Abstract
Morbidity and mortality associated with radiation-induced secondary malignancies (RISMs) have shifted treatment paradigms to minimize or eliminate radiation from treatment regimens. In this case, a 48-year-old woman was diagnosed with Hodgkin lymphoma (HL) and treated with radiotherapy in 2000. In 2018, she was diagnosed with ductal carcinoma in situ (DCIS) of the right breast and treated with a mastectomy. Soon after, she developed triple-negative invasive ductal carcinoma (IDC) in her reconstructed breast. The patient underwent a left lumpectomy, and pathology showed ER–/PR–/HER2+ IDC. This patient’s multi-phenotypic DCIS and IDC presentation are suspected to be RISM due to her previous HL treatment regimen.
Introduction
Radiotherapy (RT) is an integral part of cancer treatment, and nearly, 50% of patients with cancer will require RT as part of their treatment regimen.1,2 Although RT is a vital and effective treatment modality in the treatment of various types of cancer, it is a “double-edged sword” that comes with the potential for secondary malignancies, termed radiotherapy-induced secondary malignancies (RISMs). 3 Radiotherapy-induced secondary malignancy is seen in cancer survivors, constituting 19% of all cancer diagnoses in the United States.3,4 Cancer survivors have a 14% higher cancer rate than the general population, often as a direct result of prior treatments.3,4 The pathogenesis of RISM is due to various factors, including patient age at the time of exposure, the type of irradiated organ or tissue, the specific type of RT, the given dose and field size of radiation, concurrent use of chemotherapeutics, accompanying lifestyle habits, and genetic predispositions. 1
Survival rates for Hodgkin lymphoma (HL) patients have significantly improved over the past few decades due to developments in treatment techniques.5,6 However, RT’s long-term and late effects are becoming more apparent and can be challenging to treat, as survivors live long enough to develop RISM.1,7 Strikingly, secondary malignancies are the leading cause of death in patients with a history of HL, with survivors having an increased risk of breast cancer 8-fold.8,9 In this case report, we discuss a 48-year-old woman who developed HL and multi-phenotypic breast cancer 18 years after her initial diagnosis and treatment.
Case Report
This patient was a 48-year-old woman with a history of stage III HL that was treated with chemotherapy and radiation via mantle/inverted Y radiation fields in 2000. In follow-up, she was judged to be cured. In 2018, she was diagnosed with ductal carcinoma in situ (DCIS) of the right breast and was treated via modified radical mastectomy with 4 lymph nodes removed. All excised nodes were negative for malignancy. During this same period, the patient had a prophylactic contralateral simple mastectomy. The patient presented in 2020 with a mass in the upper outer quadrant of her surgically reconstructed right breast. A biopsy confirmed triple-negative (ER–/PR–/HER2–) invasive ductal carcinoma (IDC) (Figure 2A-2F). Staging imaging done at that time showed bone metastases. While receiving palliative capecitabine, she was found to have a new mass in the reconstructed left breast. She underwent a left lumpectomy, in which pathology showed IDC. This IDC was ER–/PR–/HER2+, phenotypically different from her recently diagnosed right breast cancer (Figure 2E and 2F). The patient was also noted to have a malignant pleural effusion, which was sampled and found to be triple-negative breast IDC. After several cycles of capecitabine, restaging computed tomography (CT) imaging showed progressive liver and pulmonary metastases. To treat her progressive triple-negative disease in parallel and to treat the possibility of concurrent HER2+ metastatic deposits, she was started on sacituzumab with concurrent trastuzumab/pertuzumab (Figure 1).

Timeline of patient’s diagnoses and secondary malignancy.
It is suspected that this patient’s diagnosis of DCIS and IDC were likely RISM, caused by her original HL treatment regimen (Figure 2). There was no family history of breast cancer. The oncogenic potential of RT is demonstrated in this patient by the 2 distinct breast cancer phenotypes: triple-negative IDC and ER–/PR–/HER2+ IDC. This patient’s scenario highlights the challenges associated with curative therapies and the need to reduce the risk of development of secondary malignancy. This study required a retrospective review of Electronic Health Records and literature review.
