Abstract
Gastric cancer ranks as the fifth leading cause of global cancer incidences, exhibiting varied prevalence influenced by geographical, ethnic, and lifestyle factors, as well as Helicobacter pylori infection. The ATM gene on chromosome 11q22 is vital for genomic stability as an initiator of the DNA damage response, and mutations in this gene have been associated with various cancers. Poly ADP-ribose polymerase (PARP) inhibitors, such as olaparib, have shown efficacy in cancers with homologous recombination repair deficiencies, notably in those with ATM mutations. Here, we present a case of a 66-year-old patient with germline ATM-mutated metastatic gastric cancer with very high CA 19-9 (48 000 units/mL) who demonstrated an exceptional response to the addition of olaparib to chemo-immunotherapy and subsequent olaparib maintenance monotherapy for 12 months. CA 19-9 was maintained at low level for 18 months. Despite the failure of a phase II clinical trial on olaparib in gastric cancer (NCT01063517) to meet its primary endpoint, intriguing findings emerged in the subset of ATM-mutated patients, who exhibited notable improvements in overall survival. Our case underscores the potential clinical utility of olaparib in germline ATM-mutated gastric cancer and emphasizes the need for further exploration through larger clinical trials. Ongoing research and clinical trials are essential for optimizing the use of PARP inhibitors, identifying biomarkers, and advancing personalized treatment strategies for gastric cancer.
Introduction
Gastric cancer, also called gastric adenocarcinoma, is the fifth leading cause of cancers worldwide, and its incidence is influenced by factors such as geographic location, ethnicity, lifestyle, and infection with H pylori bacteria. 1 In general, Eastern Asia (including countries like Japan, South Korea, and China), Eastern Europe, and the Andean regions of South America have higher incidence rates of gastric cancer than other regions. 2 In the United States, about 26 500 new cases are diagnosed annually; approximately 11 130 people died from gastric cancer the United States in 2023. 3
The ATM gene (ataxia telangiectasia mutated), located in chromosome 11q22, plays a crucial role in maintaining genomic stability as an initiator of the DNA damage response. This process is essential for preventing the accumulation of genetic mutations that could lead to the development of cancer. 4 Heterozygous germline mutations in ATM and sporadic ATM mutations have been identified in various types of cancer, including breast cancer, pancreatic cancer, lung cancer, and a wide range of other solid tumors. 5
PARP (poly ADP-ribose polymerase) inhibitors are a class of drugs that have shown effectiveness in the treatment of certain cancers, particularly those with specific DNA repair deficiencies such as in homologous recombination repair (HRR).6-12 One such context where PARP inhibitors have demonstrated efficacy is in cancers with mutations in the ATM gene. ATM is involved in the repair of DNA double-strand breaks (DSBs). ATM gene mutation, as is the case in some cancers, can lead to impaired DNA repair mechanisms. PARP inhibitors take advantage of this situation by exploiting a concept known as synthetic lethality. 13 Cancers with mutations in ATM are thus considered more sensitive to PARP inhibitors due to this synthetic lethality. Breast and ovarian cancers with BRCA mutations are another well-studied example of interactions with PARP inhibitors leading to synthetic lethality. 5
Despite the promising benefits of the use of PARP inhibitors in prostate, breast, and ovarian cancers with HRR deficiency, particularly ATM mutations, the exact roles and benefits of PARP inhibitors in gastric cancer patients with ATM mutations are not well studied. Herein, we report a case of a patient with germline ATM-mutated metastatic gastric cancer who demonstrated an exceptional response to olaparib combined with platinum chemoimmunotherapy followed by olaparib maintenance. This case highlights the potential roles of PARP inhibitors in ATM-mutated gastric cancer.
Case Presentation
A 66-year-old gentleman with significant family history of gastric cancer in his sister and brother initially presented with worsening epigastric pain and reflux disease in February 2022. He underwent esophagogastroduodenoscopy (EGD) which revealed an infiltrative and ulcerated bleeding mass with malignant appearance at the incisura (the lesser curvature of stomach near pylorus) (Figure 1). The initial CA 19-9 count was 48 000 units/mL. Staging CT of the chest, abdomen, and pelvis showed peritoneal carcinomatosis (Figure 2).

EGD which revealed an infiltrative and ulcerated bleeding mass with malignant appearance at the incisura (the lesser curvature of stomach near pylorus).

Initial staging CT of the chest, abdomen, and pelvis showed peritoneal carcinomatosis.
