Abstract
Aortic dissection is an infrequent diagnosis that usually presents with acute onset of sharp and severe tearing pain. It rarely presents with atypical symptoms, accompanied by a higher mortality risk that arises the delay in diagnosis. In this report, we discuss a type A aortic dissection case with a presentation of heaviness-like chest pain with no evidence of aortic dissection in his first echocardiography. The patient was treated for acute coronary syndrome (ACS), but on the follow-up, echocardiography aortic dissection was diagnosed accidentally. Differentiation between ACS and aortic dissection is critical in patient management. Each one has an entirely different treatment approach, and misdiagnosis can lead to catastrophic outcomes.
Introduction
Aortic dissection is a life-threatening medical emergency. Usually, patients with aortic dissection experience sharp and severe pain in the chest, back, or abdomen. Atypical painless aortic dissection has been reported but is commonly accompanied by other symptoms such as syncope. Atypical symptoms and features are seen in about 5% of acute aortic dissections, which are associated with poor outcomes. 1 Misdiagnosis between acute coronary syndrome (ACS) and aortic dissection can lead to catastrophic outcomes. Usually, presentation and physical examination can differentiate these two situations, but sometimes aortic dissection may present with unusual symptoms and mimic ACS. This misdiagnosis can almost lead to patient’s death as a result of differences in treatment approach, using fibrinolytic and primary percutaneous intervention (PCI) in ACS. Herein, we report a case with a presentation compatible with unstable angina that, fortunately, before any revascularization therapy, ascending aortic dissection was diagnosed through transthoracic echocardiography (TTE).
Case Presentation
A 65-year-old man with a medical history of stage 1 hypertension (based on 2018 European Society of Cardiology and the European Society of Hypertension guidelines) 2 who had not been using any medication presented to our emergency department with new-onset chest heaviness which started an hour ago. Nitrates (sublingual trinitroglycerin) consumption after initial chest pain resolved pain at home, but after 15 minutes, the pain started again, causing patient’s referral to the hospital. The patient did not have nausea and vomiting, but his pain was radiating to his left shoulder and he had cold sweating. Upon arrival in the emergency department, the patient’s arterial oxygen saturation was 96%, his pulse rate was 68, and he did not have any dyspnea. Blood pressure of right arm was 129/55 mm Hg and left arm was 125/50 mm Hg, and distal pulses were normal. Heart auscultation was significant for a 3/6 midsystolic murmur at the apex and a 3/6 diastolic murmur at the right sternal border. His medical record did not note any ischemic heart disease or diabetes mellitus. He was an 8 pack-year ex-smoker who had quitted 10 years ago. His pain was heaviness-like without any tearing or sharp feeling, and when he got to the hospital, his pain was resolved with nitrate use in the ambulance, and the pain duration was about 20 minutes. The patient was initially diagnosed with ACS. The initial electrocardiogram (ECG) showed biphasic T waves in V3-V6 leads (Figure 1). Early treatment with anticoagulants and antiplatelet agents was started. He was admitted to the coronary care unit (CCU) to perform cardiac monitoring, serial ECG, and laboratory tests. The patient’s cardiac troponin levels were negative for the first two times. Three days later, TTE revealed left ventricular ejection fraction (EF) of 50% to 55%, mild to moderate aortic insufficiency (AI), and mild left ventricle hypertrophy (LVH), and no dissection flap or pericardial effusion was seen. After that, he felt heaviness which resolved spontaneously after 15 minutes, and he did not tell CCU staff. On the fourth day of admission, the patient progressed to dyspnea, which we performed another TTE that demonstrated intimal flap in the ascending aorta which was extended to the proximal part of ascending aorta, mild to moderate pericardial effusion on the anterior part of the right ventricle with early diastolic right ventricle and right atrial collapse. The patient was scheduled for computed tomography angiography (CTA), which demonstrated ascending aorta dissection flap (type A), which started from the aortic valve and expanded to the aortic arch with intact coronary arteries accompanied by pericardial effusion, demonstrating opening of dissection to the pericardium space (Figure 2). The patient did not develop any sharp or compression chest pain. The patient was transferred to the operating room, and his ascending aorta was removed and replaced with 30 mm graft. In operating room, he also had a pericardial hematoma. Full-thickness clot formation was distinguished in a gross examination of the aortic wall, indicating old dissection. His aortic root and aortic valve were intact.

Initial electrocardiogram showed biphasic T waves in V3-V6 leads.
