Abstract
Zieve syndrome is a rare alcohol-related disorder characterized by a triad of Coombs-negative hemolytic anemia, cholestatic jaundice, and transient hyperlipidemia. Its recognition is often delayed due to overlapping features with other hepatic pathologies, particularly alcoholic hepatitis. We present the case of a 35-year-old man with a history of alcohol use disorder, autoimmune hepatitis, and malnutrition who presented with jaundice, severe anemia, and hyperlipidemia. Laboratory evaluation revealed hemolytic anemia and cholestasis, and imaging showed hepatomegaly and hepatic steatosis. A clinical diagnosis of Zieve syndrome was made. Supportive care and alcohol cessation led to rapid clinical and laboratory improvement. This case highlights potential contributing factors beyond alcohol, including autoimmune liver disease and nutritional deficiency. A review of 11 published cases revealed similar patterns, suggesting that hepatic comorbidities may predispose to or exacerbate this syndrome. Clinicians should maintain a high index of suspicion for Zieve syndrome in patients with alcohol use disorder and anemia, particularly when bilirubin elevation and hyperlipidemia coexist. Prompt diagnosis may avoid unnecessary interventions and improve patient outcomes.
Keywords
Introduction
Zieve syndrome, first described in 1958 by Dr Leslie Zieve, is a rare clinical syndrome characterized by a triad of Coombs-negative hemolysis, cholestatic jaundice, and transient hyperlipidemia in individuals with heavy alcohol use or episodes of binge drinking.1,2 Once the diagnosis has been established, treatment is supportive, and resolution occurs with alcohol cessation or return to baseline alcohol consumption.1,2 We present a case of Zieve syndrome in a 35-year-old man with a history of alcohol use disorder and malnutrition to raise awareness of this often-overlooked entity. Prompt diagnosis would prevent unnecessary testing and interventions.
Case Presentation
A 35-year-old man with a history of alcohol use disorder with withdrawal seizures, autoimmune hepatitis, and malnutrition presented to the emergency department due to conjunctival icterus. He regularly consumed 2 beers and up to 4 shots of liquor daily for over 24 years. He denied fevers, abdominal pain, nausea, emesis, or changes in bowel patterns or consistency.
Physical examination was notable for a heart rate of 155 beats/minute. He was in no acute distress and alert but appeared malnourished. Conjunctival icterus and diffuse jaundice were present. There was no tenderness on palpation of the abdomen. Laboratory work revealed significant derangements including sodium of 125 mmol/L, potassium of 2.4 mmol/L, bicarbonate of 18 mmol/L, total bilirubin of 22.0 mg/dL, direct bilirubin of 17.0 mg/dL, alkaline phosphatase of 686 IU/L, alanine aminotransferase of 78 IU/L, aspartate aminotransferase of 309 IU/L, ethanol 316 mg/dL, and lipase 111 U/L. In addition, he had a white blood cell count of 11.57 10E9/L, hemoglobin of 4.8 g/dL, hematocrit of 13.6%, platelet count of 126 10E9/L, lactate dehydrogenase (LDH) 818 IU/L, haptoglobin <10 mg/dL, ferritin 2863 ng/mL, iron 61 µg/dL, and negative direct Coombs test. Mean corpuscular volume was 102.3 fL, despite normal levels of vitamin B12 and folate. The prothrombin time was 13.3 seconds, the international normalized ratio was 1.17, the cholesterol 931 mg/dL, the triglyceride was 401 mg/dL, high-density lipoprotein was 4 mg/dL, and the low-density lipoprotein could not be calculated. Comprehensive testing for autoimmune and infectious etiologies was negative. Acetaminophen assay, serum copper, and ammonia levels were within normal limits. A computerized tomography (CT) abdomen and pelvis revealed significant hepatomegaly and hepatic steatosis without signs of cirrhosis.
Treatment consisted of packed red blood cell transfusion, IV fluids, antiemetics, and electrolyte replacement. Total and direct bilirubin, lipid panel, transaminases, and hemoglobin improved with supportive care (Figures 1 and 2). An esophagogastroduodenoscopy showed pre-pyloric gastropathy but a normal esophagus and duodenum. He was discharged on hospital day 6 on daily pantoprazole and follow-up with primary care and gastroenterology.
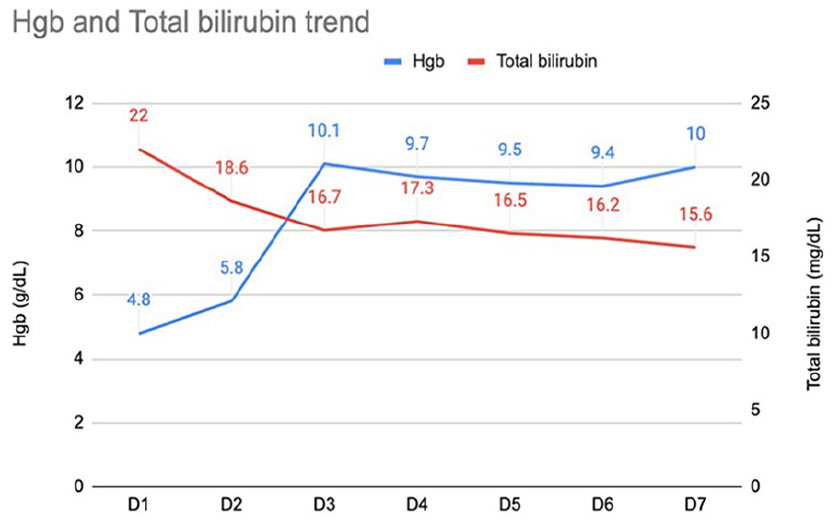
Trends in Hgb and total bilirubin over 7 days. This figure illustrates the temporal relationship between Hgb levels (g/dL) and total bilirubin (mg/dL) from day 1 through day 7. The blue line represents Hgb, which shows a gradual increase from 4.8 mg/dL on day 1 to 10 mg/dL on day 7. The red line represents total bilirubin, which trends downward from 22 mg/dL on day 1 to 15.6 mg/dL on day 7. Hgb, hemoglobin.

