Abstract
Chronic lymphocytic leukemia (CLL) is the most common leukemia in adults and is characterized by monoclonal proliferation of B-cell lymphocytes which are morphologically mature, but immunologically dysfunctional. The primary sites of disease involvement include peripheral blood, lymph nodes, spleen, and bone marrow. CLL can also present locally and aggressively at extranodal sites. We describe the case of a 74-year-old gentleman with multiple medical comorbidities who was Foley catheter–dependent at baseline for bladder outlet obstruction. He was detected to have Rai stage I CLL following an inguinal lymph node biopsy and was on regular outpatient surveillance. Later, he underwent a prostate biopsy for evaluation of hematuria, results of which were consistent with CLL involvement in the prostate and urinary bladder. The patient was started on single-agent ibrutinib, and demonstrated an excellent clinical response to bladder outlet obstruction. His long-term Foley catheter was discontinued within 5 days of ibrutinib therapy. Unfortunately, 1 year later, he had disease progression, and therapy was changed to a single-agent rituximab, to which he is responding well. Our case is unique as it brings up the first reported case of prostate and bladder wall CLL.
Introduction
Chronic lymphocytic leukemia (CLL) is a lymphoproliferative disorder characterized by progressive accumulation of immunologically dysfunctional CD-5-positive B lymphocytes. The disease is referred to as CLL when it is limited to the blood and small lymphocytic lymphoma (SLL) when the disease progresses to the lymphoma phase with lymph node involvement. The median age at diagnosis is 70 years. CLL most commonly affects the blood, bone marrow, and secondary lymphoid tissues (liver and spleen), and is usually asymptomatic at diagnosis. Rare sites of disease involvement include skin, breast, and meninges.
Case Report
A 74-year-old male with a past medical history of stroke (wheelchair-bound at baseline), dyskinesia of upper extremity, seizure disorder, benign prostatic hyperplasia (Foley dependent), paroxysmal atrial fibrillation, hyperlipidemia, and chronic obstructive pulmonary disease (COPD), was hospitalized for generalized weakness.
Initial laboratory evaluation showed the presence of lymphocytosis. Computed tomography (CT) chest, abdomen, and pelvis showed multiple mediastinal and inguinal lymphadenopathy (Figure 1). He underwent a CT-guided inguinal lymph node biopsy; the results were consistent with chronic lymphocytic leukemia (CLL). Fluorescence in situ hybridization (FISH) studies was negative for deletion of 11q, 17p, and TP 53, and positive for trisomy 12 and deletion of chromosome 13, all of which were consistent with a diagnosis of CLL. He met the diagnostic criteria for Rai stage I; hence the decision was made for regular outpatient hematology/oncology surveillance.

Computed tomography imaging showing axillary lymphadenopathy (a), porta hepatis lymphadenopathy (b), and inguinal lymphadenopathy (c).
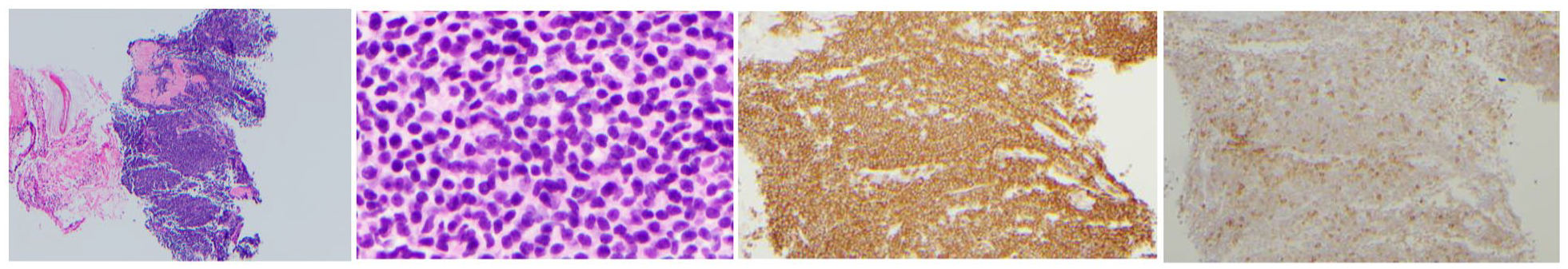
One year following the diagnosis of CLL, he had an episode of hematuria. On further evaluation, he was found to have an enlarged prostate with calcification at the apex. He subsequently underwent a prostate biopsy which showed atypical lymphocytic infiltrates, consistent with CLL involvement of the prostate gland. Considering the disease progression and performance status of the patient, he was started on single-agent ibrutinib. He showed an excellent symptomatic response to the therapy with a successful trial of void, and his chronic foley was discontinued within 5 days of the initiation of ibrutinib therapy. He was placed on a maintenance dose of ibrutinib. But unfortunately 14 months into the treatment, he was detected to have nodularity at the base of the urinary bladder, which was concerning for bladder carcinoma. He underwent a cystolitholapaxy with bladder wall biopsy, which was positive for dense lymphoid infiltrates consistent with lymphoma involvement of the bladder.
As the patient had disease progression while on ibrutinib, and considering the poor performance status with multiple medical comorbidities, the decision was made to proceed with single-agent rituximab. He completed 4 weekly doses of rituximab and was transitioned to a maintenance dose of rituximab monthly. Currently, the patient is on regular outpatient follow-up and has not had another further episode of recurrence.

