Abstract
BK virus (BKV) is a small DNA virus, a member of the polyomavirus family, that causes an opportunistic infection in immunocompromised patients, especially kidney transplant patients. This virus establishes a lifelong infection in most of the population, and once it reactivates in an immunocompromised state, leads to BKV nephropathy. This review seeks to assess the correlation between severe immunosuppression, evident by low CD4 cell counts in HIV-positive patients, and the reactivation of BKV, causing nephropathy. A literature review was conducted, extracting, and analyzing case reports of HIV-positive patients showing correlations between their degree of immunosuppression, as evidenced by their CD4 counts, and the degree of BKV infectivity, confirmed by kidney biopsy. A total of 12 cases of BKV nephropathy in HIV-infected patients were reviewed. A common finding was the presence of profound immunosuppression, with most patients having CD4 counts ≤50 cells/ mm3. A substantial number also had comorbid malignancies, with some undergoing chemotherapy, potentially increasing the risk of BKV reactivation. In addition to the HIV status and malignancies, other risk factors for BKV reactivation included older age, male gender, diabetes mellitus, Caucasian race, and ureteral stent placement. BKV nephropathy in HIV patients with native kidneys is closely correlated with severe immunosuppression. Although therapeutic strategies exist for post-transplant patients, aside from the treatment of HIV with highly active anti-retroviral therapy (HAART), which potentially helps with clearing BKV by increasing CD4 count, there is no definitive treatment for a native kidney BKV nephropathy in patients with AIDS. The complexity of the cases and severity of comorbidities indicate the need for further research to develop therapeutic strategies tailored to this population.
Introduction
The Polyomaviruses are a family of small, non-enveloped viruses with circular double-strand DNA surrounded by icosahedral capsids.1,2 They are widespread among vertebrates, especially humans, who are considered a natural host for polyomavirus hominis 1 and 2 or BKV and JC virus (JCV).2,3 BKV is known for its pathogenic role in causing disease, mainly in the genitourinary tract. In contrast, the JCV is primarily known for causing multifocal demyelination of the central nervous system (CNS), known as progressive multifocal leukoencephalopathy (PML).2,3 BKV was first found unexpectedly during a study of another virus called cytomegalovirus (CMV) in a kidney transplant recipient with ureteric stenosis. 4 The urine sample belonged to a patient with the initials “B.K.”; thus, the BKV was first isolated as one of the few human polyomaviruses. 3 Epidemiologic studies estimate that approximately 80% of the adult population globally is seropositive for both JCV and BKV, with prevalence reaching 91% at 5 to 9 years of age and primary infection beginning as young as 6 months.3,5,6 The incidence of BKV nephropathy in the native kidney has increased in solid organ and bone marrow transplants.7-10 The transmission of BKV remains uncertain, with no natural route of transmission established, although the serological association of upper respiratory infection suggests transmission by aerosol or fomites.2,9,11,12 Other studies indicate uro-oral, fecal-oral, blood transfusion, sexual transmission via semen, transplacental, and even contaminated water.3,11-14 After primary infection, the virus travels to the uroepithelium and renal tubular epithelial cells and establishes latency until reactivation in the setting of immunosuppression.2,13 These complications arise mainly in post-kidney transplant patients on immunosuppressants but also occur, although rarely, in non-transplant patients. A spectrum of complications after BKV reactivation in the native kidney has been noted in past reports among HIV-positive patients, including severe tubulointerstitial nephritis, hemorrhagic cystitis, hydronephrosis, and ureteral stenosis.12,14-24 Detection of BKV in the urine often coincides with detection of the virus in the blood.9,12 Two or more consecutive urine samples with BK viruria can be 100% sensitive and 94% specific for BK viremia. 15 Commonly, the diagnosis of BKV nephropathy is suspected when urine polymerase chain reaction (PCR) is positive for BKV in kidney insufficiency.9,12,16 Plasma BKV PCR has also been used in kidney transplant recipients. However, this test is more suitable for ruling out BKV nephropathy as the negative predictive value can be as high as 100% but with a positive predictive value of 50%.2,12 Urine cytology positive for decoy cells (virally infected epithelial cells with enlarged nuclei similar to neoplastic cells) also indicates BKV infection. However, the test is not specific as they are also positive for JCV and adenovirus infection.9,17,18 Urinary BKV PCR can be nonspecific as asymptomatic patients with BK viruria are common or can remain negative.12,19 However, if suspicion of infection remains high, a kidney biopsy can confirm through visualization of viral infiltration and tubular damage.9,12 Nevertheless, the early stages of BKV infection are localized mainly in the medulla and distal tubules and are associated with a false-negative of up to 30%, given its focal characteristic. 