Abstract
Shone complex (SC) is a rare congenital heart disease characterized by four obstructive anomalies, including parachute mitral valve (PMV), left atrial supra-valvular ring, subaortic stenosis, and coarctation of the aorta. Typically, SC manifests early in life. However, we encountered a 52-year-old female with a history of hypertension diagnosed at 26 years and left-sided weakness poststroke. She presented with worsening dyspnea and palpitations, prompting a thorough investigation. Echocardiography revealed a heavily calcified bicuspid aortic valve with severe aortic stenosis and parachute mitral valve with severe mitral stenosis and preserved ejection fraction, raising suspicions regarding the presence of SC. Cardiac catheterization, aortic-angiography, and noncontrast chest computed tomography (CT) revealed abrupt occlusion of the postductal aorta, giving a picture of aortic coarctation with well-established collateral vessels including prominent right and left internal mammary arteries. So, she was diagnosed with an incomplete SC at the age of 52. Shone complex is a rare congenital heart disease that typically presents in early childhood, but late presentations due to misdiagnosis or incomplete work up are possible. This case emphasizes the rarity of late presentations of SC and highlights the importance of early diagnosis and intervention to improve outcomes. An incomplete SC should be considered in adult patients presenting with left-sided obstructive lesions.
Keywords
Introduction
Shone complex is a congenital heart disease (CHD) that was first described in 1963 by Dr John D. Shone. It is a very rare disease and accounts for approximately 0.6% of all CHDs. 1
Shone complex is characterized by the presence of 4 obstructive anomalies that include parachute mitral valve, left atrial supravalvular ring, muscular or membranous subaortic stenosis and coarctation of the aorta. 2 When all 4 of these findings are present, it is known as complete SC, while the incomplete form is more common and consists of a defect in the left ventricular inlet including parachute mitral valve (PMV), mitral stenosis, or supra-valvular mitral ring, in addition to the presence of at least 1 left ventricular escape lesion, including bicuspid aortic valve, subvalvular aortic stenosis, or coarctation of the aorta. 3
Due to recessive genotypes in MYH6, SC was shown to have an underlying genetic inheritance in 11% of instances.
RGs in MYH6 in the SC, a disease with a previously unidentified origin, are examples of novel phenotypes induced by recessive mutations in genes previously implicated in CHD caused by monoallelic mutations. The discovery of abnormal ventricular function in a number of these patients, as well as in other patients with monoallelic MYH6 mutation, raises the possibility that individuals with SC and biallelic MYH6 mutations may be especially at risk for ventricular dysfunction, allowing for the possibility of early detection and intervention. 4 Although it typically presents in early childhood, late, undiagnosed presentations have been rarely reported in literature. 5 Echocardiography can diagnose SC or raise suspicion of its presence but multimodality imaging allows better evaluation of intra- and extra-cardiac obstructive lesions. 2
Case Presentation
We present the case of a 52-year-old female with a history of early onset hypertension diagnosed at the age of 26, which was not well controlled on 3 medications including a diuretic. She also had left-sided body weakness poststroke 3 years prior to admission. The patient sought medical advice 3 months after complaining of gradually progressive shortness of breath and palpitations on minimal activity.
On physical examination, she had high blood pressure in both upper limbs, asymmetrical pulse strength between upper and lower extremities, and a systolic murmur diffusely heard throughout her precordium. An electrocardiogram (ECG) showed a heart rate of 78 beats/min, normal sinus rhythm, and left ventricular hypertrophy criteria with P mitrale indicating left atrial enlargement.
Two-dimensional (2D) transthoracic echocardiogram (TTE) and 2D transesophageal echocardiogram (TEE) revealed normal ejection fraction (EF) of 60%, heavily calcified bicuspid aortic valve (type I) with severe stenosis, maximum velocity (Vmax) of 4.28 m/s, mean pressure gradient (P-mean) of 40 mm Hg, and left ventricular outflow tract area was 1.9 cm2 (Figures 1 and 2).

Two-dimensional transesophageal echocardiography demonstrates an anterior-posterior bicuspid aortic valve.