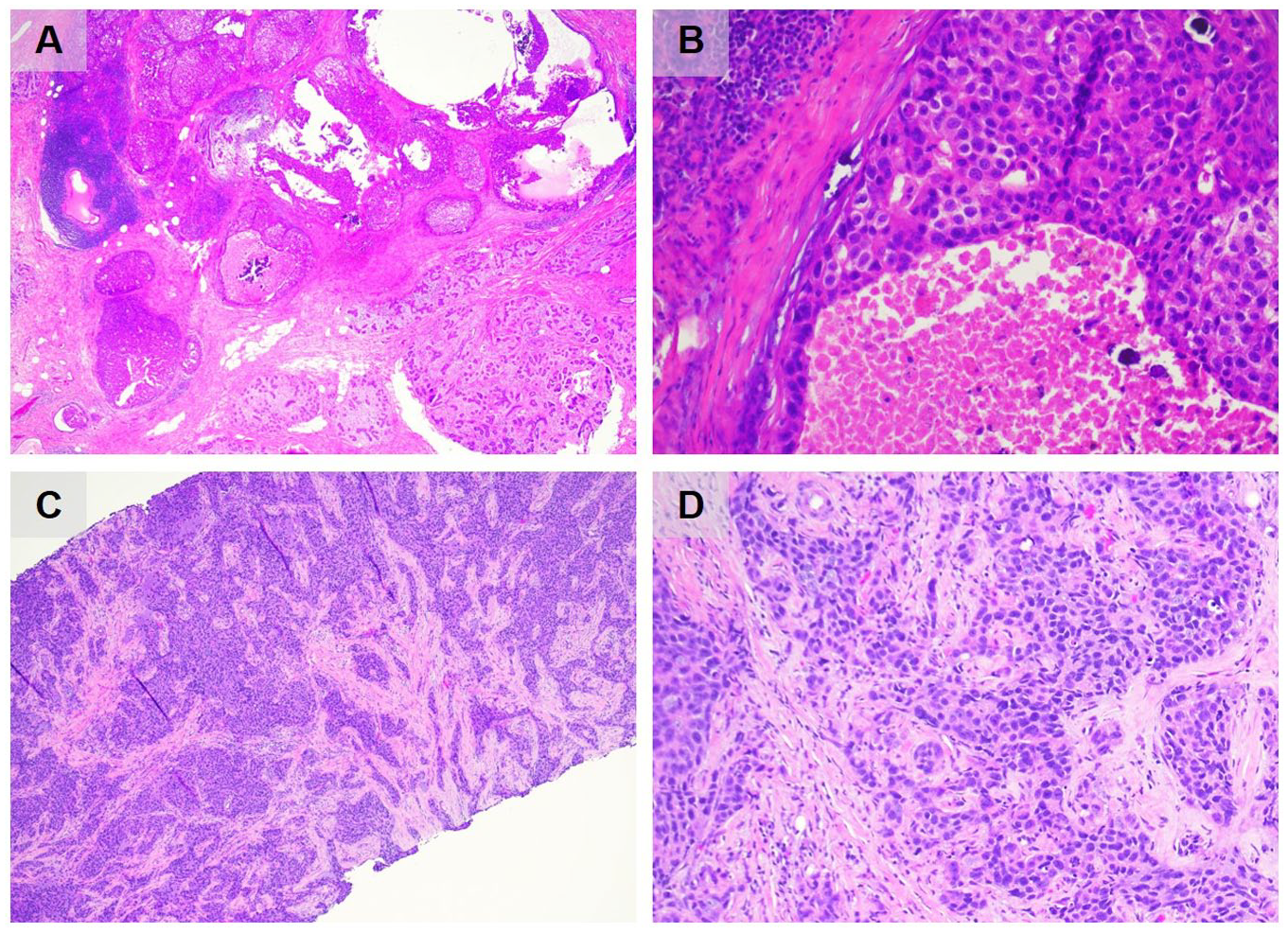
(A) H&E image of patient’s DCIS with solid and comedo patterns at 4× magnification. (B) H&E image of patient’s high nuclear grade DCIS at 40× magnification. (C) H&E image of grade 3 invasive ductal carcinoma of no special type at 4× magnification. (D) H&E image that demonstrates solid nest of neoplastic cells with significant nuclear pleomorphism in patient’s ductal carcinoma at 40X magnification.
Discussion
Although RT has been an essential component of cancer treatment for decades, its carcinogenic properties are well-known. Radiation causes double-stranded DNA breaks, leading to genetic mutations and eventually malignant transformation of the irradiated cells. 10 Because of the increased risk of developing RISM, numerous variables must be considered treating HL patients with radiation. 11 Radiation exposure during childhood dramatically increases risk of RISM compared with receiving it at an older age. 11 Older radiation techniques have been shown to increase the risk of RISM, typically involving larger radiation fields and non-specific targeting. 12 Newer methods have been developed only to include the nodes and sites involving HL.13,14 In addition, it is estimated that each Gy of radiation increases the rate of solid cancers by about 58% in females.13,14 Furthermore, the total intensity of chemotherapy and radiation therapy correlates with the risk of second malignancy after Hodgkin disease. 11 In addition, female patients have a greater risk of developing RISM compared with males. 15
Particularly for female patients with HL, RISM is associated with an elevated risk of breast cancer later in life.8,9 Despite consistent breast cancer screening and aggressive surgical prophylaxis, this patient developed multi-phenotypic breast cancer with metastatic disease. If diagnosed today with stage III HL, this patient would likely be treated with multi-agent chemotherapy alone with ABVD (doxorubicin (Adriamycin), bleomycin, vinblastine, and dacarbazine), had interval positron emission tomography (PET), then either continued ABVD or had therapy switched to escalated BEACOPP (bleomycin, etoposide, doxorubicin (Adriamycin), cyclophosphamide, vincristine (Oncovin), procarbazine, and prednisone). Thus, RT would likely not be required. Much narrower fields would be used if radiation therapy is needed rather than the relatively large field mantle/inverted Y technique.
Conclusion
Understanding the associated risk factors and the timeline from RT to the development of secondary malignancy have essential implications in the initial design of future treatment plans. Comprehensive and consistent screening practices, and lifestyle interventions, such as quitting smoking, reducing alcohol consumption, daily exercise, and weight loss also, may be effective in preventing RISM. Furthermore, oncologists should provide specific instructions for the patient’s family physician to prevent and detect RISM, as a comprehensive and multidisciplinary approach is required to manage patients’ post-RT properly. Although immense progress has been made in understanding RT for HL, further research must be done to determine the best treatment regimens for patients at risk of RISM.
Footnotes
Author Contributions
D.W.: design, writing, and editing; M.S.S.: figure design, editing; B.B.: conceptualization, supervision, and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
All images comply with journal requirements and have no identifying information of the patient to the best of our knowledge. Institutional Review Board’s approval was not required for this case report. Ethical approval is not required for this study in accordance with local or national guidelines.
Informed Consent
Informed consent for patient information to be published in this article was not obtained because the patient is deceased and their next of kin were non-responsive when contacted for consent. All patients’ data have been de-identifiable.
Prior Presentation of Abstract Statement
The abstract for this case report was previously presented at the Southern Regional Meeting sponsored by the Southern Society of Clinical Investigation in New Orleans, LA, on February 22, 2023.
