Abstract
Gastric glomus tumor is a rare mesenchymal tumor of the gastrointestinal tract, accounting for approximately 1% of all gastrointestinal soft tissue tumors. We describe a unique case of a 27-year-old female patient who presented with recurrent episodes of overt gastrointestinal bleeding requiring multiple blood transfusions. The patient was diagnosed with a gastric ulcer detected on esophagogastroduodenoscopy (EGD), which was grossly suggestive of an ulcerated gastrointestinal stromal tumor (GIST). Preoperative diagnosis was difficult, requiring laparoscopic robotic-assisted local wedge resection of the gastric mass. Pathological diagnosis and immunohistochemical (IHC) studies were consistent with a glomus tumor. We emphasize that the gastric glomus tumor might present with life-threatening recurrent gastrointestinal hemorrhage. In addition, it might mimic GIST and require surgical resection. Pathological diagnosis and IHC studies are needed to confirm the diagnosis.
Introduction
Glomus tumors are mesenchymal neoplasms that arise from the glomus body. 1 First described by the French researchers Masson and Barre in 1924, these thermoregulatory arteriovenous shunts originate from the glomus body and are mainly located in the skin and soft tissue of distal extremities. 2 Accounting for less than 2% of soft tissue tumors, glomus tumors are commonly seen in areas with large numbers of glomus bodies, including the subungual regions of digits or the dermis in the hand and foot. 3
Very few cases of gastric glomus tumors have been described in the literature. It was first described by Kay et al 4 in 1957. These tumors are usually challenging to diagnose because of their rarity and their overlapping presentation with other, more common diseases. Accurate diagnosis requires surgical intervention and histopathologic evaluation. Overt gastrointestinal (GI) bleeding has rarely been reported as a primary presentation of gastric glomus tumors. We present a rare case of glomus tumor of the stomach where overt upper GI bleeding was the main clinical presentation.
Case Presentation
A 27-year-old female patient with no significant medical history presented with new-onset melena and lightheadedness. She was out of her hometown and started having black stools associated with lightheadedness. On her flight back, she had a syncopal episode where she passed out. She denied any chest pain, palpitations, or shortness of breath. She had never experienced these symptoms previously. She endorsed regular menstrual periods with normal flow. She denied using nonsteroidal anti-inflammatory drugs (NSAIDs) or iron supplements. Vital signs were within normal limits. The physical examination was unremarkable. Hemoglobin (Hb) was 6.5 g/dL, though red blood cell (RBC) parameters, including mean corpuscular volume (MCV) and mean corpuscular hemoglobin (MCH), were normal. She denied any previous history of anemia. The patient was started on pantoprazole. Esophagogastroduodenoscopy (EGD) showed a single 15-mm cratered ulcer with an overlying blood clot in the gastric body (Figure 1). There was no active bleeding. An endoclip was placed for site marking for any possible interventional radiology intervention if bleeding recurs. The esophagus and duodenum were unremarkable. However, Hb continued to fall, and the patient needed multiple blood transfusions. Computed tomography (CT) scan of the abdomen showed a 3-cm rounded prominence along the inferior wall of the gastric antrum adjacent to the metallic clip (Figure 2). A repeat EGD was performed, which showed a fresh clot and oozing at the previous site. No discrete vessel or lesion was visible for clipping or cauterization. Hemostasis was achieved with epinephrine injection. Her Hb then stabilized. The patient was discharged, with a Hb of 7.6 g/dL, with a plan to repeat EGD in 6 weeks to reassess the affected area and take biopsies if it does not return to normal with proton-pump inhibitors (PPIs) therapy.

EGD showing a 1- to 2-cm submucosal nodule with an overlying bloody ulcerated mucosa in (A). A fresh-appearing blood clot consistent with recent active bleeding from a mucosal ulcer is apparent in (B).
