hepatosplenomegaly (orange arrow) and wedge-shaped infarcts (red arrows) favoring splenic infarcts and (B) CT abdomen and pelvis with IV contrast showing hepatosplenomegaly and wedge-shaped infarcts favoring splenic infarcts.
His vital signs on admission were blood pressure 124/45 mmHg, heart rate 105 beats per minute, respiratory rate of 26 breaths/minutes on room air, and temperature of 38.0°C. A grade 4 decrescendo diastolic murmur was auscultated on physical examination. Laboratory studies were remarkable for pancytopenia (white blood cell [WBC] 1800/mm3, hemoglobin 7.5 g/dL, platelet count 156 000/mm3, absolute neutrophil count [ANC] 900/mm3), creatinine 1.03 mg/dL, glucose 119 mg/dL, total bilirubin 0.6 mg/dL, aspartate transaminase 20 U/L, alanine transaminase 25 U/L, alkaline phosphatase 78 U/L, erythrocyte sedimentation rate 25 mm/h, c-reactive protein 4.382 mg/dL, ferritin 330.7 ng/mL, and lactate dehydrogenase 252 U/L, and he screened HIV-negative (Figure 2). Prior to induction chemotherapy, transthoracic echocardiography (TTE) was obtained and confirmed a bicuspid aortic valve (AV) with severe AV regurgitation likely due to AV mass (Figures 3 and 4). A follow-up transesophageal echocardiogram (TEE) displayed calcified echodensities on the AV and remnant aortic abscess (Figures 5 and 6). CT chest angiography showed heavy AV calcification and right hemithorax consolidation with scattered tree-in-bud and ground-glass opacities concerning for infection (Figure 7). Cardiothoracic surgery (CTS) was consulted and recommended to defer surgical repair until the patient completed chemotherapy for his malignancy given hemodynamic stability and lack of heart failure symptoms. Infectious diseases (ID) service was consulted on hospital day five due to negative blood cultures and echocardiogram findings.

Initial laboratory results.
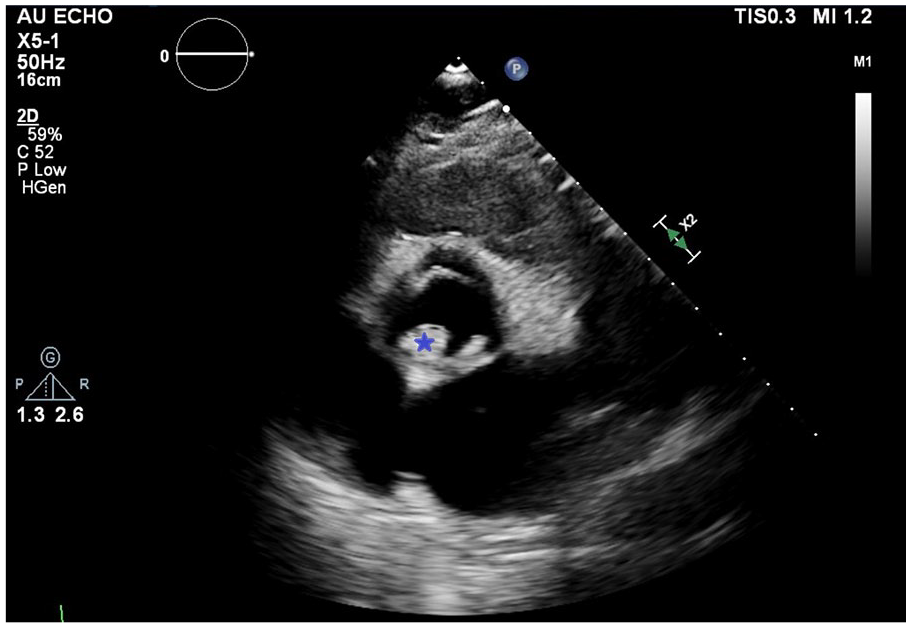
TTE parasternal short axis showing calcified echodensity (blue star) on the non/left coronary cusp of bicuspid AV.
