Abstract
Pneumomediastinum is a rare complication among non-coronavirus patients but has been published with increased incidence in patients positive for SARS-CoV-2 infection. Most of these studies report patients on mechanical ventilation and an understanding of mechanisms causing this remains limited. We aim to use an increasing occurrence in patients not on mechanical ventilation to further explore mechanisms that predispose patients to pneumomediastinum and to assess characteristics potentially related to poor outcomes. We report a case series of 37 patients diagnosed with COVID-19 and pneumomediastinum at a 2-hospital institution between January 1, 2020 and April 30, 2021. At 28 days after diagnosis of pneumomediastinum, 19 (51.4%) were dead and mortality was significantly higher among those who were older (t = 2.147, P = .039), female (χ2 = 10.431, P = .015), body mass index ≥30 (χ2 = 6.0598, P = .01), intubated (χ2 = 4.937, P = .026), and had pre-existing lung disease (χ2 = 4.081, P = .043). Twenty-three patients (62.2%) were identified to have pneumomediastinum without receiving invasive mechanical ventilation, of which 11 (47.8%) were diagnosed without receiving noninvasive ventilation. The increased diagnosis of pneumomediastinum in patients with COVID-19 while not on mechanical ventilation, in this case series and in comparable studies, may attribute to mechanisms aside from positive pressure ventilation such as patient self-induced lung injury and pulmonary frailty.
Keywords
Introduction
Pneumomediastinum, or mediastinal emphysema, is described as air within the mediastinum. 1 While relatively small, the exact incidence of pneumomediastinum is unclear and likely underestimated as symptoms are nonspecific and may go unnoticed leading to underdiagnosis. 2 It can be classified as traumatic, either due to physical trauma or pressure-generated trauma from mechanical ventilation, or spontaneous. 1 The mechanism for spontaneous pneumomediastinum has been described by an increase in the pressure gradient between intra-alveolar and lung interstitial pressures causing alveolar rupture and subsequent dissection of air along bronchovascular sheaths into the mediastinum. 3
While a relatively rare complication in non-coronavirus patients, the incidence of pneumomediastinum has been reported to be 6 times higher in patients positive for SARS-CoV-2; 90% of these cases were in patients on mechanical ventilation. 4 Another retrospective analysis comparing mechanically ventilated patients with coronavirus disease 2019 (COVID-19) ARDS (acute respiratory distress syndrome) with those with non-COVID-19 ARDS also found the COVID-19 group to have a significantly higher incidence of pneumomediastinum (13.6% versus 1.9%). 5 In non-coronavirus patients, the occurrence of pneumomediastinum has been theorized to be due to ventilator-induced lung injury (VILI) in the form of barotrauma or volutrauma. 6 Successful reduction in the incidence of barotrauma in non-coronavirus ARDS patients has been credited to following lung-protective strategies per ARDSnet protocol, focused on avoiding high tidal volume ventilation and plateau pressures.7,8 However, this mechanism of VILI causing pneumomediastinum has not been largely supported as a primary factor in patients with COVID-19. Lemmers et al 6 showed that while patients with COVID-19 on a mechanical ventilator who developed a pneumothorax (PTX) or pneumomediastinum had significantly higher maximum minute ventilation compared with those without barotrauma, there were no significant differences in tidal volumes per ideal body weight or plateau pressures. Furthermore, in a larger analysis of 53 hospitals in the United Kingdom, 205 patients with COVID-19 were identified who were not mechanically ventilated when pneumomediastinum was identified. 9 This suggests that while VILI may provide an explanation for pneumomediastinum in intubated non-coronavirus patients, it does not explain the occurrence in coronavirus patients, especially in nonintubated patients.
There may be mechanisms inherent to the coronavirus as evidence from prior coronavirus pandemics demonstrates a relatively higher than expected incidence of pneumomediastinum. Twelve percent of mechanically ventilated patients during the SARS coronavirus outbreak of 2003 developed pneumomediastinum and 30% developed barotrauma during the MERS (Middle East respiratory syndrome) coronavirus pandemic.10,11 Case series also exist describing pneumomediastinum in nonventilated patients for both the SARS and COVID-19 pandemics.2,12-18 Within these papers, one such explanation raised for the development of pneumomediastinum is patient self-induced lung injury (P-SILI).16-18 Literature describing the vicious cycle of P-SILI explains that patients with pulmonary damage have high respiratory drive and that this increase in inspiratory effort can further damage lungs from overdistention, repeated opening and closing of alveoli resulting in shear stress, and increased transpulmonary pressures from increased muscular effort. 18 This is further compounded by secondary vascular permeability resulting in dependent edema further reducing effective lung volumes and further increasing transpulmonary pressures.19,20 This leads to a vicious cycle where damaged lungs promote increased respiratory drive leading to further damage.
