Abstract
Cutaneous paraneoplastic syndromes due to Hodgkin lymphoma present with a wide spectrum of clinical manifestations from generalized pruritus to exfoliative erythroderma. We summarize the clinical findings and outcomes of 14 patients with Hodgkin lymphoma and associated cutaneous paraneoplastic syndromes treated at Mayo Clinic over the past 3 decades. Cutaneous paraneoplastic syndromes may be present at the time of lymphoma diagnosis, whereas in other patients, it may appear at the time of relapse, including patients with initial absence of cutaneous manifestations during the initial lymphoma presentation. Our results indicate that complete resolution of the paraneoplastic syndrome is associated with significantly improved overall survival. Recognition of cutaneous paraneoplastic syndromes is a crucial surrogate of relapsed malignancy and treatment requires targeting the underlying malignancy.
Background
Cutaneous paraneoplastic syndromes (PS) due to Hodgkin lymphoma (HL) present with a wide spectrum of clinical manifestations, including paraneoplastic pemphigus (PNP), pyoderma gangrenosum, Sweet’s Syndrome, and others. Prior literature suggests that nodular sclerosing HL subtype is the predominant subtype displaying cutaneous disease. 1 New-onset dermatitis or eczema may precede HL diagnosis by up to 6 months and may be accompanied by xerosis, lichenoid changes, and excoriations that resolve with systemic therapy. 2 Chronic pruritus longer than 6 weeks has been reported in up to 30% of patients with HL and is considered a PS. 2 Herein, we summarize the clinical findings and outcomes of 14 patients with HL with associated cutaneous PS.
Methods
The medical charts of 335 adult patients were reviewed from an initial database of patients with lymphoma and suspected PS seen at Mayo Clinic between 1994 and 2022. Of the 335 patients, 33 patients had confirmed HL with cutaneous manifestations. Patients under the age of 18 years and patients with multiple active malignancies were excluded. Patients who had pre-existing dermatologic conditions with suspected exacerbation at HL onset were excluded. Several patients were also excluded for common cutaneous drug reactions. Given that no formal diagnostic criteria exist across the spectrum of cutaneous PS, Bradford Hill criteria were assessed in the remaining patients for clinical diagnosis of PS, including strength of effect, specificity, temporality, plausibility, and reversibility. 2 Only patients with at least three of these criteria were included in the study. Patients meeting all inclusion criteria were retrospectively analyzed based on common endpoints and clinical features, including age, sex, race, HL subtype, HL stage at diagnosis, bulky disease, clinical PS syndrome, first-line chemotherapy, and surgical or radiation therapies. Overall survival was defined as the time from HL diagnosis to death. Overall survival probabilities were calculated using Kaplan-Meier estimates. Statistical analysis was performed using BlueSky Statistics software packages.
Results
Inclusion criteria were met in 14 patients with HL and cutaneous PS. Clinical and pathologic features of each case are described in Table 1. The clinical spectrum of PS included generalized pruritus/dermatitis (11), exfoliative erythroderma (1), granulomatous dermatitis (1), and dermatomyositis (DM) (1). Baseline demographics of our study population included 57% female and 93% White patients with median age at HL diagnosis of 72 years (range, 22-83 years). Eleven patients had classical HL, whereas 3 patients had nodular lymphocyte-predominate HL. Median follow-up time from HL diagnosis was 38.3 (range: 3.8-343.4) months.
Clinical and Pathologic Features in Patients With Persistent Versus Resolved Cutaneous Paraneoplastic Syndrome.

Abbreviations: x—Dermatopathology not available for review.
Paraneoplastic syndrome was present in 8 patients at the time of HL diagnosis, whereas 5 patients had PS at the time of relapse, including 3 patients who did not have PS at initial HL presentation. Eleven patients experienced complete resolution of PS, while 3 had no resolution of cutaneous PS with HL treatment. The median time to PS resolution was 3.8 (range: 0.5-17.6) months from the time of HL treatment initiation. Of the 11 patients who experienced complete symptomatic resolution, 3 experienced cutaneous PS relapse, which was temporally linked with malignant relapse. The three patients who had persistent PS failed to improve due to treatment failure and/or relapse of disease. The median time to PS relapse was 16.9 (range: 8.5-25.6) months.
Four patients were deceased at the time of review. All deaths were attributed to progression of disease rather than complications of treatment or PS. Median overall survival (OS) was significantly lower in patients without complete resolution of PS (29.5 months) versus complete resolution with median OS not reached (

Kaplan-Meier estimate of overall survival in patients with persistent versus resolved cutaneous paraneoplastic syndrome.
Four patients had EBV-data available. Two patients had polymerase chain reaction (PCR)-confirmed Epstein-Barr viremia at the time of diagnosis of malignancy and PS, whereas the other two patients were IgM-negative and IgG-positive, consistent with prior exposure.
Discussion
Cutaneous involvement of HL is rare and unlikely to be found on pathologic examination from skin biopsy. 3 Most cases of cutaneous PS in our series manifested with generalized pruritus and/or dermatitis. Owing to skin barrier disruption and subsequent inflammation secondary to itching, clinical findings of dermatitis may be partially explained as a consequence of chronic pruritis, a well-recognized paraneoplastic manifestation of HL. 4 The mechanism of paraneoplastic pruritus in the setting of underlying malignancy is likely multifactorial, including hypersensitivity to tumor-specific antigens, eosinophilia, and production of histamine and other chemical mediators.1,5,6
One patient specifically presented with infiltrative smooth plaques that showed pan-dermal interstitial granulomatous dermatitis on histopathology with a clinical image shown in Figure 2. Prior literature has shown that various patterns of granulomas inflammation, including sarcoidal, palisaded, and tuberculoid-like, may be found in both Hodgkin and non-HLs, and these findings may serve as a sign of an underlying lymphoma in the absence of an alternative explanation. 7 Our findings suggest that these reactive noninfectious granulomas may arise as a PS associated with HL.

