Abstract
Median arcuate ligament syndrome (MALS) is characterized by the constriction of the celiac trunk caused by fibrous connections originating from the median arcuate ligament (MAL) and diaphragmatic crura. It presents with symptoms often leading to misdiagnosis. In this study, we present three cases of MALS, with distinct manifestations. These cases were diagnosed through comprehensive investigations and managed successfully using laparoscopic decompression. The diagnosis of MALS poses challenges due to its variable presentations and overlap with other conditions. Diagnostic imaging techniques such as Doppler ultrasound, computed tomography (CT) scans and angiography play a role in confirming the diagnosis. Laparoscopic decompression has proven to be a treatment option that relieves symptoms and restores blood flow. This series highlights the importance of considering MALS as a cause for abdominal pain cases. Early detection and the use of diagnostic techniques can result in favorable outcomes.
Introduction
Median arcuate ligament syndrome is an uncommon disorder that arises from the constriction of the celiac trunk caused by the downward positioning of fibrous connections originating from the MAL and diaphragmatic crura. 1 A triad of symptoms including weight loss, nausea, vomiting, and postprandial epigastric pain, is frequently observed. 2 Despite its infrequency, MALS remains a subject of medical interest, yet its true prevalence remains uncertain due to its variable clinical expression. 2 The condition tends to favor a female phenotype (4:1) and typically emerges within the age range of 30 to 50 years. 3
This syndrome presents a challenging diagnostic workup, partly owing to its varied clinical manifestations and poorly understood pathophysiological mechanism, requiring comprehensive investigations to exclude more common causes of abdominal pain.2,3
Various imaging modalities, such as Doppler ultrasound, computed tomography (CT), magnetic resonance imaging (MRI), and mesenteric angiogram, play a crucial role in highlighting celiac axis compression. 1 The treatment goal revolves around celiac trunk decompression, using approaches like open, laparoscopic, or robotic methods, accompanied by adjunctive interventions like percutaneous transluminal angioplasty (PTA) and stenting.1,2
In this context, we present a case series involving three distinct instances of MALS, each characterized by unique presentations and managed through laparoscopic decompression.
Case Presentation
Case 1
A 36-year-old male patient presented to our hospital with a persistent 50-day-history of abdominal pain. The pain was severe, diffuse, and characterized by a dull sensation. It worsened after eating and remained unrelieved by pain medications. Furthermore, it radiated to the back and was associated with nausea and anorexia. Recurrent episodes of pain were experienced leading to several hospital admissions and medical evaluations. The patient had no preexisting medical conditions and did not mention experiencing any decrease in appetite, or alterations in bowel habits. Although, a weight loss of 9 kg in 40 days was noted.
Initially, the patient sought medical advice at a local hospital after the pain was accompanied by facial and lip swelling, shortness of breath and a papular rash. Despite extensive investigations, he was misdiagnosed with gastritis and given multiple medications that failed to provide relief. Subsequently, upper and lower endoscopy revealed antral gastritis, leading to the recommendation to continue treatment under the assumption of gastritis.
The persistence of similar episodes without a clear diagnosis prompted him to seek care at another local hospital. He presented with additional respiratory symptoms, including shortness of breath and a dry cough, necessitating supplemental oxygen and ipratropium bromide. Laboratory results, including complete blood count (CBC), kidney function tests (KFT), liver function tests (LFT), C-reactive protein (CRP), coagulation studies, serum electrolytes, urinalysis, amylase, and lipase, all remained within normal limits. COVID-19 and H1N1 tests were negative, and genetic studies related to familial Mediterranean fever yielded no conclusive findings. In light of the accompanying skin rash and respiratory issues, suspicion shifted toward neuroendocrine tumors such as carcinoid. Further investigations included 24-hour urine collection for chromogranin and 5-hydroxyindoleacetic acid (HIAA) levels, as well as upper and lower endoscopy with jejunal and ileal biopsies, all of which were unremarkable. Magnetic resonance elastography indicated mild wall thickening in the distal ileum and transverse colon.