6 Histologic examination of kidney biopsy shows viral particles in tubular epithelium cells and prominent intranuclear inclusions.9,12,20 Studies on treatment for BKV nephropathy are ongoing, as no specific antiviral drug exists. Reduction in immunosuppression post-kidney transplant has been the main therapy of choice that has been implemented, as shown in past case reports.9,12 One study on fluoroquinolones has shown antiviral activity when assayed against Papovaviruses (a family comprising of both polyomavirus and papillomavirus) by interfering with DNase I activity in the SV40—a chromatin specific to all Papovaviruses. 21 Cidofovir, a cytosine phosphate analog, has been shown to target BKV and is used in polyomavirus-associated nephropathy and PML in kidney transplant patients.5,6 However, this compound has yet to be studied in affected native kidney cohorts. 5 BKV clearance from plasma can be used as a surrogate marker to resolve kidney tissue infection. 6 Despite being rare, BKV nephropathy in native kidneys has been a growing topic in case reports and case series. 22 Patients with HIV, and especially those with advanced AIDS, are at risk for many different organ complications, not limited to the central nervous system, lungs, genitourinary, and eyes.22-24 Collective case reports suggest that the degree of immunosuppression correlates with the risk of BKV reactivation in the kidneys and other possible infections. 6 This review evaluates 12 reported cases of HIV-positive patients that show correlations in the severity of their immunosuppression evident by their low CD4 counts and the degree of kidney involvement found on biopsy. Our report presents a case of BKV nephropathy in the native kidney in an immunocompromised patient diagnosed with HIV infection 10 years prior and delivered to our medical center with renal dysfunction.
Case Report
Our patient is a 28-year-old Caucasian male who was diagnosed with HIV at the age of 18 years. He presented with fever and diarrhea of 1-month duration. He was cachectic and febrile with no other relevant findings on physical examination. At presentation, temperature was 103°F, heart rate was 150 bpm, blood pressure was 145/95 mm Hg, and body mass index (BMI) was 18 kg/m2. Initial laboratory workup revealed elevated serum creatinine of 3.0 mg/dL (with prior baseline of serum creatinine of 0.9 mg/dL), protein-to-creatinine ratio of 2.4, and urinalysis with specific gravity 1.008 and no red blood cells or white blood cells. The absolute CD4-positive T-lymphocyte count was 1 cell/μL (0.001× 109/L). Other laboratory findings showed a white blood cell count of 3800 × 106/L, hemoglobin 7.4 g/dL, platelet count of 107 × 109/L, serum sodium 146 mmol/L, serum potassium 4.8 mMol/L, and blood urea nitrogen (BUN) 27 mg/dL. Kidney ultrasound revealed bilateral echogenic kidneys without hydronephrosis. Hemodynamically mediated “pre-renal” acute kidney injury (AKI) due to volume loss from diarrhea and sepsis due to an underlying infection was suspected, and intravenous fluids and antibiotics were initiated. Over the course of 3 days, the level of serum creatinine worsened and increased to 4.8 mg/dL. Considering the proteinuria, lack of response to initial management, and echogenic kidneys on ultrasound, a decision was made to perform a kidney biopsy. HIV-associated nephropathy (HIVAN), tubulointerstitial nephritis, and acute tubular injury (ATI) were the main diagnostic considerations at the time of biopsy. The biopsy showed severe tubular injury with cytopathic nuclear alterations typical of BKV nephritis. This was confirmed using immunohistochemical antibody staining for SV40 large T-antigen, which cross-reacts with BKV (Figure 1). There was extensive background chronic injury, with about 50% fibrosis and tubular atrophy, but no evidence of HIVAN or any other significant glomerular disease. BK viremia of 600 000 copies/mL was confirmed with PCR.

The kidney biopsy showed severe tubular injury, with several foci displaying nuclear cytopathic change typical of BKV infection (arrows). Inset: an SV40 stain for BKV antigen is strongly positive in the abnormal tubular nuclei, confirming the presence of BKV.
Treatment with HAART and intravenous immunoglobulin (IVIG) therapy was initiated. Unfortunately, the patient, who had a prior history of non-compliance with his treatment, left the medical center against medical advice and was lost to follow-up.