Laparoscopy was not performed because the CT of the abdomen pelvis showed peritoneal carcinomatosis. Pathology from the stomach biopsy showed invasive adenocarcinoma with signet ring features, an Alcian blue/PAS stain highlighted adenocarcinoma, and Giemsa statin was negative for H pylori, mismatch repair (MMR) proficient, HER2 negative, and PDL-1 was 15%. In light of the patient’s family history of gastric cancer, he underwent germline mutation testing by Invitae which was positive for an ATM c6415G>T heterozygous pathogenic mutation (Figure 3). The patient had good performance status, with a Karnofsky performance score ≥60%.

Germline genetic testing by Invitae showing a pathogenic mutation of the ATM gene.
The patient was initially started on fluorouracil, leucovorin, oxaliplatin, and docetaxel in February 2022; nivolumab was added in March 2022. He demonstrated a positive response, including an improvement of the CA 19-9 level, which decreased from 48 000 to 16 units/mL within 4 months of initiation of chemoimmunotherapy. Nonetheless, patient was not able to tolerate the intensive chemotherapy regimen, oxaliplatin was discontinued in August 2022, and patient remained on nivolumab with fluorouracil and docetaxel for another 4 to 5 months. Because of the patient’s germline ATM mutation, olaparib 300 mg orally twice daily was added to his chemoimmunotherapy regimen in July 2022. The patient continued on this triple therapy regimen from July 2022 to November 2022, tolerating it well, with CA 19-9 remaining low (<34 units/mL). Nonetheless, at the end of November 2022, after about 14 doses of nivolumab, the patient developed grade 3 adverse immunotherapy events, including pneumonitis that required oxygen and hospitalization. Thus, chemoimmunotherapy was discontinued in November 2022.
In light of the patient’s poor performance status and immunotherapy adverse event, therapy was transitioned to maintenance olaparib monotherapy at 300 mg orally twice daily. The patient continued to have a great response until December 2023 (Figure 4). Surveillance EGDs in June 2023 and October 2023, performed by 2 different gastroenterologists, both showed normal gastric mucosa and disappearance of the previously seen tumor. Random biopsy tissue was negative for malignancy (Figure 5). In December 2023, after 1 year of olaparib monotherapy, the patient had a mild elevation of CA 19-9 to 45 units/mL and a restaging CT scan of the chest, abdomen, and pelvis showed reappearance of the peritoneal ground-glass opacities, suggestive of a recurrence of the peritoneal metastasis. A subsequent restaging positron emission tomography/computed tomography (PET/CT) scan showed multiple fluorodeoxyglucose (FDG)-avid peritoneal nodules consistent with carcinomatosis and linear increased FDG uptake in a portion of right pleural thickening that was suspicious for cancer involvement (Figure 6).
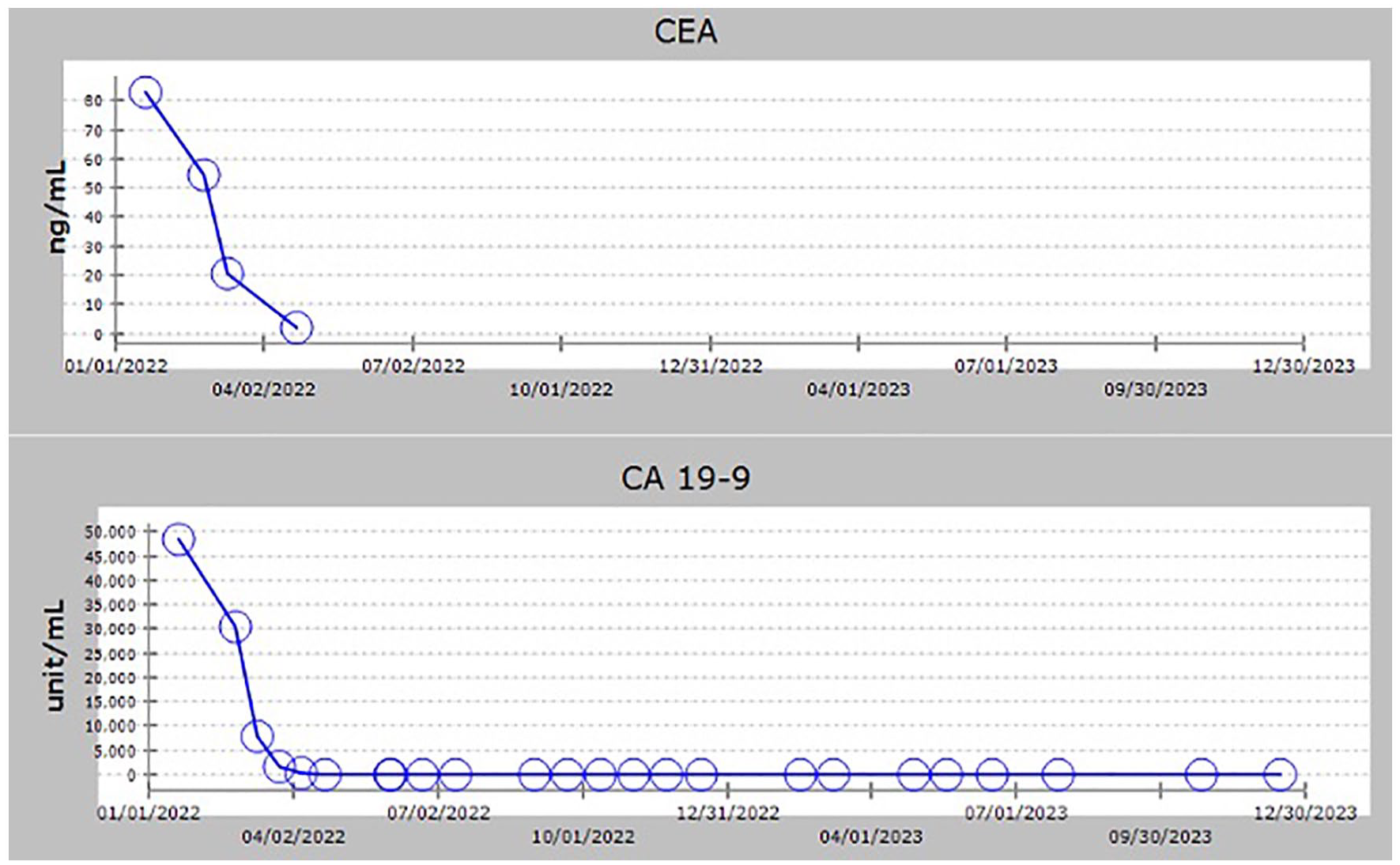