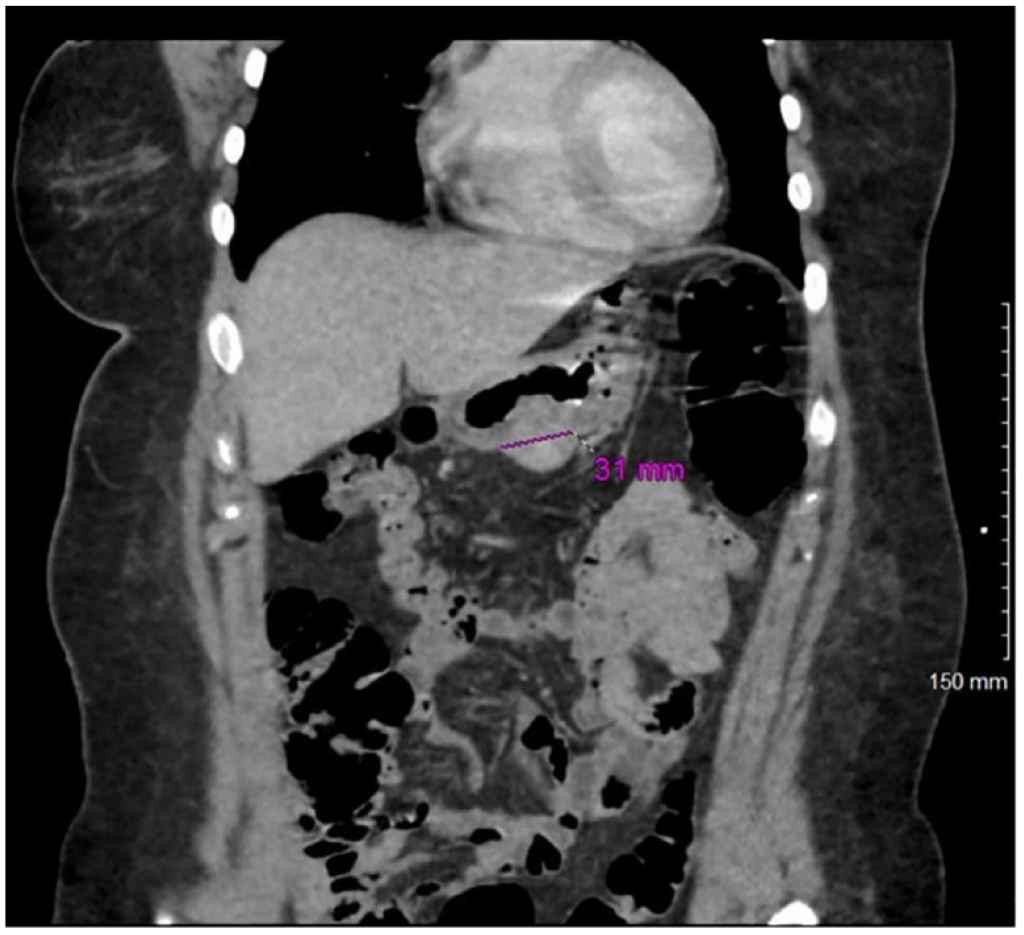
CT scan of the abdomen showing a 3-cm heterogeneous slight rounded prominence along the inferior wall.
The patient returned to the emergency department 2 days later with dizziness and melena. On arrival, blood pressure was 89/65 mm Hg. Hb was found to be 5.2 g/dL requiring transfusion with multiple units of blood and serial monitoring of Hb and hematocrit. On EGD, an ulcer with an adherent clot was seen in the gastric antrum. This appeared to arise from a 1- to 2-cm submucosal nodule, grossly suggestive of an ulcerated gastrointestinal stromal tumor (GIST). The defect was closed with endoclips. Hemoglobin continued to drop despite blood transfusions. With imaging and endoscopy findings suggestive of an intramucosal neoplasm of the stomach, the surgery team was consulted. Given the ongoing bleeding, necessitating nearly daily transfusions, the multidisciplinary team decided to proceed with laparoscopic robotic-assisted local wedge resection of the gastric mass. A 4.0 × 3.5 × 2.6 cm partial gastrectomy specimen was removed. The mucosal surface displayed a 0.7 × 0.6 × 0.3 cm hemorrhagic ulcer with diffusely raised edges. Serial sectioning of the specimen revealed a 3.6 × 2.5 × 2.0 cm tan-red, hemorrhagic, well-circumscribed mass within the stomach wall. The mass diffusely abutted the overlying mucosa at the ulcerated surface. Microscopic examination revealed a tumor located within the muscularis propria with encroachment on the submucosa and mucosa with mucosal ulceration (Figure 3A). The tumor was composed of solid sheets of neoplastic cells in a background of blood vessels with varying caliber. The cells were rounded to polygonal, with rounded nuclei having fine chromatin and pale cytoplasm (Figure 3B). Remarkably, proliferation of glomus cells around blood vessels was noted at the periphery of the lesion. Mitotic activity was less than 1 per 10 high-power fields, and no necrosis or lymphovascular invasion was seen. A battery of IHC studies was performed, revealing tumor positivity for calponin and smooth muscle actin (SMA), consistent with a glomus tumor (Figure 3C and D). Additional stains, including pan-cytokeratin, chromogranin, synaptophysin, INSM1, CD117, CD34, S100, SOX10, and pan melanoma markers, were negative, ruling out the possibilities of carcinoma, neuroendocrine neoplasm, GIST, or melanoma.