Trend in aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels over 7 days. This figure illustrates the progressive decline in liver enzyme levels. AST (blue line) and ALT (red line) from days 1 to 7. AST levels show a significant decrease from 309 U/L on day 1 to 106 U/L on day 7, while ALT levels decrease more gradually from 78 to 45 U/L over the same period. This trend reflects hepatic recovery, likely in response to supportive care and cessation of alcohol use in the context of Zieve syndrome.
Discussion
Zieve syndrome is defined by a triad of Coombs-negative hemolytic anemia, cholestatic jaundice, and transient hyperlipidemia, typically resolving with alcohol cessation. The triad is often accompanied by right upper quadrant abdominal pain, sometimes associated with pancreatitis or cholecystitis. 3 Since its first description in 1958, additional features such as myalgia, retinal injury, and intracranial hemorrhage have been reported.4-6
The mechanism of Zieve syndrome remains unclear. Alcohol is thought to alter RBC membrane composition through accumulation of lysolecithin and lysocephalin and cause oxidative stress via glutathione dysfunction, promoting hemolysis. 7 Hyperlipidemia is theorized to arise from mobilization of fat from a fatty liver, pancreatic alpha-cell injury, or lipoprotein lipase deficiency.3,7 Atypical cases with normal lipid levels have been documented, complicating diagnosis and likely contributing to under-recognition. 8 Hyperbilirubinemia reflects both hemolysis and hepatocellular dysfunction. While hemolysis leads to hyperbilirubinemia, most of the direct bilirubin elevation observed in Zieve Syndrome is secondary to hepatocyte injury due to alcoholism.9,10 Differentiation relies on identifying hemolytic anemia, low haptoglobin, elevated LDH, indirect hyperbilirubinemia, and a negative direct antiglobulin test (DAT) rather than macrocytic anemia more typical of alcoholic hepatitis. 11 Elevated transaminases or pancreatitis have been reported as associations with ZS.3,10,12 Some case reports highlight malnutrition, particularly vitamin E deficiency, as a risk factor for the development of ZS. It is thought to promote oxidative stress, erythrocyte glutathione, and polyunsaturated fatty acids depletion, and red cell instability eventually leading to hemolysis.9,13 In addition, autoimmune hepatitis, even when inactive, may impair hepatic function and lipid metabolism, lowering the threshold for decompensation.14,15 In our patients, the combination of chronic alcohol use, malnutrition, and autoimmune hepatitis likely contributed to the development of Zieve syndrome.
Alcohol cessation is the mainstay of management. It is thought to lead to excessive lipase activation, causing a shift of lipids from the plasma and liver to the adipose tissue, leading to a decrease in lipids. Figures 1 and 2 show the rapid recovery and trend of bilirubin, hemoglobin, and liver enzymes, following supportive care and alcohol cessation, consistent with typical Zieve syndrome recovery. While hyperlipidemia typically improves within 10 days and resolves by 6 weeks, hemolysis and jaundice may persist longer. 15 Plasmapheresis is rarely needed but may be considered in severe hypertriglyceridemia or complications like pancreatitis or intracranial hemorrhage. 16 In cases overlapping with alcoholic hepatitis, it is crucial to determine the bilirubin source before using Maddrey discriminant function scores to avoid unnecessary corticosteroid use. This is because many of the scoring systems incorporate bilirubin levels which are typically elevated in ZS, causing superficially elevated Maddrey discriminant function scores and unnecessary glucocorticoid use which may worsen outcomes in Zieve syndrome. Data have shown that treating critically ill patients with steroids often leads to adverse outcomes along with an increased incidence of hospital-acquired infections.13,17
In our review of 12 reported cases (Table 1), the classic triad of hemolytic anemia, jaundice, and hyperlipidemia was universally present. All patients had a history of alcohol use disorder, most with alcohol intake within 48 hours of symptom onset, and several following binge episodes.2,3,9 This supports the hypothesis that acute-on-chronic alcohol exposure may act as a trigger for Zieve syndrome.
Summary of published Cases of Zieve Syndrome: Demographics, Clinical presentation, Associated Conditions, and Outcomes.

Note: Past medical history was variably reported and often limited to alcohol use disorder.
Common comorbidities included cirrhosis, hepatitis C, spontaneous bacterial peritonitis, ascites, and pancreatitis, conditions that can either mimic or exacerbate the ZS clinical presentation.2,11,16 Notably, hepatitis C and cirrhosis may contribute to hemolysis by impairing clearance mechanisms and altering red blood cell membrane stability. 16
Among patients who received corticosteroids,8,20,21 1 experienced delayed clinical recovery, suggesting steroids may prolong the disease course. 21 Two cases had poor outcomes: 1 patient required hospice care, and another died from encephalopathy and septic shock, though these complications were not directly attributable to Zieve syndrome.2,11,18
Through our case and review, we emphasize that alcohol alone may not fully explain Zieve syndrome. Our patient’s presentation marked by alcohol use, malnutrition, and autoimmune hepatitis illustrates how hepatic vulnerability and nutritional deficiencies could contribute to Zieve syndrome. Our findings call for increased awareness, clearer diagnostic criteria, and further research into its pathogenesis and risk factors.
Footnotes
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases.
Consent for Publication
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Prior Presentation of Abstract Statement
This abstract was previously presented at the Society of Hospital Medicine (SHM) Converge Conference, held in Las Vegas, Nevada, in April 2025.
