Abstract
Ventricular septal rupture, a formidable complication of acute myocardial infarction (AMI), is linked to significant morbidity and mortality. The clinical manifestation typically involves pronounced hemodynamic compromise necessitating prompt surgical intervention. This report outlines the case of a 60-year-old male presenting with acute heart failure 3 weeks post a presumed AMI. On evaluation, a substantial ventricular septal defect with left-to-right shunt was observed. The patient, although hemodynamically stable with mild symptoms, underwent surgical closure of the defect and coronary artery bypass graft for multivessel coronary artery disease. This case contributes to the literature on the delayed presentation of post-myocardial infarction (MI) ventricular septal rupture, a scenario deviating from the anticipated severe hemodynamic instability given the timing of the MI and the extent of the septal defect.
Introduction
Post-infarction ventricular septal rupture (PIVSR) represents a rare yet grave complication arising from acute myocardial infarction (AMI), accompanied by substantial morbidity and mortality. The incidence of ventricular septal rupture (VSR) after ST-segment elevation myocardial infarction (STEMI) has witnessed a decline, diminishing from 1% to 3% in the pre-perfusion era to a range of 0.17% to 0.31% following primary percutaneous coronary intervention. 1 The manifestation of PIVSR is contingent upon the pathophysiological characteristics of the AMI, the implicated coronary artery, and the temporal proximity to the index AMI.
The presentation timing of PIVSR exhibits a bimodal distribution, with most cases emerging within hours or 3 to 5 days following the onset of AMI. 2 Instances of presentation beyond several weeks after the index AMI are exceedingly rare.3,4 Patients may present with varying degrees of severity, encompassing mild hemodynamic instability in the early stages, which can swiftly escalate to cardiogenic shock if left unattended. The therapeutic approach entails surgical closure, achieved through either open thoracotomy combined with concurrent coronary artery bypass grafting (CABG) or transcatheter repair, the latter being employed for small defects or as a salvage intervention for defects inadequately addressed via surgical means.
This report highlights the rare case of a patient who presented with PIVSR, 3 weeks after the initial AMI, demonstrating a state of relative hemodynamic stability and underwent surgical closure accompanied by CABG.
Case
A 60-year-old male with no past medical history, not on any medications, and who had not seen a health care provider in over 30 years presented to the emergency department with worsening dyspnea and lower limb edema. He reported that approximately 3 weeks prior, he had an episode of crushing substernal chest pain, palpitations, and diaphoresis while mowing his lawn. His chest pain had resolved spontaneously in a few hours’ time, and he deferred seeking medical care at that time. However, over the next several days, he suffered progressively worsening shortness of breath, orthopnea, and lower limb swelling, which prompted him to seek medical attention at our institute. He reported current tobacco use with a 45, pack-year smoking histor,y along with heavy alcohol use until about a year ago, and no illicit drug use. He had a family history of coronary artery disease in his father, who underwent a CABG at the age of 60 years.
