Abstract
Portal venous thrombosis (PVT) is a rare diagnosis in the general population. However, it is seen in patients with liver cirrhosis with an estimated prevalence ranging from 0.6% to 26%. Literature reports that about one-third of PVT cases have an unknown etiology. Identifying the precipitating factors implicated in the development of PVT is imperative as it may help guide therapy. Although the association between liver cirrhosis and PVT has been well established in current literature, there continues to be a relative lack of awareness of alcoholic hepatitis (AH) as a risk factor for PVT. Identifying AH as a trigger for thrombosis can help avoid extended anticoagulation and its complications. In the following case report and brief review, we discuss an uncommon case of a 33-year-old male who came to the hospital emergency department with complaints of nauseousness, abdominal discomfort, and yellow discoloration. Lab investigations showed transaminitis. The diagnosis of AH was established, and an abdominal duplex ultrasound revealed PVT. Heparin drip was started as a part of treatment, which improved his abdominal discomfort. He was eventually discharged on apixaban 5 mg twice daily for 3 months and a repeat abdominal duplex ultrasound in 3 months to check for the resolution of the PVT.
Introduction
Alcoholic hepatitis (AH) is a clinical entity characterized by rapid-onset jaundice and liver-related complications in patients with excessive alcohol use. In the United States, the incidence of AH continues to rise, particularly in the younger demographic. It results in high rates of morbidity and short-term mortality.
Portal venous thrombosis (PVT) is defined as a sudden partial or complete occlusion of the portal vein. It is believed to be multifactorial in about 30% of cases. 1 However, about 30% to 40% of the cases of PVT are of unknown origin. 2 As per current literature, PVT is an infrequent complication of AH. In the following case report and brief review, we bring an unusual presentation of an unprovoked PVT leading to severe abdominal pain in a healthy young male diagnosed with acute AH. Through this case, we want clinicians to consider PVT as a differential diagnosis which causes abdominal pain in patients with AH.
Case Presentation
A 33-year-old man with no significant past medical history presented to the emergency department (ED) with chief complaints of abdominal pain for 3 days. He reported that the pain was sharp in nature, generalized, 1/10 in intensity at rest, and aggravated to 7/10 upon movement. He also reported associated poor appetite due to nausea, vomiting, and constipation for the past 7 days. Earlier in the day, he noticed yellowish discoloration of his eyes, which prompted him to seek medical attention and report to the ED. He has been drinking half a liter of liquor daily for the last 2 years. However, he has gradually stopped drinking over the last 10 days. He is not a smoker and denied cannabis or intravenous (IV) drug use. The patient has not had any sick contacts, not participated in recent outdoor activities, not consumed any raw food, or not traveled abroad. The family history was non-contributory. On examination, the patient was not in acute distress, and scleral icterus was noted. He was afebrile with a heart rate of 123 beats/min, respiratory rate 18/min, and blood pressure 145/65 mm Hg. Abdominal examination revealed right-upper-quadrant tenderness along with a palpable liver and spleen. No pertinent findings were noted on cardiovascular and respiratory examinations.
Laboratory investigations were ordered, which showed hemoglobin of 10.2 g/dL (normal range = 13.5-17.5 g/dL), hematocrit 28.7% (normal range = 41%-50%), white blood cell count 35.5 K/L (normal range = 4.5-11 K/L), platelet count 198 000 (normal range = 150 000-450 000 platelets/mL), total bilirubin 10.9 mg/dL (normal range = 0.1-1.2 mg/dL), direct bilirubin 9.2 mg/dL (normal range = less than 3 mg/dL), aspartate aminotransferase 306 IU/L (normal range = 6-34 IU/L), alanine aminotransferase 215 IU/L (normal range = 29-33 IU/L), alkaline phosphatase 335 IU/L (normal range = 37-116 IU/L), and lactic acid 4.6 mmol/L (normal range = 0.4-2.0 mmol). Serum electrolytes were within normal limits. However, serum ferritin was elevated at 2081 (normal range = 24-336 mcg/L), serum iron 89 (60-170 mcg/dL), transferrin saturation 90% (normal range = 20%-50%), and total iron-binding capacity 78 (normal range = 240-450 mcg/dL) (Tables 1 and 2). Additional workup entailed an abdomen and pelvis computed tomography (CT) scan which demonstrated an enlarged, fatty liver and splenomegaly without acute bowel obstruction.