Discussion
CLL is the most common leukemia in adults, accounting for 25% to 35% of all leukemias in the United States. 1 CLL is a malignancy of CD-5 positive B lymphocytes, characterized by the accumulation of mature-appearing lymphocytes in the blood, bone marrow, and lymphoid tissues.
Patients are usually asymptomatic at presentation. About 50% to 90% of the patients can present with painless lymphadenopathy, and 5% to 10% can present with “type B” symptoms including fever, night sweats, weight loss, and extreme fatigue.2,3 CLL can involve any lymphoid tissue including liver, spleen, and Waldeyer’s ring in the pharynx. Less common presentations of CLL include leukemia cutis of the skin affecting less than 5% of the cases, meningeal leukemia in less than 2% of the cases, and membranoproliferative glomerulonephritis.4-6 Prostate and bladder involvement by CLL has never been reported in the past, and this is the first reported case, which makes our case unique.
Laboratory abnormalities include lymphocytosis with anemia and thrombocytopenia. Immunophenotypic analysis using flow cytometry is an important component in the diagnosis of CLL. The 3 most common flow cytometry findings include (1) positive expression of B-cell antigens CD 19, CD20, and CD23; (2) positive CD-5, expressed on mature B-cells; and (3) low levels of surface membrane immunoglobulins including IgM and IgD. 3 FISH is used to evaluate tumor markers in CLL. The mutations which are commonly tested in CLL include deletion of chromosome 17p, 11q, 13, trisomy 12, and TP53 mutations.7,8 Our patient tested negative for TP53 mutation, 11q and 17p deletion, but positive for deletion of chromosome 13 and trisomy 12.
The Rai staging system is a prognostic system that uses physical examination and laboratory results to stratify patients with CLL into 3 groups. 2 Low-risk group/stage 0 has only lymphocytosis, intermediate-risk group/stage I and II has lymphadenopathy, liver and/or spleen involvement, and high-risk group/stage III and IV has anemia (hemoglobin <11g/dL) and thrombocytopenia (platelet count <100 000/mm3). 2 With recent advances, flow cytometry, immunophenotypic, and molecular markers are also used as prognostic indicators. 9 Overexpression of CD38, CD49d, deletion of 17p, 11q, and TP53 mutations are associated with poor prognosis and usually follow an aggressive clinical course, compared to 13q deletion and trisomy 12. 9
Patients who are asymptomatic and have early-stage disease (Rai stage 0) are closely monitored with a wait-and-watch approach and do not require chemotherapy. 9 Treating these patients is not associated with any survival benefits. 10
Therapy is recommended for symptomatic patients and those with disease progression. 3 There are several FDA-approved drugs for the treatment of CLL that are used either as monotherapy or in combinations. Chemo-immunotherapy with fludarabine, cyclophosphamide, and rituximab remained as the standard of care for years in younger CLL patients <65 years with good performance status. 11 Ibrutinib is an irreversible Bruton tyrosine kinase (BTK) inhibitor that induces apoptosis of B-cells and is used in patients with previously untreated CLL. 12 Since its introduction in 2016, ibrutinib has been used as a monotherapy and combination therapy both in the first-line setting and relapsed/refractory setting. 9 The RESONATE-2 is a phase III, open-label, randomized study that established ibrutinib monotherapy as a first-line agent in CLL. 13 The combination of ibrutinib with rituximab has shown superior efficacy in younger patients to the above-mentioned chemo-immunotherapy as evidenced by progression-free survival and overall survival. 14
Literature has shown that patients with CLL have a higher risk of developing a second primary malignancy in comparison to patients without CLL, and this phenomenon is attributed to dysregulation of the immune system. 15 A retrospective analysis and genome sequencing study by Steiner et al 16 postulated that a rare germline polymorphism may predispose to development of prostate cancer in a patient with CLL.
The 3 most common solid organ malignancies that occur in patients with CLL are prostate, breast, and lung cancer, 15 although other malignancies may arise. Chawla et al 17 described the case of a 61-year-old male was found to have coincident malignancies with both CLL and high-grade urothelial carcinoma. Early recognition of a second primary malignancy is essential to correctly staging and managing disease in this subset of patients.
Almost all patients with CLL will relapse at some point. 9 Patients with 17p deletion are at increased risk of relapse. 18 Venetoclax is a highly selective BCL2 inhibitor (a protein expressed on CLL cells) approved for use in relapsed CLL or SLL including those with poor prognosis. 19 Anti-CD20 antibodies which are approved in CLL include rituximab, obinutuzumab, and ofatumumab. 9 Following binding with CD20, they activate complement-mediated cytotoxic activity. These agents are either used as monotherapy or used in combination with chemotherapy. 9
Conclusion
CLL is the most common leukemia in adults and is usually asymptomatic at presentation. CLL usually involves the peripheral blood, lymph nodes, spleen, and bone marrow, with skin and meninges being the less common sites. Prostate and bladder wall involvement of CLL has not been reported in the past, and this is the first reported case. Asymptomatic cases can be monitored for years without any intervention. There are several treatment options available in the first-line setting and also in the relapsed/resistant setting depending on the molecular characteristics and performance status of the patient.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Informed consent for patient information to be published in this article was obtained.