On presentation, he was afebrile with a blood pressure of 111/89 mm Hg, pulse rate of 110/min, respiratory rate of 18/min, and was saturating 91% on room air. A neck exam was notable for a visible jugular venous distention with a height of 10 cm, and cardiopulmonary auscultation revealed the presence of S3 murmur along with a harsh holosystolic murmur on the left sternal border with a palpable thrill, decreased breath sounds on the bilateral lung bases, and diffuse crackles bilaterally. Radial pulses were equal on both sides, with 2+ bilateral pitting edema to the knees. Labs on presentation showed an unremarkable complete blood count, complete metabolic panel, lipid profile, and hemoglobin A1c. The patient had an elevated NT-pro BNP of 7575 pg/mL (normal = 0-125 pg/mL), high-sensitivity troponin of 40 ng/L (normal <19 ng/L), and D-dimer of 1.27 µg/mL (normal <0.6 µg/mL). An electrocardiogram (EKG) showed sinus tachycardia with evidence of deep Q waves in the anterior and inferior leads without ST-segment changes. Chest X-ray revealed pulmonary venous congestion with moderately sized pleural effusions bilaterally. He underwent a computed tomography (CT) pulmonary angiogram, which did not show any evidence of pulmonary emboli but did confirm bilateral pleural effusions and interstitial edema. The patient was started on a heparin drip and started on diuresis with furosemide. Transthoracic echocardiogram (TTE) revealed ejection fraction (EF) of 50% to 55%, pseudo normal pattern of left ventricular (LV) diastolic filling (grade 2), mildly enlarged right ventricle (RV) and hyperdynamic global RV systolic function, pulmonary artery pressure of 39 mm Hg (normal <35 mm Hg), and large muscular ventricular septal defect (VSD) in the distal septum with left to right shunt (Qp:Qs 3.56). A transesophageal echocardiogram was performed which showed a similarly described VSD (Figure 1). He subsequently underwent a right and left heart catheterization, which showed 99% mid-left anterior descending (LAD) artery stenosis with a thrombotic lesion and 100% right coronary artery (RCA) stenosis with visualized collaterals along with a step up in oxygen saturation from 56% in the right atrium to 74% in the RV (Figures 2-4). Owing to multivessel coronary artery disease complicated by a VSD, the patient was transferred to a comprehensive surgical center to undergo further management. He was evaluated by the surgical team and a decision was made to implant mechanical circulatory support (MCS) in the form of an Intra-Aortic Balloon Pump (IABP) as a bridge to surgical VSD closure and CABG. However, the IABP ruptured within 2 hours of implantation and repeat implantation was aborted. The patient subsequently underwent double-patch closure of VSD (Figure 5) as described in the literature by Tabuchi et al, 5 exclusion of LV pseudoaneurysm, and CABG with saphenous vein graft to the first diagonal branch. His post-operative course was complicated by atrial fibrillation and right-sided pneumothorax. Post-operative TTE showed an EF of 30%, successful double-patch closure, and no interventricular blood flow (Figure 6). The patient improved clinically over the course of the following week and was discharged home on appropriate medical therapy along with a wearable defibrillator for the prevention of sudden cardiac death. On the 1-month follow-up, he reported feeling weak but denied any chest pain or shortness of breath. On the 6-month follow-up, he reported further improvement in his symptoms and improvement in his exercise tolerance. The echocardiogram at that follow-up showed improvement of his EF to 38% and a large LV thrombus measuring 33.9 mm by 13.3 mm. His life-vest was removed; anticoagulation was initiated with coumadin after bridging with apixaban.

Transesophageal echocardiogram showing the ventricular septal defect (arrow) in deep transgastric short-axis view (1) and mid-esophageal 4-chamber view (2) (LV: left ventricle; RV: right ventricle).