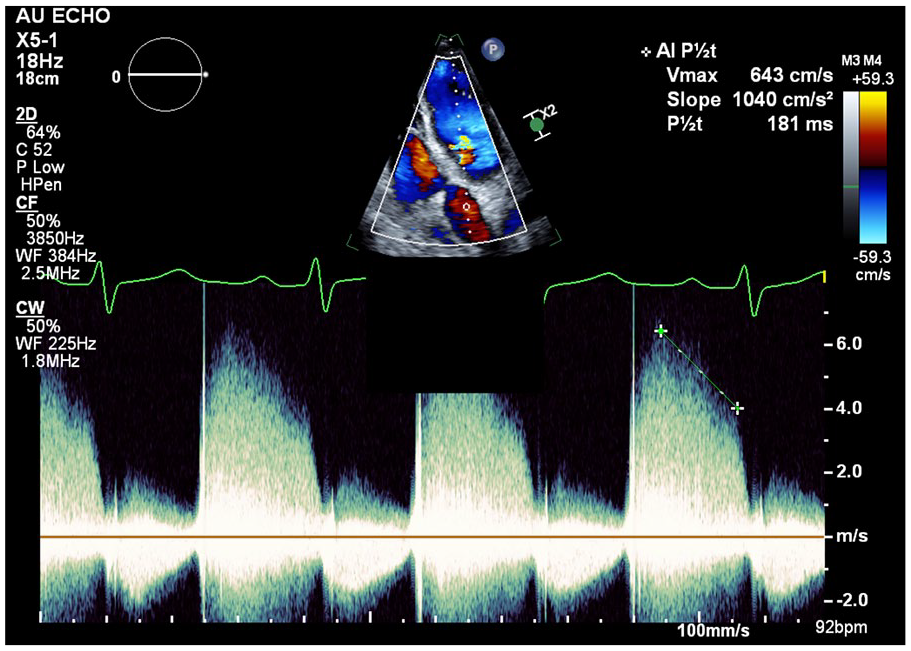
Pulsed wave Doppler of the AV showing severe aortic insufficiency with P1/2t 181 m/s.

TEE bi-plane demonstrating remnant periannular abscess (blue star) with Doppler flow.
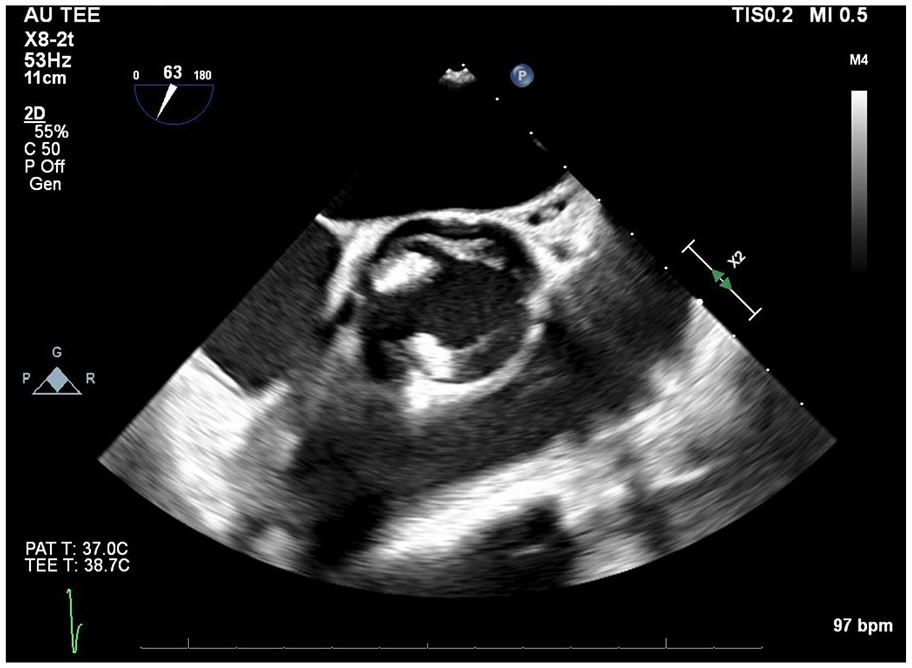
TEE bi-plane of the AV short axis showing echodensities and remnant periannular abscess.
