Abstract
With the previous worldwide initial coronavirus disease 2019 (COVID-19) pandemic, a notable rise in spontaneous pneumomediastinum with/without pneumothorax (SPP) has been noted. Most cases were initially reported as complications secondary to barotrauma from mechanical ventilation (MV) with COVID-19. However, with the Delta strain, starting from December 2020, there have been multiple reports of SPP. The SPP is an uncommon complication outside use of assisted ventilation with either noninvasive positive pressure ventilation (NIPPV) or MV. COVID-19 has been linked to higher incidence of SPP without use of NIPPV or MV. We present a series of 5 cases with a polymerase chain reaction–confirmed COVID-19 diagnostic testing whose hospital course was complicated by SPP unrelated to the use of either NIPPV or MV.
Introduction
Spontaneous pneumomediastinum with/without pneumothorax (SPP) is an uncommon complication outside the use of assisted ventilation with noninvasive positive pressure ventilation (NIPPV) and mechanical ventilation (MV) with a prevalence of 0.002%.1-3 During the coronavirus disease 2019 (COVID-19) pandemic, there has been an increase in SPP, most seen with the Delta strain. 1
Pulmonary barotrauma results in air leakage due to wall rupture of marginal pulmonary alveoli from high intra-alveolar pressure usually due to artificial positive pressure ventilation, forceful coughing, vagal maneuvers, or straining. 4 The SPP is described as free air or gas in the mediastinum and/or pleural space that is not caused by chest trauma. 4 Barotrauma commonly occurs with positive pressure ventilation, however, is less common with use of high flow oxygen through nasal canula. 4
Case Descriptions
In this section, we will present individual case descriptions. Please note that all patients were initially managed with conservative measures which are oxygen supplementation, dexamethasone 10 mg intravenously, 5 baricitinib 4 mg by mouth once daily, Remdesivir, and self-proning as tolerated. In addition, all patients were admitted from the emergency department (ED), had positive COVID polymerase chain reaction (PCR) test, and managed initially in a step-down unit. Table 1 summarizes important clinical data points.
Summarization of Cases.

Abbreviations: SPP, spontaneous pneumomediastinum with/without pneumothorax; HHFNC, heated high-flow oxygen via nasal cannula; CVA, cerebrovascular accident; CRP, C-reactive protein.
Case 1
A 60-year-old man with medical history significant for diabetes mellitus, gastroesophageal reflux disease, no prior tobacco use and cerebrovascular accident, was admitted to the hospital for acute hypoxic respiratory failure secondary to COVID-19 pneumonia. He was diagnosed with COVID-19 infection 6 days prior to presentation. He was not vaccinated against COVID-19. On admission, he was requiring 100% fraction of inspired oxygen (FiO2) through heated high-flow oxygen via nasal cannula (HHFNC) at 60 L/m. C-reactive protein (CRP) was 8.82 mg/dL (range 0.00-0.30 mg/dL),

Computed tomography chest showing spontaneous pneumomediastinum with/without pneumothorax in patient 1.
Case 2
A 35-year-old man with medical history significant for obesity and 10-pack-year smoking history was admitted to the hospital for a 10-day history of worsening shortness of breath after he had been diagnosed with COVID-19. Chest x-ray (CXR) showed bilateral infiltrates and patient was requiring up to 3 L of oxygen through nasal canula. He was not vaccinated against COVID-19. On admission, CRP was 4.34 mg/dL and

Patient 2: spontaneous pneumomediastinum diagnosed with computed tomography chest.
Case 3
A 54-year-old man with a past medical history significant for obesity, gastric cancer with resection and adjuvant chemotherapy 10 years prior (in remission), and no prior tobacco use presented to the ED for worsening shortness of breath requiring 5 L of oxygen. He was not vaccinated against COVID-19. Initial CXR showed bilateral infiltrates consistent with COVID-19 pneumonia. His COVID-19 PCR was subsequently positive. He required HHFNC 45 L/m at 55% FiO2 shortly after admission. C-reactive protein was 5.24 mg/dL and

Chest computed tomography demonstrating subcutaneous emphysema in the neck.

Chest computed tomography demonstrating extensive pneumomediastinum.