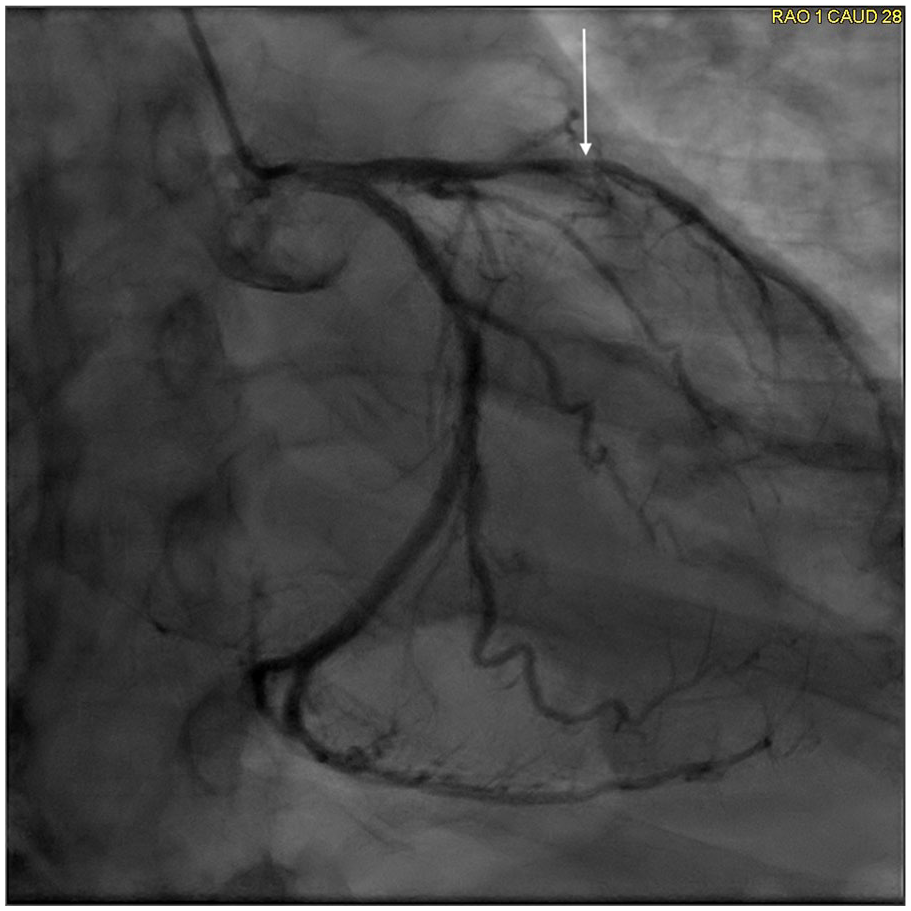
Anterior-posterior caudal view showing patent left main and left circumflex arteries with severe mid-left anterior descending artery disease (arrow).

Right anterior oblique cranial view showing severe subtotal occlusion of the left anterior descending artery with filling defect consistent with thrombotic occlusion (arrow). Left to right collaterals are visualized as well.

Left anterior oblique view showing chronic total occlusion of right coronary artery (arrow).

The double-patch technique for ventricular septal perforation shown in a heart cross-section (LV: left ventricle; RV: right ventricle).

Apical 4-chamber view of transthoracic echocardiogram showing the area of double-patch repair (arrows) of the ventricular septal defect (1) with the Doppler image showing no blood flow through the interventricular septum (2) (LV: left ventricle; RV: right ventricle).
Discussion
The patient presented following an episode of classical cardiac chest pain, indicative of what we identify as his primary AMI. Regrettably, timely reperfusion therapy was not administered. The presence of predisposing factors associated with an elevated propensity for PIVSR include advanced age, female gender, initial occurrence of AMI, anterior AMI, and delayed reperfusion, 6 several of which manifested in our patient.
Upon presentation, the EKG exhibited telltale signs of an infarct in both the anterior and inferior territories, marked by profound Q waves in V1-V4 and in leads 3 and AVF. The LAD and RCA emerge as the predominant infarct-related arteries contributing to PIVSR, constituting 42% and 46% of all instances, respectively, as per data from the SHould we emergently revascularize Occluded Coronaries in cardiogenic shocK (SHOCK) registry. 7 In our patient, left heart catheterization unveiled a chronic total occlusion in both the LAD and RCA, underscoring the severity of the ischemic insult.
The customary temporal trajectory for PIVSR is typically confined to a span of a few hours or within 3 to 5 days after the index AMI. 1 Instances of presentation beyond this timeframe are exceedingly uncommon. The investigations of the SHOCK registry and the APEX MI trial have delineated the intervals for the identification of PIVSR, reporting durations of 16 and 7.7 hours, respectively.6,8 In our case, the precise temporal evolution of the VSR subsequent to the index AMI remains elusive, as it took only a few weeks of progressively exacerbating dyspnea and lower limb edema to prompt the patient to seek medical attention. We posit that the robust physical conditioning of our patient, a farmer by profession, coupled with the absence of other medical comorbidities, potentially augmented his resilience to angina and hemodynamic decompensation.