Ill-defined hyperpigmented, erythematous infiltrative plaque on the left arm, consistent with granulomatous dermatitis.
Exfoliative erythroderma is most commonly seen in the setting of severe spongiotic dermatitis or psoriasis. 8 The patient with erythroderma in this series demonstrated severe lichenoid interface dermatitis on histopathology with a clinical image shown in Figure 3. This pattern may by seen with PNP and, when accompanied by eosinophils, as manifestation of a drug eruption. However, the absence of stomatitis clinically excluded a diagnosis of PNP, and there were no new medication exposures temporally associated with this eruption. On occasion, interface and lichenoid dermatitis may be a paraneoplastic effect secondary to antigen cross-reactivity between tumor and self-antigen, which we posit is the most likely explanation in this case. 9

Confluent macular erythema with overlying exfoliative scale and notable absence of mucositis on the labial mucosa, consistent with exfoliative erythroderma.
In our study, patients with clinically apparent dermatitis often had histopathology demonstrating features indistinguishable from those seen in conventional spongiotic dermatitis. Acute eczematous papules and patches may present with crusting and vesiculation as seen in Figure 4. Although topical and oral steroids are often implemented in the treatment of dermatitis, consideration should be given to the possibility of cutaneous PS when skin disease remains refractory to these measures. Thus, treatment should be focused primarily on treatment of HL when cutaneous PS is suspected.
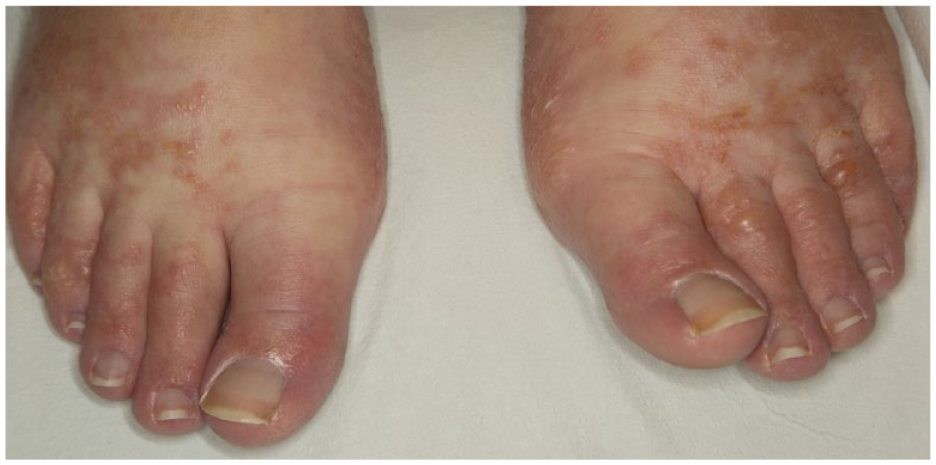
Acute eczematous papules and patches with vesiculation and crusting on bilateral feet.
Dermatomyositis occurs in the setting of internal malignancy in approximately 20% of patients, with the most common underlying malignancies being gastrointestinal, lung, breast, ovary, prostate, and non-HL. 10 Paraneoplastic DM due to underlying HL is rare. 11 One patient in our study demonstrated clinical signs of DM, including proximal myopathy, elevated creatine kinase, positive antinuclear antibody, macular erythema on dorsal fingers (Gottron’s sign), hand xerosis, and nailfold abnormalities including periungual telangiectasias. These findings were present at the time of HL diagnosis and resolved with treatment of the underlying HL, suggesting a paraneoplastic etiology.
In our series of 14 patients with PS secondary to HL, there was a significant improvement in OS associated with complete resolution of PS. Given that all mortalities resulted from progression of disease rather than PS, refractory PS represents a surrogate of refractory HL in this patient population, explaining the difference in survival between groups. Given that dermatologic complaints in HL are commonly seen following systemic chemotherapy, radiation therapy, and graft-versus-host disease following allogeneic hematopoietic stem cell transplantation, recognition of cutaneous PS is important as these clinical features may be the presenting sign of relapsed lymphoma rather than classical features such as palpable lymphadenopathy, unexpected weight loss, or night sweats. Furthermore, recognition of cutaneous PS is essential in providing optimal therapy, which requires effectively targeting the underlying malignancy, as evidenced by prior literature and our series of 14 patients who only achieved complete resolution of PS with remission of HL.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Informed consent for patient information to be published in this article was not obtained because all patient health information was deidentified for the 14 patients included in the study.