Case 4
A 28-year-old obese man with no prior tobacco use presented to the ED with headache and nausea after he was diagnosed with COVID-19 at an outpatient clinic where a CXR showed bilateral infiltrates. The patient was started on 2 L of nasal cannula due to persistent hypoxia. He was not vaccinated against COVID-19. The CTPA on admission was negative for any pulmonary embolus, pneumothorax, or pneumomediastinum but showed multifocal ground-glass infiltrates consistent with COVID-19. Patient was begun on HHFNC on day 2 of admission. C-reactive protein was 11.8 mg/dL,
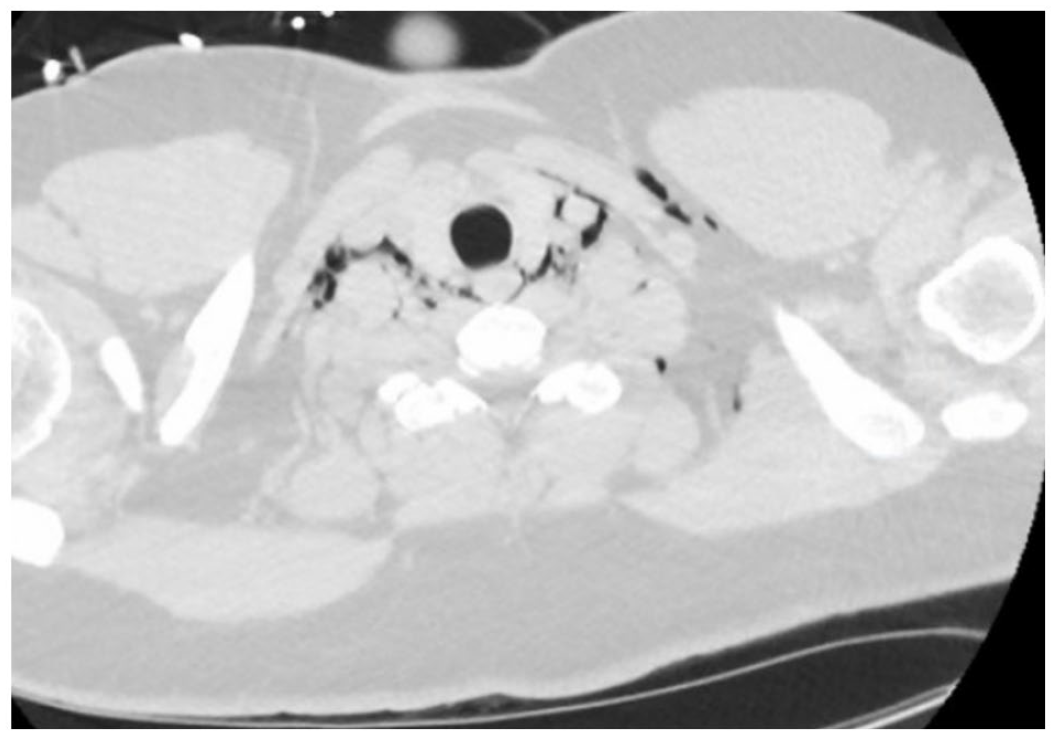
Patient 4: extensive subcutaneous emphysema in the neck.

Chest computed tomography again showing extensive subcutaneous emphysema in patient 4.
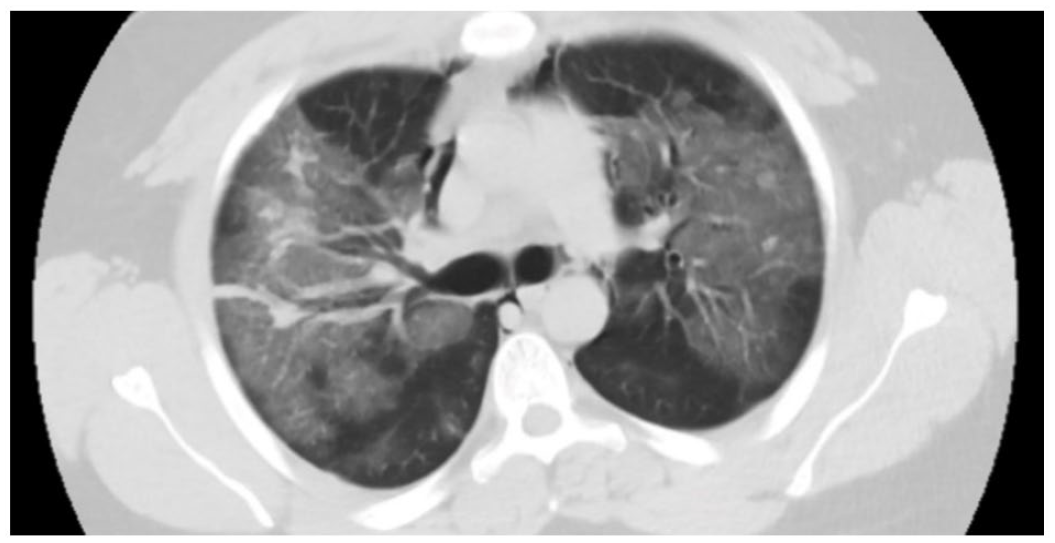
Chest computed tomography showing pneumomediastinum in patient 4.