Crucially, in the diagnostic landscape, the presence of a systolic murmur may be the sole clinical clue to the impending diagnosis of PIVSR. However, it is imperative to underscore that this seemingly benign murmur can precipitously evolve into acute heart failure and cardiogenic shock. Hence, the prompt identification of this clinical entity assumes paramount significance. To our knowledge, only a solitary report parallels our patient’s scenario—a late presenting PIVSR, 3 weeks post-AMI, characterized by a state of relative hemodynamic stability. 4
The Becker classification of free wall rupture usually correlates with clinical presentation. Becker type 3 ruptures occur from the perforation of a thinned aneurysmal myocardium in the late phase of an MI, especially in the setting of delayed reperfusion therapy. Intraoperatively, our patient was noted to have a left ventricular pseudoaneurysm with a large anterior VSD, which seems to be consistent with Becker type 3 rupture owing to his delayed presentation and multi-territory infarct. Becker type 1 is slit-like tears in the myocardium occurring within 24 hours of AMI, and Becker type 2 is slowly progressive tears secondary to coagulation necrosis.
Surgical intervention stands as the definitive course of action for the treatment of PIVSR; however, the optimal timing of such surgical measures remains a topic of substantial debate within the medical community. Arnaoutakis et al 9 conducted a comprehensive study involving 2876 patients who underwent operative repair for PIVSR. Their findings revealed an operative mortality rate of 54.1% when surgery was performed within the initial 7 days, in stark contrast to the markedly reduced mortality rate of 18.4% observed when surgery was delayed beyond this temporal threshold. This underscores the negative predictive impact of early surgical intervention on overall outcomes.
Although the 2013 American College of Cardiology/American Heart Association (ACC/AHA) guidelines 10 advocate for immediate surgical closure irrespective of hemodynamic status, the European Guidelines 11 adopt a contrasting stance, endorsing a delayed repair strategy. This delayed approach is posited to facilitate myocardial recovery from ischemia and allows for the transformation of necrotic, friable tissue into a fibrotic, more resilient scar—thereby rendering it more amenable to durable repair. In our patient’s case, the decision to pursue delayed repair was influenced predominantly by the delayed presentation. Remarkably, the post-operative course yielded favorable outcomes, as evidenced by the amelioration of functional status noted at the 1-month follow-up assessment. This outcome further underscores the nuanced consideration required in determining the optimal timing for surgical intervention in PIVSR cases.
Mechanical circulatory support has been hypothesized as a potential avenue for hemodynamic stabilization in individuals with PIVSR. However, the interaction of MCS with the unique physiological dynamics of VSR remains complex, and as such, its definitive role in this context has not been fully elucidated. A systematic review, encompassing studies exploring MCS in the pre-operative setting related to VSR, identified the IABP as the most commonly used MCS device. 8 Notably, the IABP exhibited the fewest complications in this setting. 12 The frequency of IABP rupture and entrapment is rare and has been shown to range from 1% to 1.7% based on cohort studies and registry data.13,14 In the case of our patient, IABP was strategically implanted to furnish supplementary hemodynamic support during the surgical intervention. Importantly, this pre-emptive measure was implemented not in response to impending hemodynamic collapse or cardiogenic shock, but rather to fortify the patient’s cardiovascular status during the surgical procedure. Although the precise cause of rupture remains uncertain in our patient’s case, plausible explanations include rupture secondary to abrasion over an aortic plaque or potential issues in manufacturing. It is noteworthy that despite the rupture of the IABP in our patient, the decision against repeat pre-operative implantation was made due to the inherent risks associated with such a maneuver. Consequently, the patient successfully underwent surgical intervention without the utilization of MCS, underscoring the meticulous consideration and risk-benefit assessment inherent in the management of VSR cases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
No previous presentations.