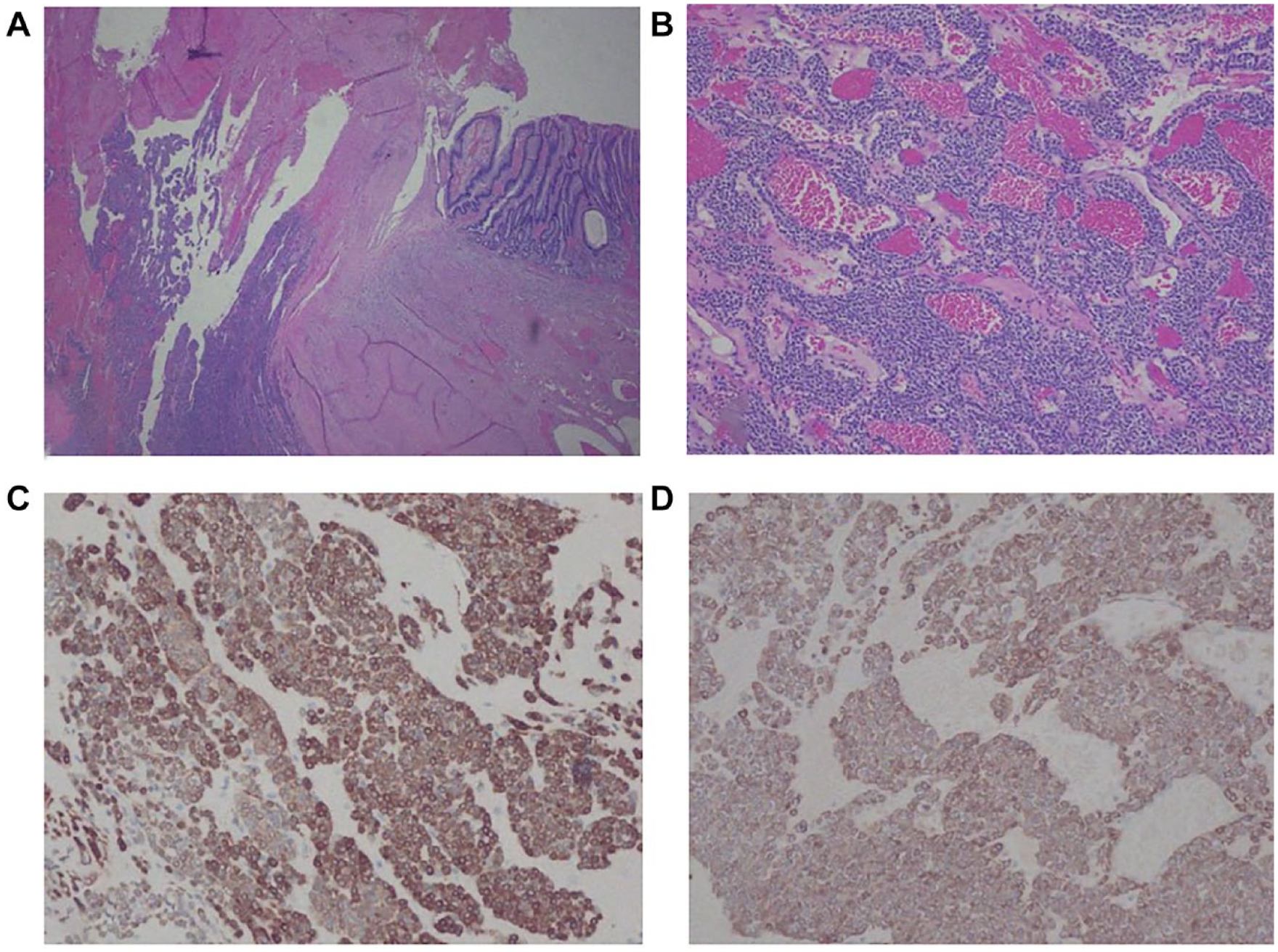
Photomicrographs of gastric glomus tumor. (A) The tumor extending into mucosa with mucosal ulceration and hemorrhage (hematoxylin and eosin, magnification 20×). (B) The classic features of glomus tumor with solid sheets of neoplastic cells in a background of blood vessels with varying caliber (hematoxylin and eosin, magnification 200×). (C) Immunohistochemical stains with calponin (magnification 200×). (D) Immunohistochemical stains with smooth muscle actin (magnification 200×).
