Abstract
The coronavirus disease 2019 (COVID-19) pandemic revealed a myriad of postinfectious severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) sequelae, many of which remain poorly understood. We describe a rare presentation of a patient developing 2 simultaneous COVID-19 sequelae: transverse myelitis and acquired von Willebrand syndrome (AVWS). There have been numerous published case reports of patients developing transverse myelitis after a diagnosis of COVID-19. However, none have described AVWS as an observed complication from SARS-CoV-2 infection. To our knowledge, this case report is the first to describe AVWS as a result of COVID-19 infection, suggesting that patients with a prior diagnosis of COVID-19 are susceptible to developing this rare bleeding disorder.
Keywords
Introduction
The coronavirus disease 2019 (COVID-19) pandemic necessitates significant scrutiny of postinfectious sequelae of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the etiologic agent of COVID-19. Much has been published regarding the multitude of COVID-19 postinfectious consequences.1 -13 However, direct attribution to prior COVID-19 infection remains a diagnostic challenge. Neurological complications of COVID-19 include myelopathy, meningoencephalitis, stroke, dysautonomia, and Guillain-Barré syndrome.1 -5 Hematologic complications include lymphopenia, venous thromboembolism, and disseminated intravascular coagulation. 6
Transverse myelitis is a rare complication of COVID-19 infection that has been reported in medical literature. Acquired von Willebrand syndrome (AVWS) is currently not known to be a hematologic complication of COVID-19. On the contrary, von Willebrand factor (VWF), the glycoprotein that is deficient in AVWS, is typically more plentiful and active during acute infection. 7 We present a case involving both clinical entities, including, to our knowledge, a novel presentation of AVWS in the setting of COVID-19 infection. This case illustrates transverse myelitis and AVWS as presenting syndromes that may suggest prior COVID-19 infection.
Case Presentation
A 58-year-old man with a history of shingles presented to the hospital with bilateral lower extremity weakness. He had been diagnosed with COVID-19 3 months prior to presentation but recovered fully without hospitalization. He noticed easy bruising on the dorsa of his hands ~2 to 3 weeks after his diagnosis. Two weeks prior to presentation, he experienced acute urinary retention causing a urinary tract infection and requiring a Foley catheter. He was prescribed ciprofloxacin for treatment. One week prior, he experienced weakness, numbness, and tingling of his bilateral lower extremities. Weakness progressed until the patient could no longer ambulate. He also endorsed fevers and constipation. He denied any weakness in his arms or saddle anesthesia. He denied any family history of bleeding disorders or autoimmune conditions.
The patient’s vital signs were normal at presentation. Neurological examination revealed 4+/5 strength in the hip flexors and extensors bilaterally, 4−/5 in knee extensors bilaterally, 5−/5 in dorsiflexors bilaterally, 3+ in patellar reflexes bilaterally, and 3 beats of ankle clonus bilaterally. Skin examination revealed multiple large ecchymoses on both hands, mostly on the dorsal surfaces.
Magnetic resonance imaging with gadolinium revealed increased spinal cord signal in the thoracic spine from T4 to T9 (Figure 1). Infectious laboratory workup, including blood cultures, arbovirus serologies, syphilis, and etiologic agents of Guillain-Barre, was negative. Analysis of VWF and factor VIII activity confirmed VWF deficiency. Laboratory investigation revealed a VWF activity of 14% and a factor VIII activity of 22%, both below the threshold of normal activity (Table 1). Lumbar puncture was delayed until correction of VWF deficiency to minimize the risk of bleeding. He was given intravenous immunoglobulin (IVIG) dosed at 1 g/kg over 2 days with improvement in VWF and factor VIII activity levels (Table 1). Lumbar puncture was performed 48 hours later; cerebrospinal fluid studies revealed pleocytosis with lymphocytic predominance, elevated protein, and elevated immunoglobulin G levels consistent with transverse myelitis. A paraneoplastic multiplex polymerase chain reaction panel for 14 autoantibodies was negative (Table 2).