Methods
To find other similar cases, a search was performed using retrieving, reviewing, and comparing different case report studies on databases and web searches, including PubMed, Google Scholar, Cochrane Library, and EMBASE, for papers published in English between 1973 and 2023, in which the full text was available. Search keywords included BKV nephropathy, HIV, AIDS, native kidney, non-transplant, renal failure, BK viruria, and BK viremia. Exclusion criteria included patients with a history of a kidney transplant, negative HIV history, no evidence of nephropathy, no biopsy-proven BKV nephropathy, non-BKV infection (eg, adenovirus, CMV, JCV), and cases described only in the abstract without full text. Out of 356 retrieved unique articles in the primary literature screen, 11 case reports met our inclusion criteria (Table 1).
AIDS-Associated BK Virus Nephropathy Cases.
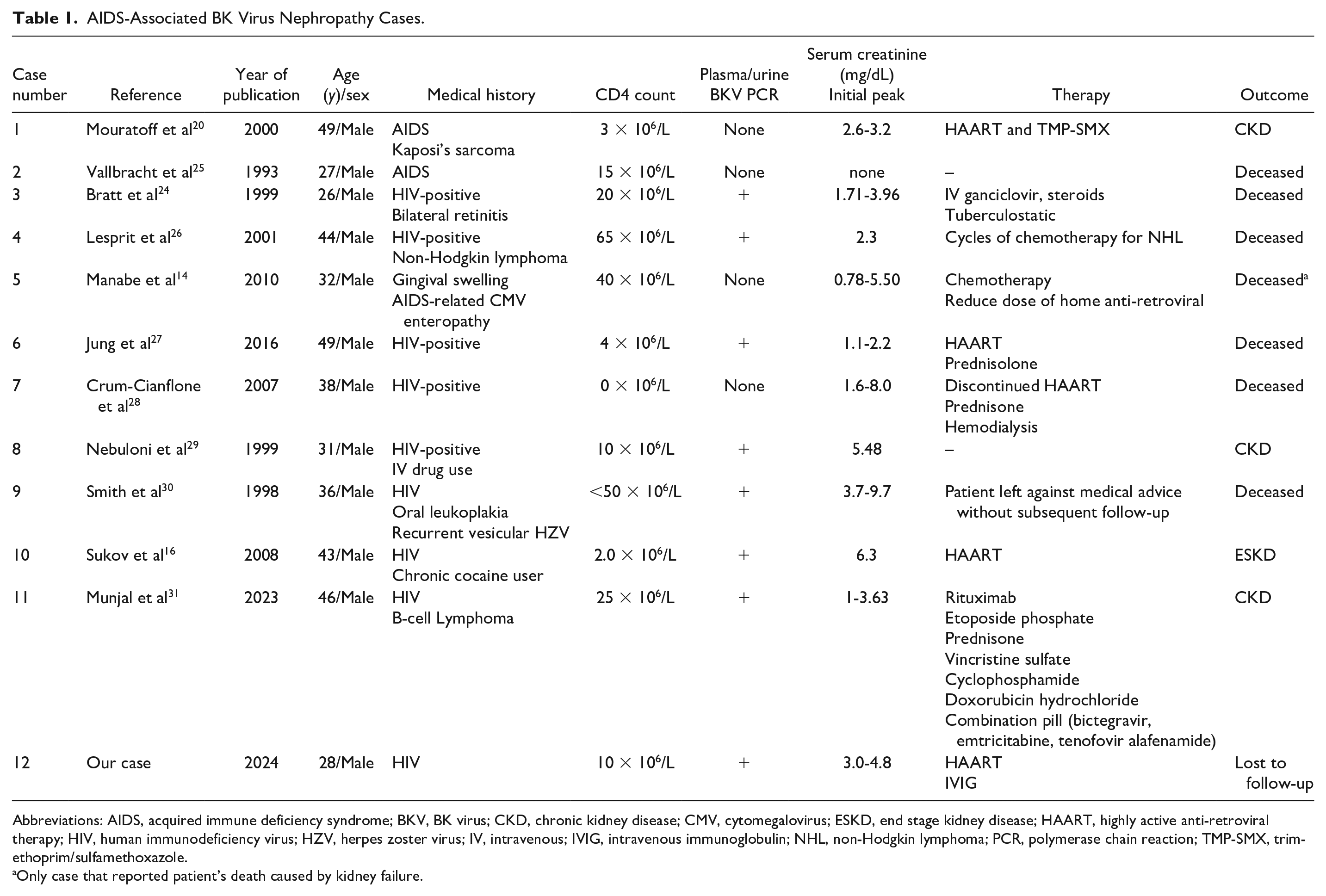
Abbreviations: AIDS, acquired immune deficiency syndrome; BKV, BK virus; CKD, chronic kidney disease; CMV, cytomegalovirus; ESKD, end stage kidney disease; HAART, highly active anti-retroviral therapy; HIV, human immunodeficiency virus; HZV, herpes zoster virus; IV, intravenous; IVIG, intravenous immunoglobulin; NHL, non-Hodgkin lymphoma; PCR, polymerase chain reaction; TMP-SMX, trimethoprim/sulfamethoxazole.