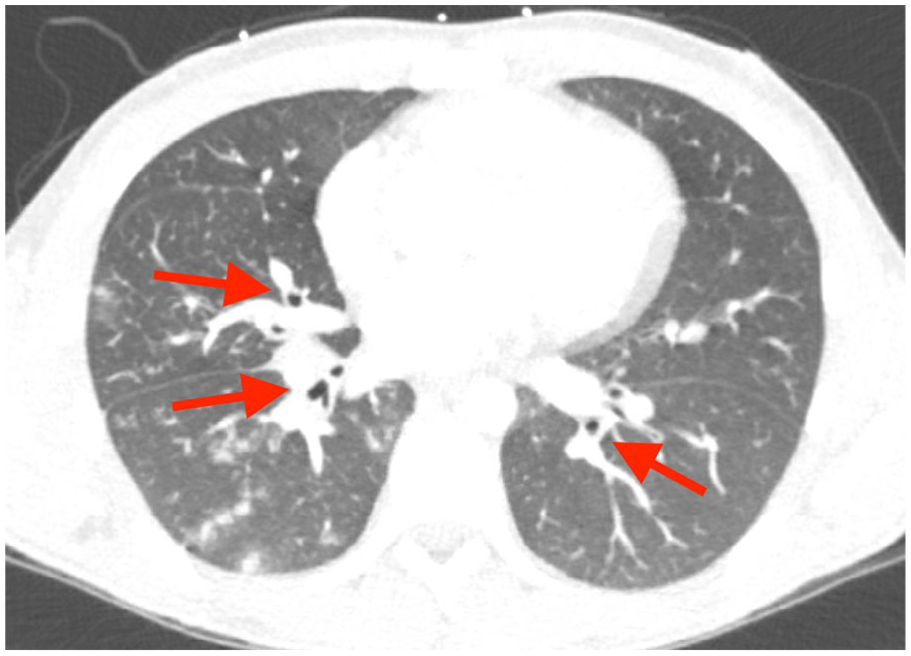
CT chest angiography showing scattered tree-in-bud and ground-glass opacities (red arrows) concerning for infection.
After further interview, the patient admitted to daily vaping and social alcohol use, but he denied engaging in high-risk sexual behavior, injection drug use, or smoking. He had no risk factors for Brucella or Coxiella given his lack of farm animal exposure or outdoor activities involving hunting, hiking, or camping. He reported having a dog and cat as indoor pets, and Bartonella serologies were ordered to rule out BCNE. Autoimmune workup revealed a positive rheumatoid factor 45.1 IU/mL and antinuclear antibody (ANA) titer 1:320; however, the rheumatology service deferred further therapy given the patient did not meet criteria for systemic lupus erythematosus or a specific connective tissue disorder. He underwent liver biopsy with interventional radiology for continued infectious and malignancy workup. He completed pre-phase chemotherapy for 3 days with concurrent prednisone along with empiric IV cefepime for 5 days while inpatient in the setting of neutropenic fever. He was discharged to follow up with his primary oncologist as an outpatient with oral levofloxacin for gram-negative prophylaxis prior to initiating future chemotherapy.
After discharge, serologic testing returned positive for IgG titer of 1:32,768 for B. henselae and IgG titer 1:256 titer for B. quintana while IgM was negative for both Bartonella species. His liver biopsy confirmed a diagnosis of hepatosplenic T-cell lymphoma (HSCTL), and liver tissue was negative for Bartonella PCR. Our patient met the diagnosis of B. henselae endocarditis given Bartonella IgG titer >1:800 and TEE findings of chronic aortic abscess. Three weeks later, he initiated his first cycle of ICE (ifosfamide, carboplatin, and etoposide) chemotherapy regimen and began antibiotic therapy for his Bartonella infection. The patient completed a 2-week course of oral rifampin 300 mg twice daily and 4-week course of oral doxycycline 100 mg twice daily to address his Bartonella-native valve endocarditis. Gentamicin was not incorporated into his treatment regimen given the risk of nephrotoxicity and drug-drug interactions with his chemotherapy. He finished his second cycle of ICE chemotherapy 1 month later, and his doxycycline was extended for additional 4 weeks as suppressive therapy by the inpatient ID team.
Per literature review, immunocompromised individuals (specifically patients living with HIV) benefit from daily suppression therapy with doxycycline or macrolide to prevent relapse after completing their primary treatment course. 8 The patient was re-evaluated in ID clinic after chemotherapy, and the decision was made to decrease doxycycline to daily dosing for suppression therapy until he could receive his surgical valve repair with CTS.