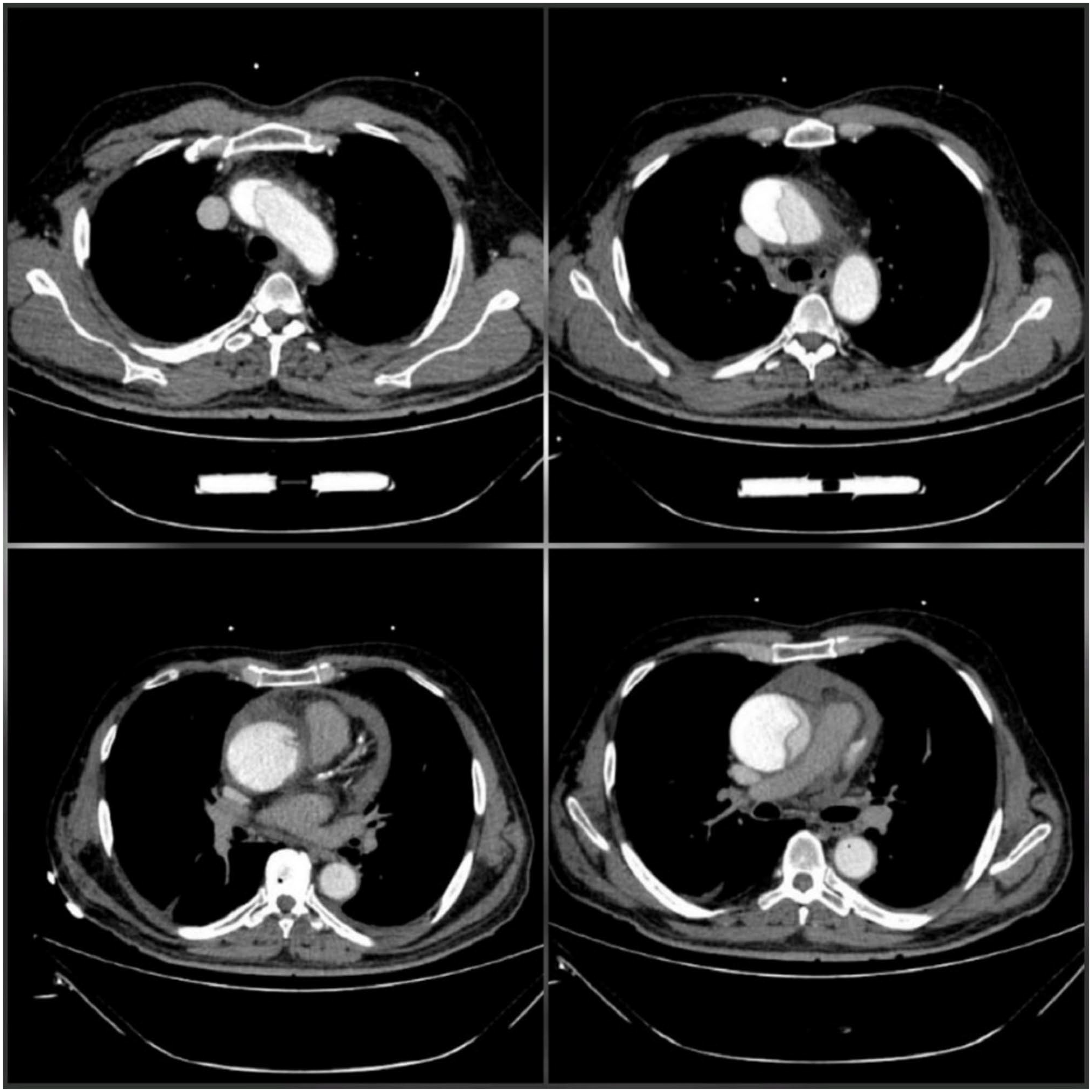
Pre-operative computed tomography angiography showed type A aorta dissection, starting from the aortic valve and expanding to the aortic arch with intact coronary arteries and pericardial effusion.
After a week of admission in the cardiac surgery intensive care unit (ICU), the patient was discharged. Two weeks later, follow-up TTE demonstrated pericardial effusion, and subsequent CTA revealed small contrast leakage, 1 centimeter above sinotubular junction, accompanied with moderate to severe pericardial effusion (Figure 3). The patient was transferred to the operation room and underwent a subxiphoid pericardial window. Four days later, the patient was discharged without any complication.
