Abstract
Breast plasmacytoma is relatively uncommon in which most of the recorded cases were related to disseminated multiple myeloma. However, many of these cases tend to be misdiagnosed with other breast lesions such as breast carcinoma. This article presents a case study on a Libyan female patient around the age of 55 who has a single breast lump, which was first diagnosed to be a malignant lesion. All the results of immunostaining for cytokeratins, GATA3, estrogen receptor, progesterone receptor, HER2, and E-cadherin were negative; hence, the possibility of a breast carcinoma was not considered. However, plasma cell tumors were indicated by the presence of CD138, MUM1, and kappa-light chain markers. In addition, the patient had multiple osteolytic bone lesions, plasma cell infiltration, a monoclonal gammopathy, and signs of renal failure, which considered to be an indication to an extra-medullary breast plasmacytoma secondary to advanced multiple myeloma. This case study emphasizes the necessity of complete histopathological and imaging evolution for proper diagnosis of breast plasmacytoma.
Introduction
Solitary plasmacytoma (SP) is a rare cancer that has affected approximately 0.191/100.000 men and 0.090/100.000 women worldwide. 1 It is characterized by the lack or minimal presence of diffuse bone marrow involvement. 1 Solitary bone plasmacytoma is defined by the presence of a single lytic bone lesion with or without soft-tissue extension, whereas extra-medullary plasmacytoma (EMP) is defined by the presence of a soft-tissue mass without bone involvement. 2 Multiple myeloma (MM), however, is described as a lesion of the bone marrow that is associated with a variety of clinical, radiographic, and laboratory characteristics. MM can also affect several extra-osseous locations and manifest as a single EMP or a relapse of MM. 3 EMPs produce only 4% of all plasma cell tumors and are usually seen in the respiratory tract, oral cavity, and soft tissues. 3
Breast plasmacytoma (BP) is a rare form of plasma cell tumor as most of the reported cases occurred as a result of the disseminated MM. According to the literature, BP accounted for roughly 1.5% of all the occurred plasmacytoma; of which, 15% of these instances were classified as primary BP, whereas the rest of the instances were secondary MM events. 4 Clinically, cases of BP are mostly presented with unilateral or bilateral, solitary or multiple breast lumps; which are similar to various lumpy lesions of the breast. 4 As a result, it can be confused with other breast cancers easily, especially when their clinical and imaging features are similar. Consequently, patients may be subjected to unnecessary surgeries such as mastectomy. Furthermore, indicating whether the BP lesion is a primary or a part of advanced MM can influence the outcomes and the treatment management. Therefore, employing advanced imaging and histopathology techniques, and a precise diagnosis of BP is absolutely essential.
In this article, a case of BP related to disseminated MM is presented, which emphasizes the importance of documenting the histological and imaging evolution of any breast lump accurately to distinguish between unusual lesions (i.e., BP) and other breast lesions.
Case Report
A Libyan patient woman around the age of 55 was examined at National Cancer Institute–Sabratha in August 2021. She had a painless unilateral breast mass on her left side. Over the course of 3 months, the size of the mass grew without any nipple secretion or skin retraction. One year ago, the patient complained of general weakness and weight loss. Based on the examination, the patient was cachectic, underweight (45 kg), and had a single breast mass in the left breast at 1 o’clock. The mass was mobile and firm, measured 1 × 2 cm in diameter without changes to the surrounded skin nor palpable corresponding lymph nodes. Mammographic evaluation of the breasts and axilla suggested a high suspicion of cancer with a BI-RADS score of V. Under ultrasound (US) guidance, a core needle biopsy was taken for histopathological evaluation.
The obtained sections demonstrated extensive tumor necrosis, with a few minor areas of malignant epithelial cell infiltration (see Figure 1). The size of tumor cells was between small and medium, with sparse pale to clear cytoplasm, vesicular nuclei with indistinct nucleoli, and minor pleomorphism. The cells lack cohesion and spread out randomly through the fibrous stroma, which resulted in creating loose cellular grouping with a few single-file linear cords without tubular formation. Mitosis was sparse, with only 8 out of 10 of mitosis that has been seen in a high-powered field. The tumor was graded with a score of 5, with no evidence of ductal carcinoma in situ (DCIS), lobular carcinoma in situ (LCIS), lymph-vascular, or perineural invasion. As a result, a primary diagnosis of invasive mammary carcinoma with lobular feature grade I was applied.

H&E stain of the tumor section revealed malignant cells with scanty cytoplasm, vesicular nuclei with indistinct nucleoli, and mild pleomorphism.