Case 5
A 38-year-old woman with a past medical history of alpha-1 antitrypsin deficiency, chronic obstructive pulmonary disease, diabetes mellitus type II, obesity, chronic migraine headaches, and no prior tobacco use presented to ED 8 days after testing positive for COVID-19 experiencing worsening shortness of breath, fevers, and body aches. The patient had received casirivimab-imdevimab infusion but was noted to have SaO2 of 80% on 4 L nasal cannula. She was not vaccinated against COVID-19. She was subsequently placed on HHFNC 50 L/m at 100% FiO2 and admitted to the hospital. The CTPA showed diffuse bilateral ground-glass opacities intermixed with areas of consolidation and air bronchograms with several prominent right paratracheal lymph nodes. There was no evidence of pulmonary embolism, pneumothorax, or pneumomediastinum. Admission
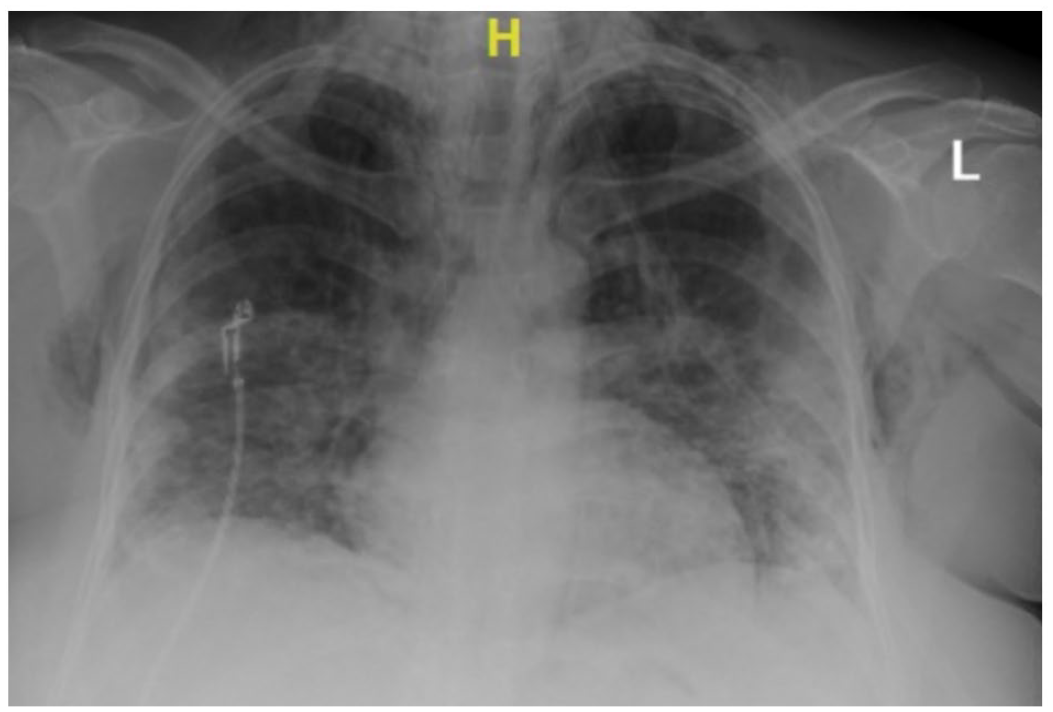
Chest x-ray demonstrating extensive subcutaneous emphysema of the chest wall and pneumomediastinum.