Doppler tracings from the patient, the aortic continuous wave (CW) recording shows a peak gradient of 73 mm Hg and a mean gradient of 40 mm Hg. These findings are consistent with severe aortic stenosis.
Mitral valve showed heavy calcifications, PMV (unifocal attachment of the chordae to posteromedial papillary muscle) causing severe stenosis, P-mean was 11, and mitral valve area of 1.5 cm2 (Figures 3-6).

Continuous-wave Doppler echocardiogram across the stenotic mitral valve showed Vmax of 240 cm/s and mean Pressure Gradient of 11 mm Hg.
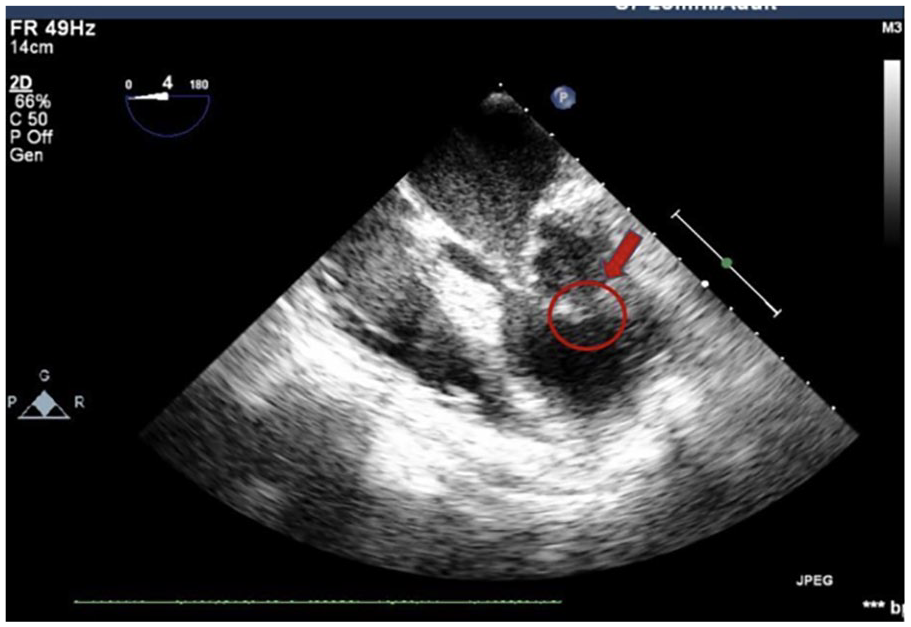
Two-dimensional transesophageal echocardiographic at midesophageal (ME) 4-chamber view demonstrated anterior and posterior mitral leaflets attached to thickened and shortened chords that originated from a single papillary muscle (PM).
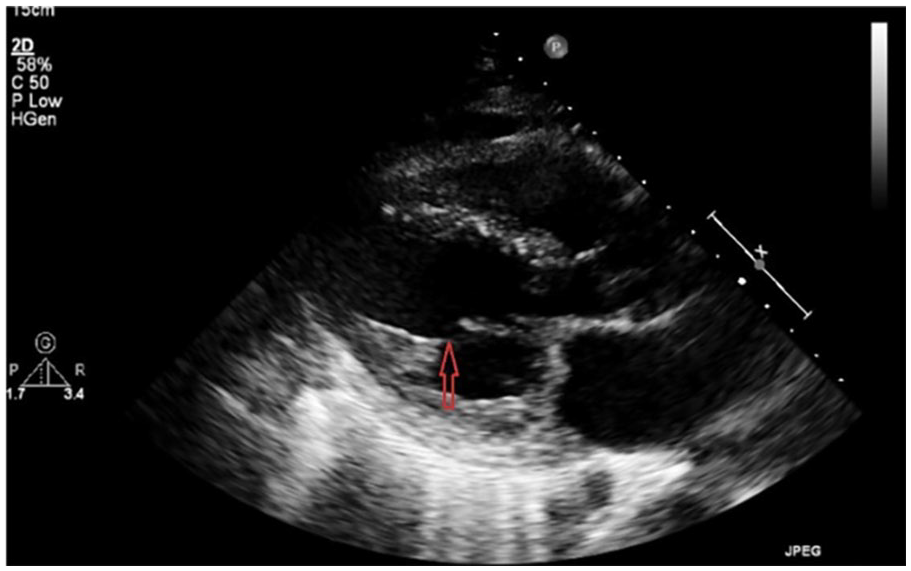
Parasternal long axis (PLAX) view shows both chordae are attached to a single point, consistent with a parachute mitral valve.