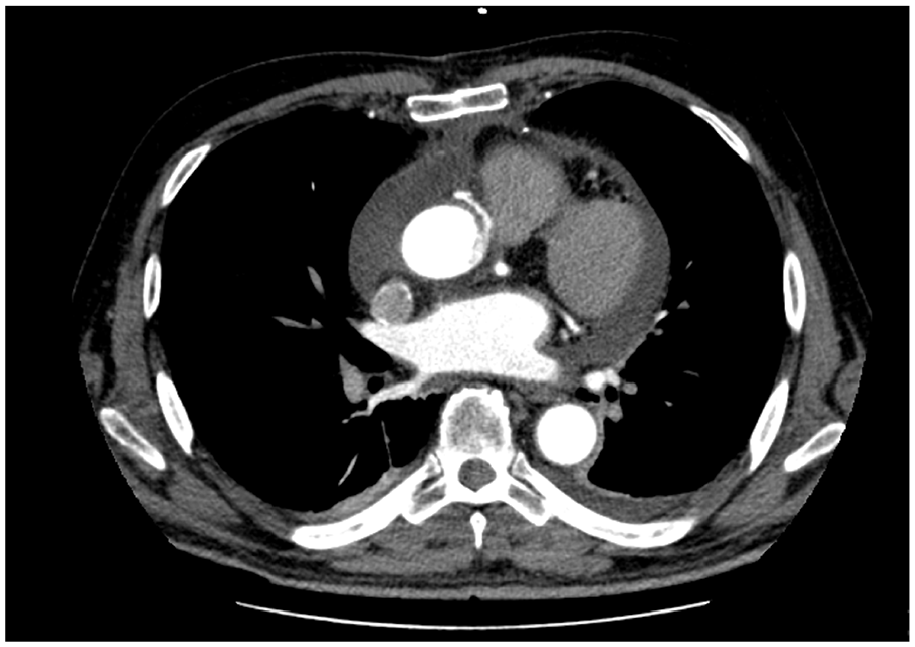
Post-operative computed tomography angiography showing contrast leakage and pericardial effusion.
Discussion
Aortic dissection classically presents with abrupt onset, severe, sharp, and tearing pain in the chest, back, or abdomen. 3 Severe chest pain is the most common presentation in type A and type B aortic dissection. However, painless ascending aorta dissection is more frequent among patients with type A aortic dissection than type B and is accompanied by increased mortality. 4 Painless aortic dissection is infrequent, with a reported incidence of 5% to 15%. 5 Although some patients did not have any chest pain on presentation, other symptoms included syncope, acute heart failure, and neurologic manifestations. 6 Diagnosis of ascending aorta dissection requires a high clinical suspicion. Physicians should always consider it in patients with the presentations mentioned above, where an exact diagnosis is uncertain. 7
Our patient was a rare situation that presented to the hospital with symptoms compatible with unstable angina. He had none of the signs compatible with aortic dissection. The patient only presented with pressure-like chest pain with an on-and-off pattern that resolved with nitrates (sublingual TNG), increasing the difficulty of aortic dissection diagnosis.
Specific comorbidities like long-standing hypertension, soft tissue diseases, bicuspid aortic valve, coarctation of the aorta, vascular inflammation, amphetamine and cocaine use, trauma, and presence of aortic disease in the family are typical risk factors for aortic dissection. 8
Although ST–T changes like elevation or depression of ST-segment or negative T waves have been commonly observed in symptomatic acute ascending aorta dissection (type A), 30% of ECGs can be without any abnormalities; ECG changes are commonly seen in ACS and are not helpful in differentiation between aortic dissection and ACS. 9
TTE has a sensitivity of 59% to 83% and a specificity of 63% to 93%, but it is operator-dependent. 10 CTA is the gold standard test to reveal aortic dissection; however, the use of this modality can be delayed in the setting of atypical presentation, when aortic dissection is not considered. 11
In the first TTE of our patient, no sign of aortic dissection was seen, but the second TTE in a day later demonstrated dissection flap. The probable reason is that dissection was limited during the patient’s initial presentation and the flap could be in a part of the aorta that was not detected with TTE. The pain recurrence in the evening of the third day which was spontaneously improved might be associated with the expansion of the flap toward the ascending aorta, leading to flap extension to the pericardium, pericardial effusion, and subsequent shortness of breath. Then, the patient underwent TTE to investigate the cause of dyspnea.
Surgical treatment is the gold standard for Stanford type A aortic dissection, and the surgical procedure methods depend on the expansion of dissection. 12 Our patient underwent surgical removal of ascending aorta and graft replacement.
Conclusion
Aortic dissection is an uncommon but life-threatening aortic emergency. Aortic dissection can present with atypical demonstration and leads to misdiagnosis which can have catastrophic outcomes. In our case, despite accurate physical examination and history taking, the initial presentation was compatible with ACS. Differentiating myocardial infarction (MI) from aortic dissection is critical and a serious dilemma for physicians because initial therapy of MI may lead to extension of dissection and impair life-saving clot formation and eventually progress to death.
Footnotes
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