As symptoms persisted, a referral to our hospital was made for further evaluation and management. Upon assessment by our medical team, the patient exhibited multiple small, painless, papular rashes on the upper and lower extremities. Upper and lower endoscopies with multiple biopsies revealed multiple, scattered hyperemic areas throughout the gastric mucosa. Pulmonary function test (PFT) indicated Forced expiratory volume (FEV)1/Forced vital capacity (FVC) of 70%, total lung capacity (TLC) of 76%, and Diffusion capacity for carbon monoxide (DLCO) of 80%.
Abdominal CT scan revealed thickening of the median arcuate ligament (Figures 1 and 2), prompting a targeted dynamic Doppler US of the celiac artery that demonstrated significant findings consistent with MALS.

Sagittal preoperative arterial phase computed tomography (CT) scan.
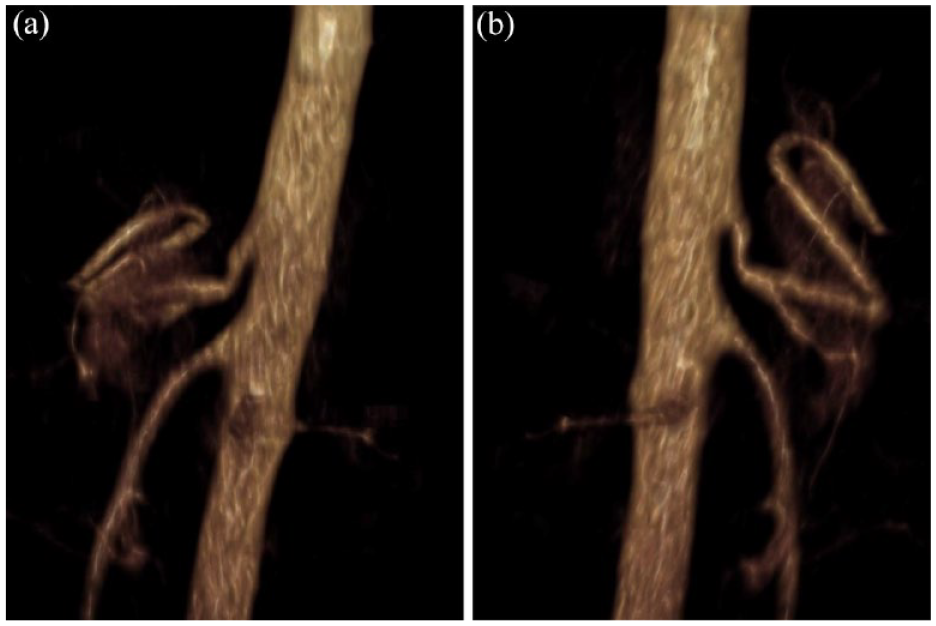
Sagital 3D reconstruction pre-operative (A) and post-operative (B).
Subsequently, laparoscopic MAL release was performed. The source of the celiac artery was located, followed by the release of the MAL and the excision of the celiac ganglion (Figure 3), resulting in improvement of symptoms. The patient was discharged in stable condition. During his follow-up 2 weeks later, the patient was well without complaints, underscoring the success of the intervention in alleviating his symptoms. However, the patient missed the subsequent follow-up, leading to the omission of a postop CT scan.
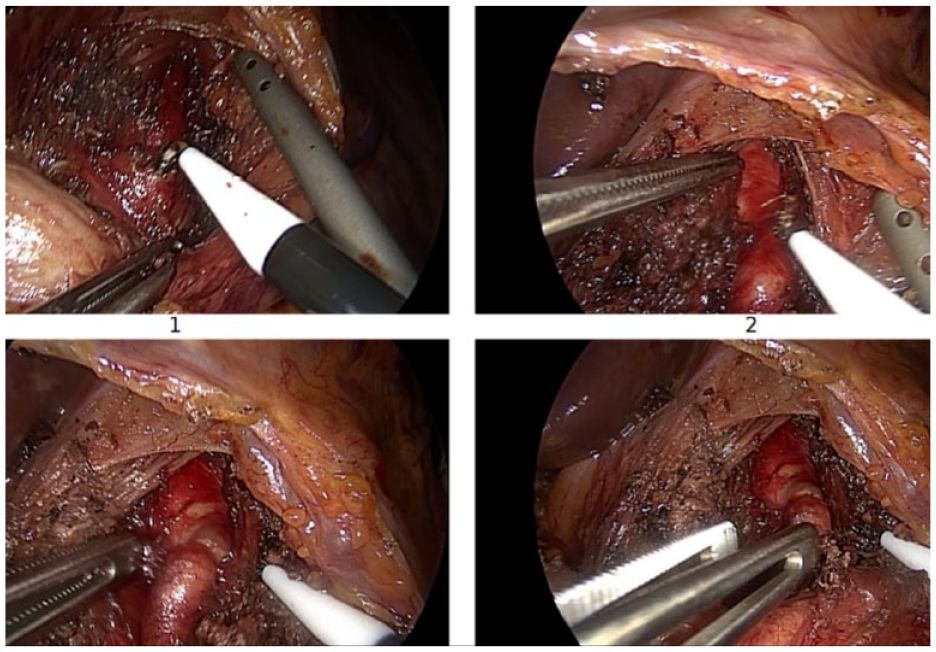
Intraoperative image of laparoscopic approach toward median arcuate ligament (MAL) release.