The postprocedure course was uneventful. Computed tomography scan of the abdomen confirmed no anastomotic leakage. The fluctuation in Hb stopped, and the patient remained hemodynamically stable, with no more transfusion requirements. Eventually, the patient was discharged on postoperative day 8. She received a total of 9 units of packed RBCs throughout her stay. On a follow-up visit 4 weeks later, she reported a complete resolution of symptoms.
Discussion
Gastrointestinal glomus tumors are rare, with very few documented cases of glomus tumors in the stomach. In most reported cases, the tumor was located in the gastric antrum. 5 A review of literature on gastric glomus tumors by Duan and Chetty 6 showed a female preponderance, with a median age of presentation of 54 years (range: 18-90).
Gastric glomus tumors present with an array of symptoms, ranging from asymptomatic anemia to overt GI bleeding in the form of melena in larger lesions complicated by ulceration. In their review of a series of gastric glomus tumors, Duan and Chetty 6 noted epigastric pain to be the most common presenting symptom (46%). Gastrointestinal bleeding was noted in a quarter of the cases, while another quarter was asymptomatic, where glomus tumors were discovered incidentally during surgery for other indications. Other symptoms included nausea, vomiting, and fatigue. The symptom duration varied, ranging from a week up to 10 years among the patients investigated by Fang et al. 7 The present case fits into this profile, as the patient was a young woman showing the most common symptoms with anemia and recurrent GI hemorrhage.
A glomus tumor is composed of 3 parts: glomus cells, smooth muscles, and blood vessels. Table 1 8 describes the 3 subtypes of glomus tumors based on the relative proportion of these components. On a genetic level, Mosquera et al 9 identified changes in the Notch signaling pathway, which regulates vascular smooth muscle development, in more than half of the 33 glomus tumors they screened. Glomulin gene 10 mutations have been linked to familial glomangiomas, which present as multifocal lesions.
Subtypes of Glomus Tumors.

Grossly, the tumor is well circumscribed, 11 with a median size of 2.5 cm. It is located in the submucosa or muscularis propria of the gastric wall, with a tendency to bulge inwards toward the mucosa or outwards toward the serosa. 12 The cross-section may be soft to rubbery and commonly exhibits signs of hemorrhage. 12
Barium study usually shows gastric glomus tumors as submucosal masses causing a filling defect. 7 Owing to their vascularity, they show strong enhancement 13 on the arterial phase of contrast-enhanced CT. Their homogenous density and relatively sharp demarcation can also be seen on nonenhanced CT. On magnetic resonance imaging (MRI), they tend to show hypo- and hyperintensities on T1- and T2-weighted images, respectively. They are hypervascular, with persistent enhancement after administration of gadopentetate dimeglumine. 14 Endoscopic ultrasound (EUS) can aid in identifying the layer of origin, with gastric glomus tumors commonly arising from the third and fourth layers (submucosa and muscularis propria). Muscularis propria surrounding the tumor cells is seen as a characteristic peripheral halo. 15 Internal calcification may show up as internal hyperechoic spots. Heterogeneity on EUS may be due to hemorrhage or calcification. The American College of Gastroenterology recently published guidelines recommending EUS with tissue acquisition via fine-needle biopsy alone or fine-needle aspiration with rapid on-site evaluation to improve diagnostic accuracy in identifying solid nonlipomatous subepithelial lesions. 16 A EUS-guided biopsy is associated with the risk of bleeding and inadequate tissue acquisition.6,17
The radiological findings described above may be seen to a certain extent in various hypervascular gastric tumors such as GIST and neuroendocrine tumors. This is where pathological evaluation with IHC analysis plays a pivotal role in accurate diagnosis. Microscopic examination reveals vasculature surrounded by small, monomorphic cells. 11 Cellular atypia, pleomorphism, and necrosis are rare. 18 Tumor stroma may occasionally show hyaline or mucous degeneration and calcification or ossification. Immunohistochemical is usually suggestive of smooth muscle differentiation, with strong positive staining 19 for SMA, vimentin, calponin, type IV collagen, and laminin.