Laboratory Investigations Including the Complete Blood Count (CBC) and Comprehensive Metabolic Panel (CMP) on the Date of Admission for the Patient.

Abbreviations: AST, aspartate aminotransferase; ALT, alanine aminotransferase; CBC, complete blood count; WBC, white blood cell; CMP, comprehensive metabolic panel; TIBC, total iron-binding capacity; MCV, mean corpuscular volume; BUN, blood urea nitrogen.
Laboratory Investigations Obtained During the Hospital Stay for This Patient.

Abbreviations: EBV, Epstein-Barr virus; MPL, IgM phospholipid units.
A decision was made to admit the patient to an inpatient setting for further evaluation of transaminitis, with broad differential diagnoses such as AH, viral hepatitis, and hemochromatosis, among others. His Model for End-Stage Liver Disease (MELD) score was 22, and his Maddrey’s Discriminant Function score was 38. A hepatitis panel was ordered, and blood culture was also sent. The patient was started on IV fluid, pantoprazole, multivitamin, thiamine, folic acid supplementation, and as needed lorazepam per the alcohol withdrawal protocol.
On the second day of hospitalization, the patient had some improvement in abdominal discomfort. Hepatitis panel did not show acute hepatitis. Labs showed noticeable improvements in transaminases; however, conjugated and unconjugated bilirubin remained elevated at 8.2 and 9.3, respectively. Additional testing for portal vein thrombosis, Still’s disease, Epstein-Barr virus (EBV), cytomegalovirus (CMV), and HIV were pursued. Next day, the patient was started on prednisone 40 mg once a day, and magnetic resonance imaging (MRI) abdomen without contrast was performed which revealed hepatosplenomegaly (Figure 1), questionable mild gallbladder wall thickening with a normal biliary tree, and normal pancreatic duct. Further investigation with contrast was recommended, which demonstrated new findings of liver with fatty infiltration and umbilical vein recanalization (Figure 2).
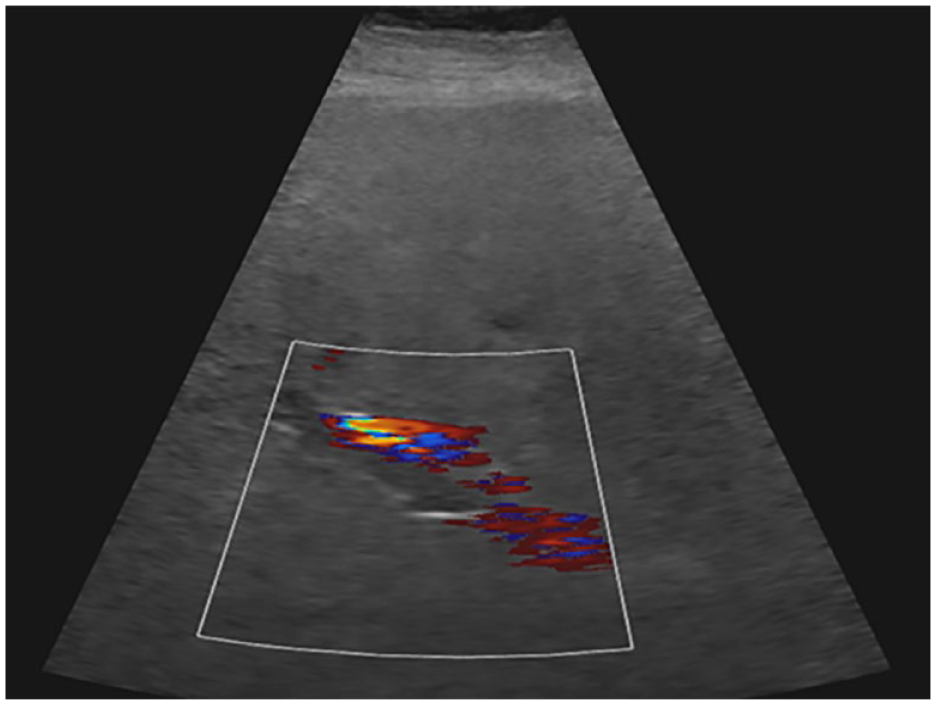
Thrombosis of the main portal vein demonstrated in ultrasound imaging.