Case 2
A 53-year-old male patient known to have sarcoidosis, presented to our hospital due to acute exacerbation of a 7-month-long clinical history of epigastric pain both postprandial and unprovoked, the pain was colicky in nature, severe, and not relieved by analgesia, associated with vomiting of gastric contents and occasional hematemesis. In addition, he had unintentionally lost 18 kg over a short period and had multiple episodes of diarrhea. A history of chronic cough without hemoptysis was noted, and he denied urinary symptoms or fever.
Despite ongoing medical management, his symptoms persisted, leading to recurrent hospital admissions as a case of gastroenteritis for intravenous fluid therapy and antiemetics.
Laboratory tests were normal, including CBC, LFT, KFT, coagulation studies, serum electrolytes, and lipid profiles. Upper and lower endoscopy were performed and showed an upper esophageal submucosal lesion of size around 2.5 × 3 cm, hard in consistency, intact overlying mucosa, a lower esophageal sphincter incompetence, mild nonerosive gastropathy, mild-to-moderate bulbar duodenitis, H. pylori rapid urease test was negative. A proton pump inhibitor was prescribed without relief.
Given the refractory nature of his symptoms, coupled with suspicion of MALS, we proceeded with a comprehensive evaluation using an abdominal CT scan during the arterial phase, specifically timed during inhalation.
The imaging revealed a thickened arcuate ligament, measuring 6 mm. This ligament displayed an acute angulation, causing indentation upon the superior aspect of the celiac trunk. Notably, this configuration created a distinctive hooked appearance, consistent with MALS, accompanied by an estimated diameter narrowing of approximately 67%. This radiographic evidence further supported the diagnosis of MALS.
The decision was made to proceed with laparoscopic release of the arcuate ligament. The surgical team performed MAL liberation, adhesiolysis, and celiac ganglion resection via laparoscopic surgery.
The patient had a good postoperatory evolution with improvement of abdominal pain, without acute complications so he was discharged shortly after. At a 1-week postoperative check-up appointment, the patient had complete resolution of previous symptoms including pain, diarrhea and vomiting.
Postoperative CT during the arterial phase, specifically timed during inhalation showed improvement of the luminal diameter at the presumed site of arcuate ligament compression (about 2 mm in preoperative images and 5 mm in postoperative images) with estimated residual diameter narrowing of about 38% (Figures 4-6).