In this study, we will examine the characteristics of those who developed pneumomediastinum and assess which risk factors are associated with higher mortality. There will be a particular focus on the nonintubated cohort, with attention to the duration of symptoms and respiratory support they were provided prior to identification of pneumomediastinum, with a goal to further understand why this population is prone to the development of pneumomediastinum.
Methods
A retrospective review was conducted at a 2-hospital institution in southern New Jersey. Data were manually extracted using the hospital’s electronic medical record system. The study was reviewed and approved by the Inspira Health Institutional Review Board (File # 2021-03-004). The requirement for informed consent was waived due to adequate provisions to protect the privacy of subjects and to maintain confidentiality of data.
Inclusion criteria were defined as patients who were 18 years or older, admitted between January 2020 and April 2021, tested positive for SARS-CoV-2 infection by nasal polymerase chain reaction on or during admission, and had documented radiographic evidence of pneumomediastinum.
Descriptive statistics was used to characterize the cohort who developed pneumomediastinum prior to mechanical ventilation, with special focus to the respiratory support provided to diagnosis. Differences in mortality at 28 days after diagnosis among all patients in this series were also compared using χ2 analysis for nominal variables or an independent 2-tailed t test for continuous variables. Significance was determined when a P value of less than .05 was achieved. Descriptive statistics, specifically measures of central tendency, were used to describe the characteristics of subjects who developed pneumomediastinum prior to or without ever requiring mechanical ventilation. Analysis was performed using Statistical Package for Social Sciences (SPSS), Version 26.
Results
Thirty-seven patients were identified to have radiographic evidence of pneumomediastinum and tested positive for SARS-CoV-2 infection on the same admission. Characteristics of subjects included mean age 62.03 ± 13.58 years, female (16, 43.2%), body mass index (BMI) of 30 or higher (20, 54%), hypertension (HTN) (21, 56.8%), diabetes (12, 32.4%), and pre-existing lung disease (7, 18.9%). Nineteen (51.4%) were dead at 28 days after diagnosis of pneumomediastinum, 25 (67.6%) were intubated during their hospital course, and 12 (32.4%) also had a PTX during the admission. Relevant clinical characteristics and outcomes were compared between those alive and dead (Table 1). Mortality was significantly higher among those who were older (t = 2.147, P = .039), females (χ2 = 10.431, P = .015), BMI ≥30 (χ2 = 6.0598, P = .01), intubated (χ2 = 4.937, P = .026), and had pre-existing lung disease (χ2 = 4.081, P = .043). It was not significantly different based on the presence of HTN (χ2 = 0.652, P = .419) or diabetes (χ2 = 1.668, P = .197).