Magnetic resonance imaging (MRI) abdomen in coronal T2 view showing hepatomegaly and splenomegaly.
Abdominal duplex ultrasound was pursued, which produced consistent findings of thrombus in the main portal vein (Figure 3). The patient was informed of the need for esophagogastroduodenoscopy (EGD) and colonoscopy to rule out varices before anticoagulation for portal vein thrombosis. However, the patient refused EGD and colonoscopy and opted for heparin infusion without endoscopy. In addition, a hypercoagulable workup was ordered before initiating heparin drip and revealed anticardiolipin IgM antibody to be slightly elevated at 15 MPL U/mL (indeterminate: 13-20 MPL U/mL, no-med positive: >20-80 MPL U/mL), antithrombin III activity slightly low at 68% (75%-135%), antithrombin III antigen 55% (72%-124%), and protein C activity also slightly low at 63% (73%-180%). With the initiation of heparin drip, his symptoms improved significantly, and liver enzymes normalized. Genetic testing for hemochromatosis which was ordered at hospitalization was also found to be negative. He was eventually discharged home on apixaban 5 mg twice a day for 3 months and with outpatient primary care follow-up. At the 3-month follow-up, the patient reported to be doing well. Follow-up abdominal duplex ultrasound revealed resolution of the extrahepatic PVT.

Magnetic resonance imaging (MRI) abdomen with and without contrast. Red arrow pointing to recannulized umbilical vein in the patient with portal vein thrombosis.
Discussion
Alcoholic liver disease (ALD) is a spectrum ranging from simple steatosis to AH to cirrhosis. Alcoholic hepatitis is acute hepatic inflammation associated with significant morbidity and mortality in patients with steatosis or underlying cirrhosis. Management of AH is primarily focused on cessation of alcohol and prednisolone in a subset of carefully selected patients. 3 In patients with liver cirrhosis, numerous risk factors for PVT have been recognized; however, there are insufficient data on the association between AH and PVT.
To establish a diagnosis of AH, it is important to rule out other chronic diseases of liver which can lead to cirrhosis such as hepatitis B, hepatitis C, non-ALD, auto-immune liver disease, hereditary hemochromatosis, Wilson’s disease, EBV, CMV, primary biliary cirrhosis, and primary sclerosing cholangitis. These were investigated in our patient and ruled out from the list of differential diagnosis. Hence, AH was established as the cause of transaminitis in our patient, aligning with his extensive history of alcohol use. Our case is unique as alcohol use was identified as the only risk factor for the acute liver injury with the presence of PVT.
The pathophysiology of acute non-cirrhotic PVT is based on the features of Virchow’s triad, that is, a hypercoagulable state, vascular endothelial injury, and reduced portal flow. Portal venous thrombosis may be secondary to vascular endothelial injury due to local risk factors such as an intra-abdominal inflammatory disease (eg, pancreatitis, cholecystitis, cholangitis, appendicitis, etc), surgery (eg, splenectomy, colectomy, portacaval shunts), and abdominal trauma. Compression or direct vascular invasion by hepatocellular carcinoma and cholangiocarcinoma may also lead to reduced portal blood flow and consequently thrombosis. Prothrombotic state, either inherited or acquired, might be the cause of PVT in patients without cirrhosis, among which myeloproliferative disease is the most common one. 4 In AH, the increased hepatic vein pressure gradient (HVPG) has a significant role in clinical manifestations. However, the amount and effect of alcohol on HVPG are unclear.