Discussion
This case series represent SPP without use of NIPPV or MV in HCA West Florida Brandon hospital during the Delta variant strain of COVID-19 spread in the United States. Noncompliant lungs in the face of miniscule pressure can develop spontaneous pneumothorax/pneumomediastinum in COVID-19 ARDS as suggested by Lemmers et al. 6 In his study, there was no difference among incidence of barotrauma in patients mechanically ventilated using low tidal volume ventilation and low pressure defined as plateau pressures 23 ± 4 cmH2O. This population of patients does not necessarily reflect our patient population as these patients have been on HHFNC which at near maximum inspiratory flow induces a mean ± SD airway pressures at 30, 40, and 50 L/min were 1.93 ± 1.25 cm H(2)O, 2.58 ± 1.54 cm H(2)O, and 3.31 ± 1.05 cm H(2)O, respectively. However, our HHFNC goes up to 60 L of flow per minute. Other potential confounders are that our patients were using incentive spirometry as there have been case reports of patients using incentive spirometers causing pneumothorax/pneumomediastinum and a case of secondary spontaneous pneumothorax in patients receiving incentive spirometers with noncompliant lungs.7-9 Incentive spirometer is used mostly in the postoperative setting to encourage long, deep breaths to prevent atelectasis and pneumonia which increases inspiratory volumes and promotes lung expansion. 9 This has commonly become employed in COVID-19 ARDS as it has been proposed that the mechanism of action of incentive spirometry to improve ventilation/perfusion mismatch and alveolar-PaO2 gradient thus reducing the intrapulmonary shunting in which is proposed to happen in COVID-19 ARDS. 10
Thus, it seems this could be a possible combination of factors such as frail noncompliant lungs and the combination of IS. Other possible contributors include oxygen toxicity from 100% FiO2 which results in free radicals causing endothelial damage with resultant increased adhesiveness and accumulation of neutrophils in the alveolar space with subsequent collagen deposition. 11 It is also believed that HHFNC generates a small amount of positive end expiratory pressure.12,13 All these factors combined would theoretically increase the chances of barotrauma; in addition to COVID-19 alveolar damage. Tobacco smoking was not an important risk for developing SPP in our patients’ cohort.
Pneumomediastinum complicating COVID-19 has been treated conservatively, even with small pneumothorax, in prior case reports. 14 The decision to place a chest tube has been an individual one, baring into consideration severity of the disease and expansion of pneumothorax with little extra guidance from prior published guidelines in spontaneous secondary disease. 15
Mortality from pneumomediastinum is mostly related to the underlying etiology and treatment is usually geared toward associated pneumothoraces. 16 Morbidity from pneumomediastinum is difficult to quantify. Possible contributing factors to increased morbidity are increased pain (as in our cases), increased monitoring time, thus increasing hospital length of stay, increased cost of care, days lost of work, and stress/cost of follow-up imaging and health care contact time. Pneumothorax has been shown to double mortality related to COVID-19. 17 This significant (and independent) risk of death also contributed to mortality in patients on MV in the same study. Another interesting finding is that most patients developed SPP before MV or noninvasive ventilation, which has been replicated in other studies. 17 Larger and longer-term follow-up studies are needed to examine whether SPP confers an increased risk for future pleural disease in patients with COVID-19.
We reported a unique finding of SPP in our patients receiving HHFNC, with relevant clinical data and radiological images. We have also reported laboratory data, clinical trajectory, and any interventions needed. There are several limitations to our study other than its inherent limitation of being retrospective; mainly the potential overestimation of smoking effect, vaccination status, and treatment offered. Another limitation is the possible missed cases of SPP as some cases were diagnosed incidentally. Furthermore, we could not confidently rule out prior undiagnosed lung disease in our cohort. With all these limitations, we think our study highlights the importance of SPP in COVID-19 patients, its clinical description, radiological features, expected recovery, and potential areas of further research.
Conclusion
The SPP seems to have a temporal relation with HHFNC in COVID-19 patients at our institution, occurring more frequently with the Delta strain. This may be due oxygen toxicity, severity of inflammation, use of IS, or small positive pressure generated by HHFNC. No specific treatment is available for SPP short of conservative measures. Further high-quality studies are needed to address the relationship between HHFNC and SPP, and how would that affect long-term incidence of pleural disease.
Footnotes
Acknowledgements
We want to thank the patient for allowing us to share this with the medical literature.
Authors’ Note
This case was submitted as a poster to Florida ACP.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in the article.