Comparison of Relevant Characteristics and Clinical Course of Patients Alive to Dead at 28 Days After Diagnosis of Pneumomediastinum.
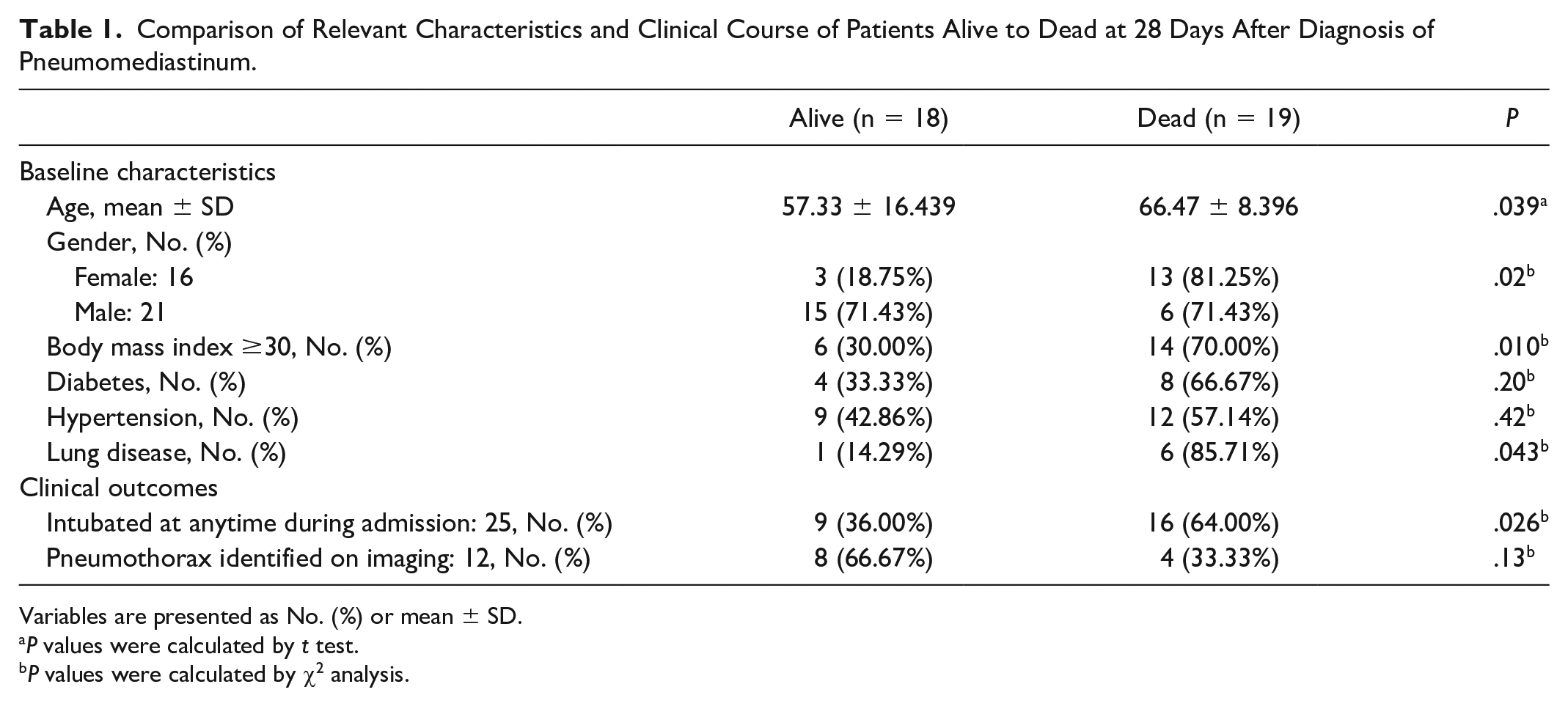
Variables are presented as No. (%) or mean ± SD.
P values were calculated by t test.
P values were calculated by χ2 analysis.
Twenty-three of the 37 subjects (62.2%) were diagnosed with pneumomediastinum prior to or without ever requiring any invasive mechanical ventilation (IMV). Of these 23, 11 (47.8%) progressed to requiring mechanical ventilation. The number of days from symptom onset, initial COVID-19 laboratory diagnosis and admission to the hospital to diagnosis of pneumomediastinum via chest x-ray or computed tomography scan were recorded (Table 2). Subjects were found to have pneumomediastinum on imaging after a mean 19.17 days from symptom onset, mean 14.09 days from initial COVID diagnosis, and mean 12.35 days from day of relevant hospital admission.
Time Course of Presentation Prior to Identification of Pneumomediastinum in Subjects Diagnosed Without Being on Mechanical Ventilation (N = 23).

Noninvasive oxygen requirements in the form of high-flow nasal cannula (HFNC) or noninvasive ventilation (NIV) administered prior to diagnosis of pneumomediastinum in these 23 subjects were also recorded (Table 3). Eleven of 23 (47.8%) were diagnosed with pneumomediastinum without receiving any NIV, 6 (26.1%) without being placed on HFNC, and 4 (17.4%) without being placed on either HFNC or NIV. The 17 subjects who received HFNC prior to diagnosis of pneumomediastinum were on it for a mean 10.65 days and 16 of these 17 were on a flow rate of at least 40 LPM. The 12 subjects who received NIV prior to diagnosis of pneumomediastinum were on it for a mean 4.92 days prior and 10 of these 12 received an inspiratory pressure of 10 cm H2O or higher. Of these 12 subjects who received NIV prior to diagnosis of pneumomediastinum, 8 (66.7%) ended up being intubated and 7 (58.3%) were dead at 28 days after pneumomediastinum diagnosis. On the contrary, of the other 11 who did not receive NIV prior to diagnosis of pneumomediastinum, 4 (36.3%) ended up intubated and 3 (27.3%) were dead at 28 days after diagnosis.
HFNC and NIV Oxygen Requirements Prior to Identification of Pneumomediastinum in Subjects Diagnosed Without Being on Mechanical Ventilation.

Abbreviations: HFNC, high-flow nasal cannula; NIV, noninvasive ventilation; LPM, liters per minute.