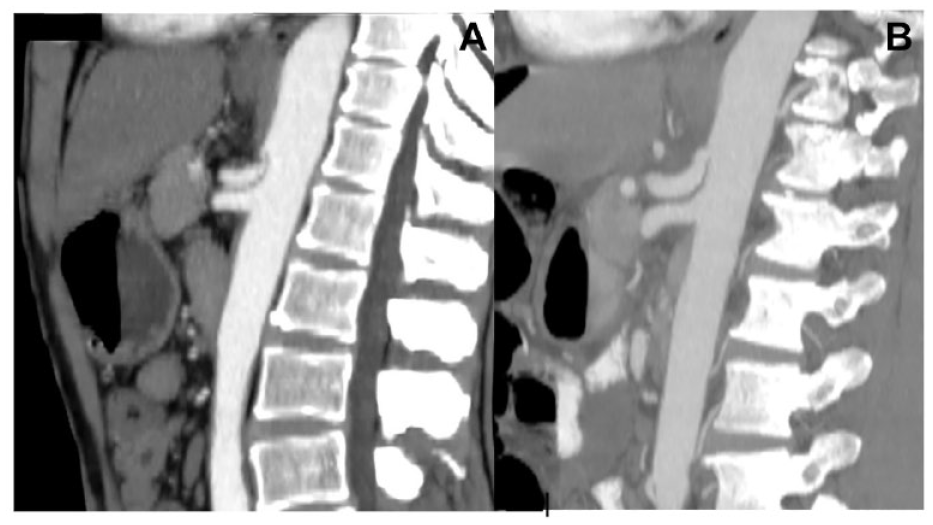
Sagittal computed tomography (CT) scan: arterial phase preoperative (A) and postoperative (B) images demonstrating celiac trunk constriction and indentation, and enhancement of postop diameter improvement.

Sagittal computed tomography (CT) scan: arterial phase postoperative with overlaying measurement at maximum narrowing and at fixed reference.

Sagittal 3D reconstruction preoperative (A) and postoperative (B).
Case 3
A 47-year-old male with past medical history of hypertension and past surgical history of laparoscopic cholecystectomy presented to our hospital with a 3-month history of postprandial pain. The pain was mainly in the epigastric region, acute in onset, moderately severe, dull in nature, nonradiating, predominantly after drinking and eating and accompanied by constipation. It was not associated with nausea and vomiting and was relieved with bowel rest. This phenomenon was more common with fatty meals. He mentioned a 14-kg weight loss in 2 months.
Despite receiving medical care and undergoing previous ureteroscopy for kidney stones after CT revealed moderate hydronephrosis, under the assumption that they were causing the symptoms, the insertion of a double J stent did not result in any relief.
The persistence of symptoms without a clear diagnosis prompted further investigations. Routine hematological and biochemical parameters were within normal limits. Aortoangiography revealed a U-shaped stenosis configuration at the celiac trunk with tight ostial stenosis and poststenotic dilation. In addition to intimal thickening at the celiac ostium with indentation of the MAL upon the stenotic segment, findings were suggestive of MALS, the celiac branches were opacified distally. Gastroscopy was also conducted showing mild gastritis at the antrum.
After confirming the diagnosis of MALS, the patient underwent laparoscopic MAL release in which the celiac trunk was identified, and the compressing band was dissected using hook cautery releasing the aorta and celiac trunk from compression. Postoperatively, he was transferred to the ward in good condition, without immediate complications.
A postoperative aortic angiogram demonstrated successful MAL release, leading to a notable improvement in the diameter of the celiac artery. The maximum diameter of narrowing preoperatively was 3.6 mm, which increased to 5 mm postoperatively (Figures 7 and 8). In addition, atherosclerotic changes were detected within the proximal segment of the splenic artery, resulting in a 60% diameter narrowing at its origin from the celiac artery. After determining that the patient was in satisfactory condition, he was discharged with complete instructions. A follow-up 15 days later revealed a complete resolution of the patient’s symptoms, affirming the effectiveness of the intervention.

Sagittal computed tomography (CT)—scan preop: arterial phase shows severe celiac trunk narrowing (1.4*10). Postobstruction dilation (10 mm) evident poststenotic dilation turbulence.

Postoperative diameter improvement and severe postobstruction dilation resolution.
Discussion
Median arcuate ligament syndrome is a rare vascular disorder characterized by the compression of the celiac trunk due to the downward positioning of fibrous connections originating from the median arcuate ligament and diaphragmatic crura, with a female predominance. It was first described by Harjola in 1963. 4
There are two current common hypotheses of MALS that try to explain the origin of its symptoms,3,5 one is neurological and the other is ischemic, the latter is said to be manifested by the compression of the celiac artery leading to either direct foregut ischemia or indirect midgut ischemia caused by postprandial steal via collaterals originating from the superior mesenteric artery ending in the celiac bed, while the former is due to compression of the celiac plexus leading to splanchnic vasoconstriction or direct sympathetic fibers irritation.