Trends of CEA and CA 19-9 from the initial diagnosis to recent follow-up.

Surveillance EGD showed normal gastric mucosa and disappearance of the previously seen tumor.

After 1 year (1/2023-1/2024) on olaparib monotherapy, there is a mild recurrence of nodular and ground-glass opacity peritoneal metastases on PET/CT, and CA19-9 increased from 13 to 45 units/mL.
Discussion
Gastric cancer frequently exhibits deficiencies in HRR. Homologous recombination repair is a DNA repair pathway that acts on DNA DSBs and interstrand cross-links. ATM is 1 of 15 HRR-related genes: BRCA1, BRCA2, ATM, BARD1, BRIP1, CDK12, CHEK1, CHEK2, FANCL, PALB2, PPP2R2A, RAD51B, RAD51C, RAD51D, and RAD54L.14,15 ATM is located in chromosome 11q22-23 and is a critical gene involved in the repair of damaged DNA, 16 as well as playing a crucial role in maintaining genomic stability by responding to DNA damage. When DNA damage occurs, the ATM protein is activated and initiates a signaling cascade that helps repair the damaged DNA or, if necessary, induces cell cycle arrest to prevent the replication of damaged DNA. This process is essential for preventing the accumulation of genetic mutations that could lead to the development of cancer. 4 In individuals with ataxia telangiectasia, mutations in both copies of ATM are present (homozygous mutations), resulting in a lack of functional ATM protein. As a consequence, these individuals have impaired DNA repair mechanisms, making them more susceptible to the accumulation of DNA damage and conferring an increased risk of cancer. 17 Heterozygous mutations in the germline ATM gene and sporadic ATM mutation have been identified in various types of cancer, including breast cancer, pancreatic cancer, lung cancer, and a wide range of other solid tumors. 5
PARP enzymes—PARP1, PARP2, and PARP3—play a critical role in cell survival and replication. They are involved in multiple cellular processes including DNA transcription, cell cycle regulation, and DNA repair. 18 PARP inhibitors hinder the repair of single-stranded breaks (SSBs) in DNA and facilitate the transformation of SSB into DSBs. This induces synthetic lethality in cancer cells lacking an efficient DSB mechanism, such as HRR. Consequently, individuals with HRR deficiency, such as those with an ATM mutation, stand to gain from the use of PARP inhibitors.6-12
Olaparib is a potent oral PARP inhibitor which causes synthetic lethality in BRCA1/2- and HRR-deficient tumor cells through the formation of cumulative DSBs which cannot be repaired, leading to disruption of cellular homeostasis and cell death of tumor cells.6-12 Olaparib has demonstrated promising responses as maintenance therapy in patients with breast, prostate, ovarian, and pancreatic cancer, and its efficacy seems more marked in patients with BRCA mutations and HRR-deficient cancers. Despite the widely observed HRR deficiency seen in gastric cancer, the clinical role of PARP inhibitors in gastric cancers remains to be determined.