Discussion
The literature shows increasing reports of pneumomediastinum in patients with COVID-19, of which the majority present patients who are mechanically ventilated.4,6 While some studies have attributed this to be due to barotrauma, from either significantly higher set positive end-expiratory pressure (PEEP) or higher minute ventilation, this does not provide an explanation for the increased disposition to pneumomediastinum in those not on mechanical ventilation. 10 This case series and the study by Melhorn et al 9 present a cohort of patients with pneumomediastinum in the setting of COVID-19 in whom most patients were not mechanically ventilated; 62.2% of our cohort and 54.4% of the cohort reported by Melhorn et al were not mechanically ventilated and yet developed pneumomediastinum. This phenomenon has also been reported in case reports and small case series.12-18 Given the sample size in this case series and in comparable studies, the development of pneumomediastinum in patients not mechanically ventilated is an unignorable complication of COVID-19 worth further investigation. This includes assessing characteristics that can predispose patients to poor outcomes and exploring the pathophysiology of COVID-19 that poses an increased risk for the development of pneumomediastinum.
It could be argued that NIV (bilevel or continuous positive airway pressure) provides the increase in PEEP that leads to traumatic pneumomediastinum, similar to that seen in patients on mechanical ventilation who received higher PEEP prior to diagnosis of pnuemomediastinum. 4 However, 11 (47.8%) of the subjects in this nonintubated cohort and 77 in the larger study by Melhorn et al 9 did not receive positive pressure from NIV prior to diagnosis. In addition, the mean positive pressure provided in these subjects who received NIV was minimal with an average of 10.8 cm H2O and 7 of these subjects never received a positive pressure higher than 10 cm H2O. The HFNC has been shown to provide some PEEP; it is approximated that with a closed mouth that every 10 L/min of oxygen flow on HFNC equates to between 0.6 and 1.0 H2O of PEEP. 21 Thus, the likely maximum generated PEEP among our patients on HFNC with a median flow rate of 40 L/min would only be 4 cm H2O. This is probably an overestimation given that most patients in respiratory distress are likely to breathe with their mouths open. In comparison with cohorts of mechanically ventilated patients with COVID-19 who developed pneumomediastinum, the median max PEEP ranged from 12 to 15.3 cm H2O.6,10 These data may support mechanisms outside of the positive pressure provided as responsible for COVID-19-related pneumomediastinum.
If positive pressure from the respiratory support provided is not responsible for the development of pneumomediastinum, then P-SILI from high minute ventilation or volutrauma from excessive tidal volumes may be the culprit. A tidal volume of 6 to 8 cc/kg is targeted in mechanically ventilated patients to minimize lung injury. 22 In a spontaneously breathing patient, it would be difficult to suppress the respiratory drive to achieve similar tidal volumes. This could allow for excessive tidal volumes and uncontrolled minute ventilation resulting in notable swings in transpulmonary pressures, which in turn could lead to a PTX or pneumomediastinum.15,23 This mechanism of how impaired respiratory drive from injured lungs contributes to a cyclical worsening of lung injury has been described as a central explanation of how P-SILI leads to barotrauma.24,25 Spontaneously breathing patients with COVID-19 monitored on NIV have been observed to take larger tidal volumes as the severity of hypoxia progresses. This increase in minute ventilation has been linked to the development of pneumomediastinum.15,26 Just as the large tidal volumes or increases in transpulmonary pressures in mechanically ventilated patients contributes to VILI, it is then reasonable to consider a similar mechanism in the nonventilated COVID-19 patient. Other authors have offered a similar explanation for the development of pneumomediastinum in the nonventilated patient.15-17 While the retrospective nature of our data and the wide minute-to-minute variations of minute ventilation witnessed at the bedside prohibited accurate recording of our subjects’ tidal volumes or minute ventilation, the number of subjects in our cohort, and in comparable studies, who developed pneumomediastinum while on NIV or HFNC are supportive of P-SILI playing a precipitating role. 9
If COVID-19 causes these exacerbations in breathing patterns that are associated with the development of pneumomediastinum, then the length of symptoms should correlate: the longer the patient is symptomatic, the more likely they will develop pneumomediastinum. Subjects in our study were symptomatic for an average of 19.17 days and hospitalized for an average of 12.35 days prior to identification of pneumomediastinum. Also, there was a shorter average use time of NIV (4.92 days) compared with HFNC (10.65 days) prior to identification of pneumomediastinum. While difficult to prove, the theory would be that a higher level of support correlates with worse respiratory status and more likely development of P-SILI and, subsequently, pneumomediastinum. As expected, patients who required NIV were more likely to get intubated and had a higher 28-day mortality than those who did not. While these data do not substantiate NIV being a greater risk of pneumomediastinum compared with HFNC, it is a relationship worth exploring further in a targeted study.