Unfortunately, the patient was readmitted with complaints of a severe frontal headache and unresponsiveness. He was emergently intubated in the ED given inability to protect his airway. Stat CT head showed a large left intraparenchymal hemorrhage with diffuse cerebral edema, midline shift, and impending herniation. He was taken urgently for decompressive hemicraniectomy and received osmotherapy postoperatively. Despite these efforts, his CT imaging worsened, and he had no brainstem reflexes or motor responses on physical exam. Apnea testing was conducted to confirm brain death, and the patient passed away. His family declined to pursue an autopsy to determine the etiology of his death; however, he had known risk factors for hemorrhagic stroke despite his young age including moderate thrombocytopenia (platelet count 51,000-100,000/mm3), the use of nonsteroidal anti-inflammatory drugs (NSAIDs), and advanced renal failure requiring initiation of hemodialysis. 9
Discussion
We present a case of BCNE caused by B. henselae. Patients with Bartonella endocarditis tend to have pre-existing valvular disease that promotes the development of IE. Typically, fevers are present (90%), vegetations are identified via echocardiography (90%), and >90% of patients require valvular surgery. 10 Bartonella endocarditis is characterized by an indolent course and frequently presents with nonspecific symptoms, underscoring the necessity for clinicians to maintain a high index of suspicion for prompt diagnosis and initiation of treatment. Individuals who have predisposing risk factors such as immunocompromised state, who inject drugs, and who are exposed to endemic areas may benefit from further serologic testing in their IE workup.
An IgG titer >1:800 is considered suggestive of Bartonella endocarditis; however, titers of <1:800 do not completely exclude the diagnosis.1,4,5,11 Bartonella antibody titers can be positive in asymptomatic and healthy individuals at a much lower concentration. Comparatively, cross-reactivity to IgG assays of other Bartonella spp., Chlamydia, and Coxiella is frequent, which reduces its specificity.1,3,11 Our patient’s IgM titers to B. henselae and B. quintana were negative; therefore, acute infection was less likely suspected. However, his IgG titers to B. quintana were concurrently positive with B. henselae and most likely due to cross-reactivity.
A delay in diagnosis and treatment of Bartonella infection can cause high mortality; and to date, no single treatment is effective for all Bartonella-associated diseases. 10 In the absence of systematic reviews, treatment guidelines are based on case reports that test a limited number of patients. Treatment of Bartonella endocarditis requires a combination of antimicrobials, and the American Heart Association guidelines currently recommend the use of combination therapy with an aminoglycoside (ie, gentamicin sulfate 3 mg/kg IV/intramuscular [IM] every 8 hours) and doxycycline 100 mg oral [PO]/IV every 12 hours.5,10 If gentamicin cannot be given, then it is replaced with rifampin 600 mg PO/IV every 24 hours.5,12 The standard of care is 2 weeks of gentamicin with concomitant 4 to 6 weeks of doxycycline. The duration of therapy for native valve endocarditis is at least 4 weeks and extended to 6 weeks in prosthetic valve endocarditis.5,12 A retrospective study reviewing 101 patients with Bartonella endocarditis found that patients receiving an aminoglycoside were more likely to recover. 13 Surgical valve repair is required for definitive cure as Bartonella endocarditis typically causes extensive valvular damage, aggravation of heart failure, and annular abscess. 3
As is commonly the case, this patient required a multidisciplinary approach to diagnosis with ID, cardiology, and CTS. In a case series of 48 patients with either B. henselae or B. quintana endocarditis, 10 were caused by B. henselae. Of these 10 patients, 9 required involvement of ID, cardiology, and CTS. 14 Our case specifically required the expertise of these departments with the addition of hematology-oncology given underlying HSCTL.
The patient’s Bartonella titers suggested a pattern indicative of past infection rather than an acute episode. Considering his upcoming chemotherapy and evaluation for bone marrow transplant, a treatment regimen consisting of doxycycline and rifampin was initiated. In addition, surgical intervention was postponed due to the patient’s operative risk, heightened by ongoing lymphoma treatment. This decision to delay surgery was made to minimize potential complications and optimize the patient’s overall prognosis.
This case underscores the diagnostic challenges and multidisciplinary therapeutic complexities associated with BCNE caused by Bartonella species, specifically B. henselae. Recognizing the indolent nature and nonspecific symptoms of Bartonella endocarditis is paramount for timely intervention especially in patients with predisposing risk factors. Serologic testing plays a crucial role in diagnosis; however, interpretation can be confounded by cross-reactivity and varying antibody titers. Treatment strategies, largely based on case reports (although with some guideline-driven strategies), emphasize combination antimicrobial therapy alongside surgical intervention for definitive management. Multidisciplinary collaboration among ID, cardiology, CTS, and in this case, hematology-oncology, can be essential for comprehensive patient care. The decision to postpone surgical intervention in favor of antimicrobial therapy reflects a nuanced approach considering the patient’s underlying lymphoma and operative risk, aiming to optimize prognosis amid ongoing treatment challenges.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published.
Prior Presentation of Abstract
This work was previously presented as a poster at the American College of Cardiology Annual Scientific Session in Atlanta, Georgia, on April 6, 2024.