The clinical presentation of MALS can be vague and nonspecific, often leading to diagnostic challenges and numerous misdiagnoses all of which could lead to nonbeneficial trials of drugs and surgical interventions resulting in unwanted complications and delaying diagnosis. 2 In our study, all 3 cases were initially misdiagnosed, illustrating the diagnostic challenge (Table 1).
Patient Characteristics.

The shared triad of symptoms, including postprandial epigastric pain, weight loss, nausea, and vomiting, concurs with previous literature.2,6
Imaging plays a crucial role in confirming the diagnosis of MALS and aids in visualizing celiac trunk compression.7,8 The use of various modalities was seen in these three cases, including Doppler ultrasound, CT, MRI, and angiogram. Case 1 underscores the value of dynamic Doppler ultrasound, demonstrating significant findings consistent with MALS across positions and respiratory phases. Case 2 highlights the role of CT in revealing the characteristic hooked appearance of the arcuate ligament, supporting MALS diagnosis. Case 3, utilizing CT and aorta angiography, underscores the need for vigilance in diagnosing MALS in unusual clinical presentations. Notably, endoscopy did not aid MALS diagnosis in these cases.
Laparoscopic approach became the favorable surgical approach over open decompression due to its long term positive outcomes, including decreased blood loss, greater postoperative pain relief, better cosmetic outcomes, and shorter hospital stays.9,10 In the context of a disease with limited standard surgical techniques due to its rarity and lack of extensive research, three approaches are commonly discussed. The first technique involves an antegrade approach, tracing down from the aorta’s upper side to decompress the MAL on the celiac body. The second technique, which we utilized in all of our cases, employs a retrograde approach, cutting the MAL while moving upward without dissecting the upper side of the artery. The third technique is a combination of both approaches, without definitive evidence favoring one technique over the others. Cautious maneuvering is necessary during surgery due to the proximity of vital structures. 11 In our cases, we successfully achieved MAL decompression using the retrograde approach. This procedure effectively released the MAL in all three cases, resulting in symptom relief and improved luminal diameter. Restoring adequate blood flow to the celiac trunk was reinforced by both preoperative and postoperative CT angiography results. The procedure was conducted under general anesthesia with the patient in reverse Trendelenburg with legs apart. A 10-mm camera port was inserted under direct vision in the epigastrium about 5 cm above the umbilicus.
After establishing pneumoperitoneum up to 15 mmHg, four 5-mm ports were introduced. The left segment of the liver was retracted and the gastrohepatic ligament was divided exposing the right crus of the diaphragm.
The left crus was identified upon stomach retraction to the left. The procedure involved skeletonizing the left gastric, proximal common hepatic and splenic arteries, progressively dividing the bands compressing the celiac trunk down to the aorta using hook diathermy. Blood loss was minimal. Postoperatively, the patients were discharged without acute complications.
This case series sheds light on the diagnostic challenges, imaging techniques, and successful laparoscopic management of MALS.
Conclusion
This case series emphasizes the difficulties in diagnosing MALS due to diverse presentations. In addition to the successful laparoscopic treatment options available. Median arcuate ligament syndrome, although rare, can display a range of symptoms that often result in misdiagnosis. Imaging techniques such as Doppler ultrasound, CT scans, and angiography are crucial for confirming the diagnosis. Laparoscopic ligament decompression effectively treats and restores blood flow. It is essential for disciplines to collaborate in order to accurately diagnose and intervene promptly. These cases highlight the significance of considering MALS as a cause of abdominal pain and emphasize the need for further research to improve our understanding and management of this uncommon vascular condition.
Footnotes
Authors Contributions
AAH, YA, MA and LA contributed to writing the first draft.
KA and TA contributed to data collection.
KA and MM supervised the project.
All authors contributed to the article and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study is exempt from ethical approval in our institution in Al Makassed Hospital.
Informed Consent
Written informed consent was obtained from the patients for the publication of this case series and accompanying images. The patients are legally competent to do so in accordance with applicable law. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Grantor
Mohammed Maree is the grantor of this case series, taking full responsibility for the integrity of the manuscript and ensuring that all questions related to its accuracy or completeness are addressed.