The three main variants of PVT include acute non-cirrhotic PVT, acute cirrhotic PVT, and chronic PVT. Our patient was diagnosed with acute non-cirrhotic PVT, which is a rare presentation. Clinical presentation for PVT ranges from asymptomatic cases, which are identified casually on imaging from acute liver failure with rapid obstruction of the vein and developmental of venous collaterals. Abdominal discomfort (61%), hepatomegaly (67%), and ascites (83%) are the most common presenting symptoms at diagnosis, and varices, specifically esophageal, could be detected in >50% of cases. 5 Patients with PVT who do not have cirrhosis or who have compensated (child A or B) cirrhosis should be evaluated for conditions that may have predisposed to the development of the clot, such as prothrombotic states. A thorough investigation for myeloproliferative disorders or other thrombophilic conditions should be considered including consultation with a hematologist. Imaging studies with Doppler ultrasound, CT scan, or multidetector CT angiogram should be used to identify PVD for further management. Doppler ultrasound has a sensitivity >75%, which also depends on the experience of the operative. 6
The primary management processes for acute PVT are anticoagulation and addressing the predisposing condition. In our patient, both anticoagulation and treatment of AH were pursued. If treatment for acute PVT is initiated prior to the onset of intestinal infarction, the patient’s prognosis turns out good. 7 Abdominal pain will start to subside within hours to days after the initiation of anticoagulation. If the superior mesenteric vein remains patent which prevents interstinal infarction, and if the portal trunk or in patients with recent DVT one of its branches remains patent or recanalized which prevents the portal hypertension. In a prospective study of 102 patients with acute PVT, enrolled over a period of 2 years, where thrombus extension and recanalization were assessed by expert radiologists, anticoagulation was given to 95 patients. Recanalization occurs in one-third of patients receiving early anticoagulation for acute portal vein thrombosis, whereas thrombus extension, intestinal infarction, severe bleeding, and death were rare. This study supports early anticoagulation for acute PVT; however, an alternative therapy should be considered when ascites and splenic vein obstruction are present. 8
Patients with diagnosis of PVT without cirrhosis should be considered for antithrombotic therapy to prevent interstitial ischemia and chronic PVT with portal hypertension. Patients should be started on Low Molecular Weight Heparin (LMWH) for rapid anticoagulation, with a switch to an oral anticoagulant (Coumadin or possibly a Direct Oral Anticoagulants [DOAC]) once the patient’s condition has stabilized and no invasive procedures are planned (if Coumadin is used, our international normalized ratio [INR] goal is 2-3). 9 Treatment is generally recommended for at least 3 to 6 months, although long-term treatment is recommended for most patients with permanent thrombotic risk factors that cannot be corrected. 10 Our patient was treated with anticoagulation therapy, and clot resolution was noted on follow-up abdominal Doppler ultrasound few months later. In our review of the literature, there are currently no cases reported for PVT in acute AH, making our case rare and unique.
Annually the probability of developing cirrhosis in cases of AH is approximately 10% to 20%, and approximately 70% of the patients with AH will end up with cirrhosis. Alcoholic hepatitis usually progresses to cirrhosis if drinking is continued. 11 For those who discontinue alcohol, hepatitis returns to normal within a few months, but the cirrhosis that has already occurred does not reverse. The gold standard for confirmation of cirrhosis is liver biopsy; however, limitation of being invasive results in frequent refusal by patients. The best noninvasive method available is MR elastography, and the next best is MRI. 12 Our patient had MRI imaging which did not show any evidence of cirrhosis and had refused the liver biopsy when offered on his follow-up outpatient appointments.
Conclusion
Acute PVT is a rare condition, but due to discrete and nonspecific signs and symptoms, it may be missed as a differential diagnosis. The origin of portal thrombosis is frequently multifactorial. Recent literature has emphasized the increasing prevalence of cirrhosis-induced PVT in immunocompetent patients. Identifying AH as a trigger for thrombosis can help to avoid extended anticoagulation. Overall, only limited amount of data is currently available on the influence of AH on PVT incidence. In this case, the temporal association of AH and the development of PVT, as well as the absence of other prothrombotic conditions, led us to conclude that alcohol use was the causative agent in the patient’s disease. To the best of our knowledge, this is the only reported case of AH causing PVT in the literature. A minimum of 3 months of anticoagulation is the mainstay of therapy to prevent the extension of the thrombus and development of complications that include intestinal ischemia and portal hypertension. With early anticoagulation, outcomes improved, and mortality can be avoided.
Footnotes
Authors Contributions
All authors have contributed equally to the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require it when reporting individual cases.
Informed Consent
Verbal informed consent was obtained.