With overlapping clinical and radiological findings, it is crucial to distinguish gastric glomus tumors from other mesenchymal tumors. These include GIST, neuroendocrine tumors, schwannoma, smooth muscle tumors, lymphomas, metastasis, and other rare lesions such as inflammatory fibroid polyp, inflammatory myofibroblastic tumor, and desmoid fibromatosis. Gastrointestinal stromal tumors are the most common 20 mesenchymal tumors in the GI tract. Given that incidence of gastric glomus tumors is estimated to be 100 times less 21 than GISTs, gastric glomus tumors are often misdiagnosed as GISTs, like in the current case. Though endoscopic findings may be similar, GISTs are distinctively positive for CD117, DOG1, and CD34 22 on IHC. On the contrary, neuroendocrine tumors have cells with salt and pepper chromatin patterns in their nuclei and stain positive for neuron-specific enolase, synaptophysin, INSM1, and chromogranin. 23 Though it may be challenging to differentiate gastric glomus tumors from lymphomas on frozen section biopsies, the lineage and developmental stage of lymphomas can be recognized with IHC and flowcytometric analysis. Neurogenic tumors usually arise in the submucosa and are diffusely positive for S100 but lack activity for CD117, DOG1, SMA, and desmin. Smooth muscle tumors, such as leiomyomas and leiomyosarcomas, are usually diffusely positive for desmin and SMA but are negative for DOG1 and CD117. Desmoid fibromatosis usually arises extrinsic to the GI tract and demonstrates nuclear labeling for B-catenin.
Surgical treatment with a wedge or segmental resection 24 is the preferred management modality. Endoscopic enucleation 25 carries the risk of bleeding and perforation. The exact rate of recurrence of gastric glomus tumors has not been documented, though cases of recurrence have been reported in cases series 12 of glomus tumors. Folpe et al 25 described histological criteria for identifying malignant glomus tumors. They proposed that deep-seated tumors, >2 cm in size, with atypical mitotic features, moderate to high nuclear grade, and ≥5 mitotic figures per 50 high-power field (HPF), can be used as criteria for identifying malignant glomus tumors. They observed that 38% of tumors meeting these criteria had metastases in their cohort. However, these are not specific for “gastric” glomus tumors. One of the patients with a gastric glomus tumor out of 32 studies by Miettinen et al 12 died 4 years after initial presentation with hepatic metastases. Criteria for the malignant potential for gastric glomus tumors have not yet been laid down, though malignant gastric glomus tumors have been documented26,27 in literature based on histopathological findings. It may be difficult to differentiate between multicentric and metastatic diseases. Surveillance after removal, especially for tumors that are large with atypical histological features, can help monitor for recurrence.
Here, we report a very unusual case of a primary GI tumor presenting with recurrent GI bleeding. The tumor mimicked GIST and required surgical resection along with pathological and IHC studies to reach a definitive diagnosis. This type of tumor is very rare and rarely metastasizes. Thus, timely clinical examination and careful histologic evaluation are critical for diagnosing glomus tumors.
Footnotes
Acknowledgements
The authors would like to thank Amira Hassan, Robert Scott Strobach, and Bradley Carmichael for their help with establishing a pathological diagnosis. We also want to thank Hasan Bader for his help with the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