Further immunohistochemistry staining of tumor sections excluded the possibility of invasive breast carcinoma as a diagnosis and instead suggested the plasma cell tumors. The obtained results of breast carcinoma-related markers such as cytokeratins (Ck and CK7), the transcription factor GATA3, estrogen receptor (ER), progesterone receptor (PR), HER2, and E-cadherin were negative as shown in Figure 2. Whereas the staining was positive for each of CD138, MUM1, EMA (focally), P53, and kappa-light chain as showed in Figure 3, which resulted in proposing plasma cell neoplasia. Other markers such as CD68, LCA, CD79a, and lambda-light chain were also shown to be negative (see Figure 3). In 5% of tumor cells, the proliferation index Ki67 was positive. In our instance, the final diagnosis for advanced multiple myeloma was extra-medullary breast plasmacytoma (a kappa-restricted form).

(A-F) Immunohistochemistry staining of breast sections for breast-carcinoma related markers: the staining was negative for cytokeratins (Ck and CK7) (A&B), estrogen receptor (C), progesterone receptor (D), the transcription factor GATA3 (E), and HER2 (F).

(A-E) Immunohistochemistry staining of breast sections for plasma cells neoplasia-related markers. The staining was negative for monocyte-lineage related marker CD68 (A), but positive for plasma cell marker CD138 (B), multiple myeloma oncogene (MUM1) (C), and kappa-light chain. Whereas, lambda-light chain was negative (D).
Further examination of the breast for tumor staging via using post-contrast CT scanning and magnetic resonance imaging (MRI) revealed a calcified lesion impeded in the breast parenchyma with no involvement of the surrounded skin, underlying muscle, or ipsilateral lymph nodes. In addition, diffuse osteolytic bone lesions were discovered in the ribs and pelvic bone, primarily at the left iliac bone as illustrated in Figure 4.
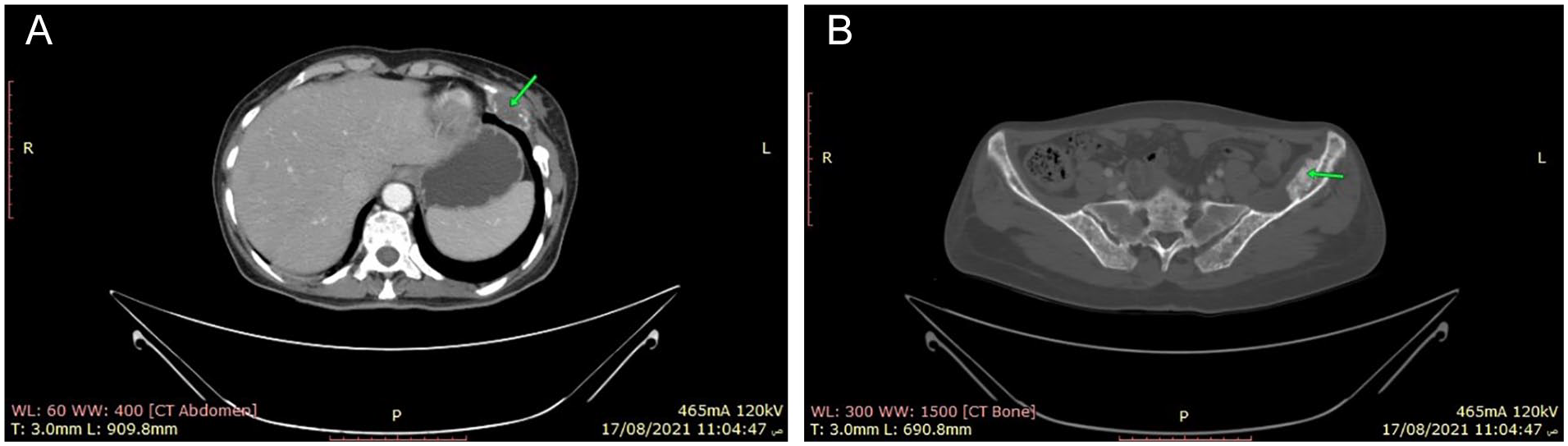
(A-B) Evidence of multiple osteolytic bone lesions mainly evident at ribs and the sternum (the green arrows). The largest lesion at the left lower ribs around 2.5 × 4.8 cm with soft tissue component (A). diffuse osteolytic appearance of whole skeletal bones, prominently seen at the left iliac bone (B).