The use of olaparib in the context of ATM-mutated gastric cancer represents a significant area of interest in oncology research. In a double-blind, randomized phase II clinical trial (NCT01063517), the combination of olaparib and paclitaxel in patients with recurrent or metastatic gastric cancer, particularly those with low ATM levels, did not achieve the primary endpoint of progression-free survival. Although the study did not meet its primary endpoint of progression-free survival in the overall population, intriguing findings emerged in the subset of patients with ATM mutations. Notably, the combination exhibited a notable improvement in overall survival over paclitaxel plus placebo (hazard ratio, 0.74; median progression-free survival, 5.29 vs 3.68 months). 19 In the phase III GOLD trial (NCT01924533), the combination of olaparib and paclitaxel was again failed to reach its primary endpoint with no statistical significance difference in overall survival. Overall survival were comparable across treatment groups both in the overall patient cohort regardless of ATM mutation status (with a median overall survival of 8.8 months [95% confidence interval (CI): 7.4-9.6] in the olaparib group versus 6.9 months [6.3-7.9] in the placebo group; hazard ratio [HR] 0.79 [97.5% CI: 0.63-1.00]; P = .026) and among patients with ATM negative (12.0 months [7.8-18.1] versus 10.0 months [6.4-13.3]; HR 0.73 [0.40-1.34]; P = .25). Nonetheless, in that trial, ATM-mutated patients treated with olaparib exhibited significantly improved remission rates compared to ATM-negative patients treated with paclitaxel alone (overall response rate, 4.24; P = .0309). This suggests that PARP inhibitors may have a potential role in ATM-mutated patients with gastric cancer (Table 1).20,21
Summary of Randomized Clinical Trial Result of Both NCT01063517 and GOLD Clinical Trials.

IV = intravenous; PFS = progression-free survival; OS = overall survival; Ref. = Reference; HR = hazard ratio; OR = odds ratio.
Olaparib is very well tolerated with minimal side effects. An aggregated examination of prior trials reveals that neutropenia and anemia are the predominant side effects. The intensity of neutropenia may be heightened when it is combined with chemotherapy. Less frequently observed side effects encompass fatigue, nausea, vomiting, thrombocytopenia, diarrhea, elevated AST and ALT levels, reduced appetite, headaches, and urinary tract infections. 22 In our case, the patient tolerated triple therapy with olaparib, nivolumab, and fluorouracil and docetaxel well with no major side effects for at least 6 months. Nivolumab and chemotherapy were eventually discontinued due to the immunotherapy-associated adverse events and subsequent poor performance status.
Our case is unique, and it demonstrates the real-world benefits of olaparib in a patient with germline ATM-mutated gastric cancer, who had an exceptional response with the remarkable reduction of CA 19-9 from an initial 48 000 units/mL to <34 units/mL (>90%) that was maintained for at least 18 months. Elevated CA 19-9 levels, as consistently reported across various studies, are linked to an unfavorable prognosis. Higher CA 19-9 levels signify increased tumor aggressiveness, a greater tumor burden, and an elevated risk of mortality. 23 Our patient was maintained on olaparib for 18 months with good tolerability. There are unknown territories worth exploring further based on our case: Did our patient’s germline HRR mutation lead to an exceptional deep durable response to chemoimmunotherapy as well as to the PARP inhibitor? Regardless, this case highlights the safety of olaparib combined with chemo-immunotherapy as well as the potential clinical utility of olaparib, particularly in germline ATM-mutated gastric cancer. Currently, there are various ongoing clinical trials in looking at the potential clinical efficacy of PARP inhibitors either as monotherapy or combination therapy in BRCA- and HRR-deficient gastric cancer. We are hopeful that there will be larger clinical trials that explore the benefits of using of PAPR inhibitors either as monotherapy or combined with chemoimmunotherapy in germline or sporadic ATM-mutated solid tumors.
Conclusion
This case report, showing an encouraging outcome in a patient with ATM-mutated gastric cancer treated with olaparib and chemoimmunotherapy, hints at the potential efficacy of PARP inhibitors in this subset of gastric cancer cases. Our example underscores the need for ongoing exploration of molecularly targeted therapies and the importance of identifying biomarkers for personalized treatment approaches in gastric cancer. Clinical trials and ongoing research are crucial in understanding the full spectrum of responses and optimizing the use of PARP inhibitors in various cancer types.
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude and appreciation to Ms. Virginia Mohlere, ELS, who helped revise the grammar of our manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethic Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