Only case that reported patient’s death caused by kidney failure.
Discussion
Although well recognized among kidney transplant recipients with kidney dysfunction, BKV nephropathy is often not considered in immunocompromised patients with kidney dysfunction who have not undergone kidney transplant. In addition to immunosuppression, a higher risk for developing BKV nephropathy has been reported to be associated with older age, male sex, Caucasian race, comorbidity of diabetes mellitus, and ureteral stent placement.19,32 All of the patients in our review series were young to middle-aged adult males with severely low CD4 cell count—a predisposing factor to BKV reactivation from latency. As shown in Table 1, 10 out of 12 cases reported a CD4 ≤ 50 cells/mm3, indicative of advanced HIV/AIDS and associated with morbidity and mortality and poor outcomes. 33 In addition, 5 of the 12 cases also had a concurrent malignancy, and 3 of them were noted to be receiving chemotherapy, likely further immunosuppression. All the reported cases had similar findings on kidney biopsy, including tubular epithelial cell injury and necrosis, interstitial edema, basement membrane erosion, and interstitial atrophy and fibrosis.
With AIDS-related kidney impairment, the differential is broad, but BKV-associated nephropathy should be considered, even outside the setting of the kidney transplant. BKV can cause significant impairment in kidney function and even progression to ESKD. In allograft kidney transplant recipients, about 10% of patients who develop viruria from viral reactivation will progress to develop BKV nephropathy within a year of the transplantation, 4 and the loss of allograft function rates can reach as high as 50% to 80%, especially if diagnosed late. 8 Screening for BK viremia or viruria is routinely done post-kidney transplant. The risk of graft loss makes early diagnosis particularly important. 34 In addition, earlier administration of anti-retroviral therapy could contribute to decreased incidence of BKV, although studies are limited.
However, according to case reviews, early screening for the detection of BK viruria or BK viremia might also be considered in other immunosuppressive states, specifically AIDS patients. Only the patient reported by Mouratoff et al had initial improvement in kidney function, although the patient was eventually diagnosed with CKD 2 months later. The rest of the patients progressed to ESKD and/or death. However, most of those patients also had other pre-existing comorbidities or contributors to their demise, such as CNS deterioration, tumor progression, or pneumonia.25,28,29 Other studies have shown that early and regular screening decreases the incidence of BKV nephropathy following kidney transplant with subsequent reduction of immunosuppressant agents. 6 Probably, a lower threshold should be considered to perform a kidney biopsy in patients with AIDS and unusual course of AKI like rapid decline in glomerular filtration rate (GFR) and significant proteinuria.
The current protocol for treating BKV nephropathy revolves around conservative management and the reduction or discontinuation of immunosuppressive agents in kidney transplant patients. Leflunomide has been used in treating BKV nephropathy post-kidney transplant. 31 One study showed inconsistent results among kidney transplant patients with BKV nephropathy when treated by reduction or discontinuation of the immunosuppression, with graft loss ranging between 10% and 80% of the cases. 19 However, aside from the treatment of HIV with HAART, there is no definitive treatment for a native kidney BKV nephropathy in patients with AIDS and there is a paucity of guidance in the literature for treatment of BKV nephropathy in non-kidney transplant patients. Further studies are needed to determine treatment options and efficacy to prevent poor outcome.
Conclusions
To the best of our knowledge, BKV nephropathy, traditionally associated with kidney transplant recipients, is increasingly recognized in immunocompromised individuals with native kidneys. A significant observation from the cases reviewed is the correlation between severe immunosuppression, as evidenced by profoundly low CD4 cell counts, and BKV reactivation. The presence of comorbidities (such as malignant tumors), particularly when coupled with chemotherapy, may further increase this risk. Kidney biopsy findings across the cases were consistent, indicating significant kidney injury. Despite existing treatment protocols for post-kidney transplant patients, there is a conspicuous lack of well-established therapeutic approaches for BKV nephropathy in native kidney patients. The mixed outcomes observed in cases that, combined with the varied reasons for morbidity, highlight the complexity of managing this condition and emphasize the need for further research to develop therapeutic strategies for this vulnerable population.
Footnotes
Acknowledgements
The authors acknowledge Dr Craig Zuppan, MD, for his assistance in providing the biopsy slide for this report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
This case was presented at the NKF spring meeting 2018, Austin, Texas.