Moreover, MRI results showed that there was multiple increased destructive osteolytic vertebral body lesions at the lumbar vertebrae as well as there was a parenchymal focal lesion in the left lobe of the liver, which was an indication of liver metastasis (see Figures 5 and 6). The hepatic lesion was a tiny hypoechoic ill-defined focal lesion, measuring about 1.2 × 0.7 cm on US examination. Pancytopenia with about 29% of plasma cell infiltration and a monoclonal gammopathy were also discovered in the patient’s bone marrow and protein electrophoresis, respectively. These findings suggested that the multiple myeloma was a secretory type with extra-medullary metastases.
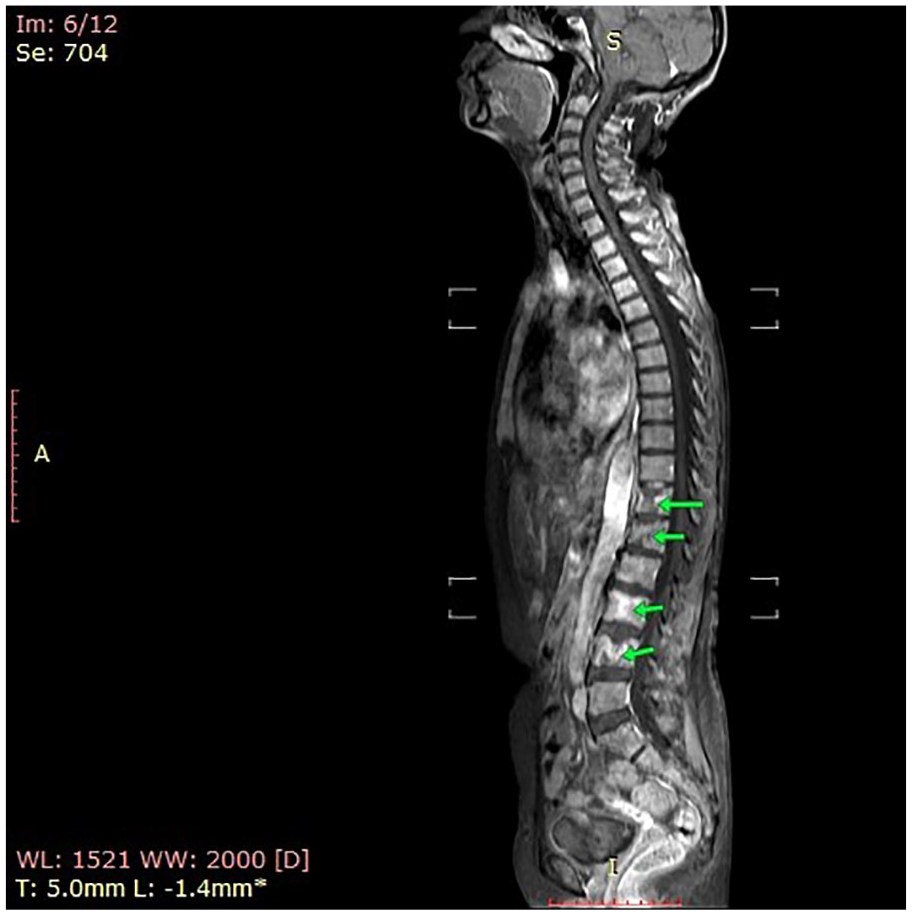
T1 fat-saturated post-contrast MRI image revealed multiple enhanced vertebral bodies at the upper thoracic region, with multiple enhanced destructive osteolytic vertebral body lesions at the lumbar vertebrae.

CT scan post-contrast image showed left hepatic lobe with small parenchymal focal lesion, along with peripheral enhancement measuring 1.5 × 1 cm.
Due to severe renal failure, the patent had renal dialysis at the time of her presentation, which demonstrated systemic involvement. The patient’s most recent biomedical tests revealed that there was hypocalcaemia and a slight increase in creatinine levels. Other tests, such as blood sugar, urea, electrolytes (eg, phosphorus, uric acid, CL-2, Na+, and K+), and the whole liver function tests (LFT) were within normal limits. The patient is currently undertaking treatment for multiple myeloma and a regular monitoring plan is considered for her.
Discussion
According to the International Myeloma Working Group (IMWG), active MM is defined as a bone plasma cell proliferation of more than 10% or biopsy-approved plasmacytoma (ie, bony or extra-medullary) with evidence of 1 or more of MM-Defining Events (MDE). 5 Such events can be indicated by (1) end-organ damage (eg, bone osteolytic lesions, renal failure, hypercalcemia, or anemia), (2) more than 60% of clonal bone marrow plasma cell, (3) involved/uninvolved serum free light chain (FLC) with a ratio of more than 100, or (4) the appearance of more than 1 focal lesion with a diameter of at least 5 mm on MRI investigations.5,6
In accordance with the above description, our index patient showed about 29% of bone marrow plasma cell proliferation, pancytopenia, renal failure, and monoclonal gammopathy. In addition, there were multiple osteolytic lesions on the lumbar vertebrae, ribs, and sternum, with liver metastases. This case verifies the diagnosis of advanced MM with extra medullary lesions.