Additional considerations based on the literature that could provide further insight into why patients with COVID-19 are at increased risk of pneumomediastinum include evidence of alveolar damage as evidenced by radiographic imaging and postmortem tissue analysis. Radiograph studies generally show a propensity for dependent edema, diffuse cystic changes, and thrombosis.23,27 In a case series of autopsies performed on patients that died from COVID-19, the primary pathology found in all autopsies was small vessel thrombosis and diffuse alveolar damage, including among those who had not been mechanically ventilated. 28 These pathological processes could increase the frailty of lung parenchyma and make alveoli prone to rupture with the slightest additional insult such as overdistention or stress from high airway pressures. A future study to further support the above proposed mechanism is to review serial computed tomographic scans of subjects as their symptoms and required respiratory support progressed.
An expected limitation of any retrospective cohort, such as exploring mortality risk of pneumomediastinum in COVID-19, is that it would present data difficult to validate and cannot control for extraneous variables. 29 Given that the mortality rate of patients with COVID-19 requiring oxygen can range anywhere from 25% to 55% for nonintubated inpatients and 43% to 75% for intubated inpatients, it would be unreasonable to correlate any increased mortality risk specifically attributed to the development of pneumomediastinum with the current study design. 30 Other studies exploring the mortality difference between COVID-19 ARDS patients who developed pneumomediastinum and between those who did not showed no significant difference.4,6,10
The mortality analysis at 28 days after pneumomediastinum performed in this study simply provides observed characteristics and does not aim to define risk factors for mortality associated with the presence of pneumomediastinum in COVID-19. Our study shows a significantly higher mortality risk among COVID-19 subjects with pneumomediastinum who are older, obese, or diagnosed with pre-existing lung disease. It would be difficult to attribute these risk factors as the reason for poor outcomes in the setting of pneumomediastinum as these are similar to the risk factors that predisposed patients with COVID-19 to poor outcomes in studies analyzing mortality without regard to the presence of pneumomediastinum. 31 While our data show a higher mortality risk among female patients with COVID-19 with pneumomediastinum compared with male patients, this may be due to chance as a recent study with a larger sample size examining mortality data without regard to pneumomediastinum shows higher mortality among male patients. 31 In a larger cohort of patients with COVID-19 identified to have pneumomediastinum, mortality at 120 days was significantly higher among patients who were male, older, had HTN, diabetes, or were mechanically ventilated patients. 9 It was not significant in patients with pre-existing lung disease or obesity with BMI >35. Given the limited sample sizes and conflicting significance in these cohorts compared with each other and with those without pneumomediastinum, current evidence is inconclusive as to which risk factors increase mortality in the setting of pneumomediastinum. The exception may be older age and mechanical ventilation as this was consistently associated with significantly higher mortality.4,9,31 It may be beneficial to further explore through targeted studies how and to what degree these risk factors in the presence of pneumomediastinum are associated with worse outcomes. Through more conclusive evidence we may increase our ability to predict when the presence of pneumomediastinum correlates with a worse prognosis, such as through scoring systems, and potentially affect clinical decision-making in advancing or withdrawing care.
Conclusions
Pneumomediastinum has become an increasingly recognized complication of COVID-19 and risk factor that could predispose these patients to higher mortality may include IMV, obesity, older age, and female sex. The percentage of subjects in this cohort, and in comparable studies, who were diagnosed with pneumomediastinum without being intubated support precipitating mechanisms aside from positive pressure ventilation. While further investigation is required to explore COVID-19’s role in precipitating pneumomediastinum, analysis of duration of symptoms, and the respiratory support provided may hint at P-SILI or pulmonary frailty precipitated by coronavirus as responsible processes.
Footnotes
Acknowledgements
The authors would like to acknowledge Dr Samantha Abate, Director of Clinical Research at Inspira Health Network, for her assistance in statistical analysis.
Authors’ Note
Prior presentation of abstract statement: Preliminary data of this case series were published as an abstract in the 51st Critical Care Congress, Critical Care Medicine, January 2022.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval to report this case series was obtained from the Inspira Health Institutional Review Board (FWA # 00003816), which is responsible for the ethical oversight of research activities.
Informed Consent
Informed consent for patient information to be published in this article was not obtained because the Inspira Health Institutional Review Board waived the needs for signed informed consent as data were collected retrospectively from closed medical records and adequate provisions were taken to protect the privacy of and confidentiality of the data.