Parasternal short-axis view showing single papillary muscle.
Further investigations, including cardiac catheterization, revealed normal coronaries, but an aortoangiography showed abrupt occlusion of the postductal aorta, consistent with aortic coarctation. Findings were also confirmed on noncontrast chest computed tomography (CT), which also showed prominent right and left internal mammary arteries (Figures 7 and 8).
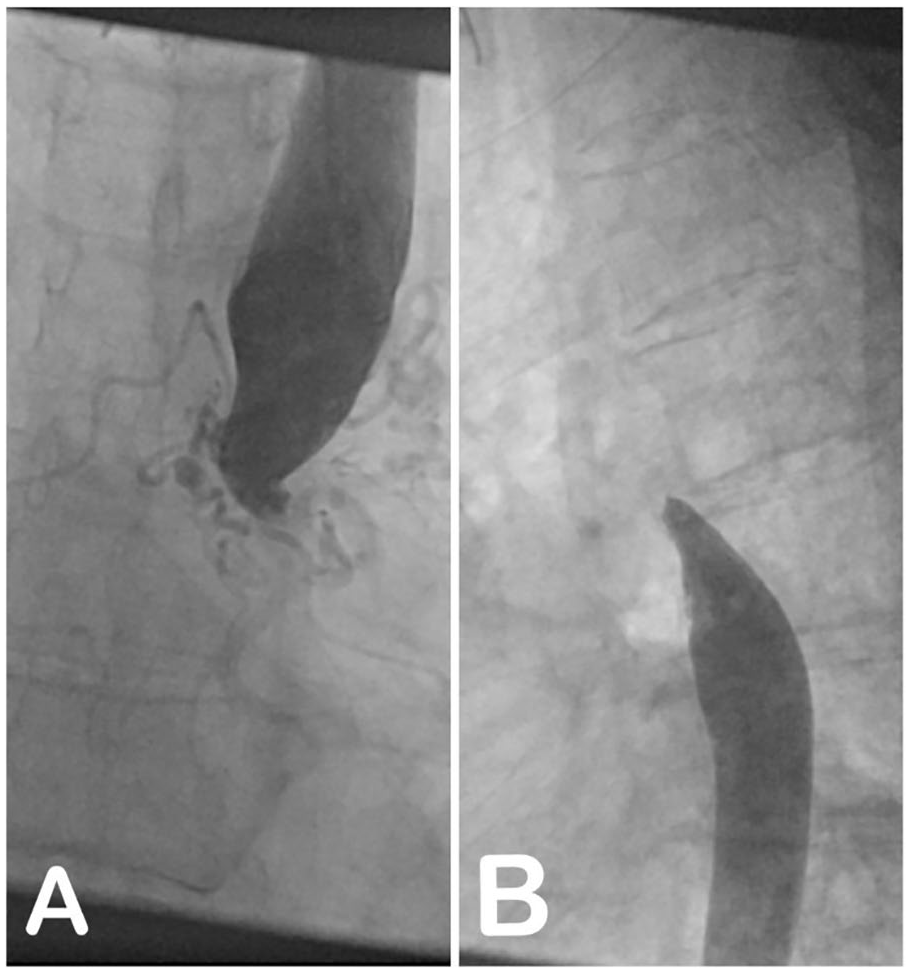
A and B aortoangiography showed abrupt occlusion of the postductal aorta, consistent with aortic coarctation.

Two-dimensional sagittal computed tomography scan imaging of coarctation of aorta showing the discrete narrowing site at the aortic isthmus level (arrow).