MM is one of the plasma cell cancer types that is counted around 1% of all cancers and about 10% of all hematologic cancers.5,7 Extra-medullary disease (EMD) is a type of MM that occurs outside of the bone marrow. 3 In certain studies, the overall incidence of EMD reached 13%, which was divided into 2 categories: primary EMD (7%), and relapse/secondary EMD (6%-20%).3,8 EMD has different subtypes which are (1) solitary plasmacytoma (SP) with no marrow involvement, (2) SP with limited marrow involvement, (3) bone-associated EMD with MM (EMM), (4) organ infiltrating EMD, and (5) plasma cell leukemia (PCL). 3 Within this context, the presented case in this article is an example of extra-medullary breast plasmacytoma secondary to disseminated MM.
Because the clinical and radiological features of BP are nonspecific, it can be difficult to distinguish it from other breast cancers. BP usually manifests as a single or numerous breast lumps, well-defined or ill-defined lesions, with or without microcalcification. 8 Traditionally, BP is defined as hyperechoic or hypoechoic solid mass lesions on US examination. MRI studies typically reveal hypointense lesions on T1WIs and hyper-intense lesions on T2WIs.8,9 The radiological evaluation of our case revealed similar findings, though it was inconclusive as to the rarity of BP and the radiological features resemblance to other lymphoproliferative diseases or even benign breast lesions.
Our case was confirmed by histopathological investigation and immunohistochemical (IHC) staining. CD138, multiple myeloma oncogene 1 (MUM1) marker, and kappa-light chain restriction were substantially positive in the examined sections. Whereas, CD68 (a selective marker for human monocytes and macrophages), CD79a, and leukocyte common antigen (LCA) were negative. These findings supported our diagnosis of extra-medullary breast plasmacytoma and enabled us to distinguish it from other lymphoproliferative neoplasms such as large B-cell lymphoma.
CD79a, CD19, CD20, and PAX5 are B-cell differentiation antigens that are frequently positive in big B-cell lymphomas, although CD138 and CD38 are plasma cell markers that are usually negative. Extra-medullary plasmacytoma, however, expresses the plasma cell markers CD138 and/or CD38 frequently. 9 The expression of CD79a antigen is almost exclusively seen in B-cell neoplasms before immunoglobulin heavy-chain gene rearrangement during B-cell ontogeny. 10 In late phases of B-cell development and plasma cell cancers, CD79a is downregulated or completely eliminated. 10 Hence, emergency medical technician (EMT) is frequently negative for CD79a expression. 10 Furthermore, in MM, a high Ki-67 index may be a prognostic predictor as well as a risk factor for early recurrence and high mortality.
Extra-medullary involvement in MM is frequently linked to high-risk cytogenetic alterations, resistance to therapy, and a poor prognosis. Few studies have identified that the translocations at (4;14) and (14;16), deletion at (17p), and gaining (1q21) are associated with extra-medullary disease. In addition, deletion of (17p13) and (13q14) may be a potential prognostic marker for MM extra-medullary illness.11,12 However, prognostic indicators for the development of extra-medullary disease are still undetermined.
Surgical excision with adjuvant radiation is the conventional treatment for localized plasmacytoma. However, because the disease was so widespread, systemic therapy (ie, chemotherapy) was suggested for our patient. Chemotherapy is the chosen treatment for large tumors (ie, greater than 5 cm in diameter), high-grade tumors, and relapsed or refractory disease.13,14 Also, intensive multi-agent therapies and allogeneic stem cell transplantation have been reported to be other options.13,14 However, because of the disease’s rarity, there is little experience in treating breast plasmacytoma.
In conclusion, extra-medullary breast plasmacytoma is a rare MM presentation that should be investigated in cases with breast lumps with plasma cell tumor characteristics. To distinguish BP from other breast cancers and to determine a suitable therapeutic approach for the disease, a comprehensive examination involving radiological, histological, and cytogenetic tests is required.
Footnotes
Acknowledgements
The authors thank the patient for allowing us to publish this case report. The authors thank the National Institute of Oncology for their collaboration and support for the publication of the case. We also appreciate Dr. Entsar Jghdam from the National Institute of Oncology in Sabrata, Libya, who provides us with constant assistance and clinical data about the patient.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for her anonymized information to be published in this article.