After consultation with the cardiology and cardiac surgery teams, the patient underwent multiple corrective surgeries. The first was coarctation of the aorta repair using the left subclavian artery to descending aorta extra-anatomical graft. During the surgery, blood pressure equalized in both upper and lower limbs after declamping the graft. The postoperative course went smoothly without complications, and the patient was discharged 6 days after the surgery.
The patient was readmitted 2 months later for her second surgery, which involved bypass mitral and aortic valve replacement. Postoperative echocardiography revealed moderate impairment of left ventricular function with an ejection fraction of 40%, a prosthetic aortic valve with a P mean of 7 mm Hg and no regurgitation, and a prosthetic mitral valve with a P mean of 4 mm Hg and no regurgitation.
During her hospital stay, the patient also experienced two tonic-clonic convulsion episodes, but remained vitally stable. A brain CT was performed, which showed no new abnormalities other than previously documented encephalomalacia in the territory of the middle cerebral artery after her stroke 3 years before. There was no evidence of brain hemorrhage or new stroke. The patient was followed up by the neurology team and maintained on Levetiracetam 1000 mg 1 × 2. She recovered well and was discharged 20 days after the surgery.
On follow-up, the patient reported great improvement in her symptoms, with normal physical examination except for prosthetic valves associated auscultatory findings. Her blood pressure was well-controlled, and she had no other complaints.
Discussion
First described in 1963, Shone’s anomaly is considered a rare congenital entity with left-sided obstructive lesions. The classic findings consist of a parachute mitral valve and muscular or membranous supra-valvular ring of the left atrium. These 2 findings combined represent the classical SC. 2
Parachute mitral valve is defined as a mitral valve with 2 leaflets and 2 commissures, in which chordae insert into 1 papillary muscle instead of 2. Supra-valvular ring of the left atrium is defined as a connective tissue ring that arises at the atrial surface base of the mitral valve leaflets and protrudes into its inlet. The third and fourth findings include subaortic stenosis and aortic co- arctation. 2
Shone complex usually presents early, a retrospective study was conducted showing that the diagnosis of SC was at a median age of 14 days. 6
Another retrospective cohort study revealed that SC is an often-overlooked diagnostic entity, with a prevalence of 0.7% among 4000 patients with CHD. The incomplete form of SC was more common than complete. Morbidity was related to arrhythmias, heart failure, and interventions, leading to high rates of cardiac hospitalization. In adulthood, patients had high rates of mitral valve, aortic valve, and left ventricular outflow tract reintervention. However, mortality in patients who reached adulthood was relatively low.
Furthermore, high prevalence of left superior vena cava was identified. These findings highlight the importance of recognizing and appropriately managing SC. 7
Our case highlights the importance of considering SC in the differential diagnosis of adults with suspected CHD, especially when presenting with obstructive lesions of the left heart and emphasizes the need for multimodality imaging to fully evaluate and characterize the extent of the disease. Furthermore, it suggests that SC can be present asymptomatically.
Conclusion
In conclusion, this case report describes a rare presentation of incomplete SC in an adult patient, which is a CHD that is typically diagnosed in infancy. The patient’s delayed presentation and diagnosis of this condition highlights the importance of maintaining a high level of suspicion for CHDs in adult patients who present with unexplained symptoms such as uncontrolled early onset hypertension. Early diagnosis and intervention can lead to improved outcomes for these patients, and therefore, it is essential for clinicians to consider CHDs in their differential diagnosis when evaluating adult patients with these symptoms.
Footnotes
Author Contributions
LHA-H, YN, and DIB contributed to writing the first draft. SM and HK contributed in data collection, and writing the final manuscript. BA supervised the project. All authors contributed to the article and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The study is exempt from ethical approval in our institution in Al-Makassed Charitable Hospital.
Informed Consent
Written informed consent was obtained from the patient for the publication of this case report and accompanying images. The patient is a legally competent person to do so in accordance with the applicable law. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Grantor
Yumna Njoum is the guarantor of this case report, taking full responsibility for the integrity of the manuscript and ensuring that all questions related to its accuracy or completeness are addressed.