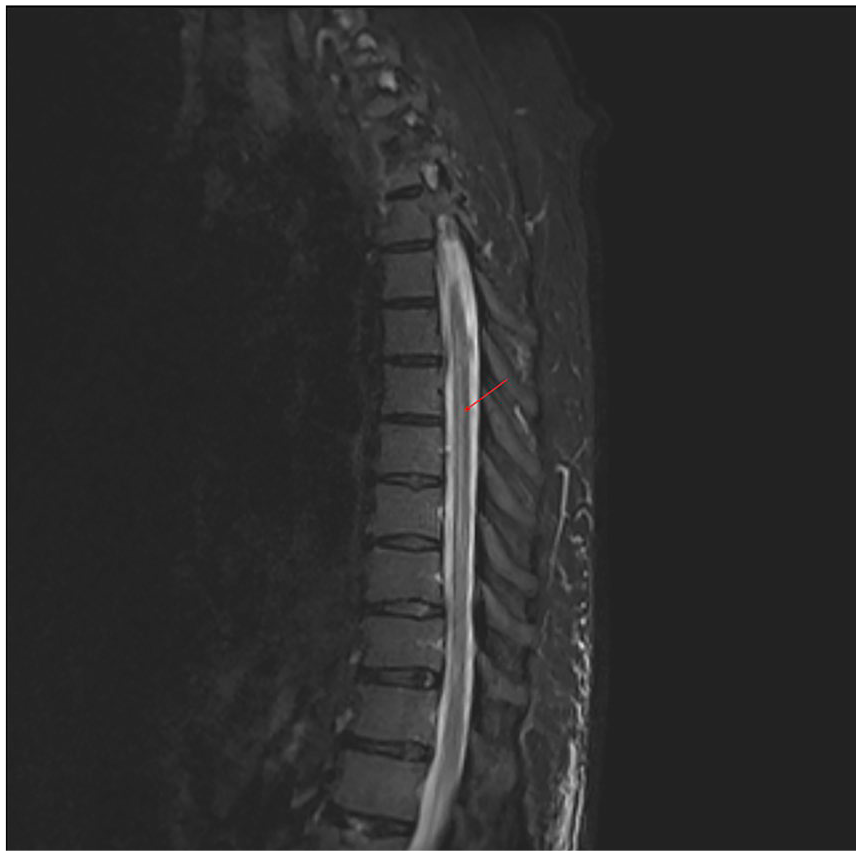
A mid-sagittal magnetic resonance imaging (MRI) view of the thoracic spine shows increased spinal cord signal spanning from T4 to T9 (arrow).
Coagulation Studies.

Abbreviations: IVIG, intravenous immunoglobulin; L, low; VWF, von Willebrand factor; HMW, high-molecular-weight; IMW, intermediate-molecular-weight; LMW, low-molecular-weight; H, high.
Cerebrospinal Fluid Studies.

Abbreviations: IgG, immunoglobulin G; H, high.
A clinical diagnosis of acute transverse myelitis was made, and he was treated with methylprednisolone dosed at 1 g/d for 5 days, during which time his bilateral lower extremity weakness markedly improved. By the time of discharge, he was able to ambulate with the assistance of a rolling walker. His urinary retention and constipation had also fully resolved. He was instructed to complete a structured steroid taper in addition to following up with outpatient physical therapy, neurology, and hematology for further management.
Discussion
Transverse myelitis is inflammation of the spinal cord that disrupts neural pathways, leading to sensory, motor, and autonomic symptoms. The major causes of transverse myelitis include infection, demyelinating diseases, autoimmune diseases, paraneoplastic syndromes, and drugs/toxins; approximately 15% to 30% of cases are idiopathic. 8
For postinfectious etiologies, symptoms of transverse myelitis typically start after the acute infection has resolved. Evidence suggests that onset of neurological symptoms of COVID-19–induced transverse myelitis ranges from 15 hours to 42 days after symptoms begin. 9 The patient in this case had a latency period more than double the upper end of this time range; this highlights the importance of inquiring about any known COVID-19 exposures as far back as 90 days prior to onset of neurological symptoms.
Acquired von Willebrand syndrome is an exceedingly rare bleeding disorder caused by inhibition of VWF activity and levels, with fewer than 700 cases reported worldwide to date. 10 The most common causes of AVWS are lymphoproliferative diseases, myeloproliferative diseases, and cardiac valvular disease such as aortic stenosis. Other causes include medications, autoimmune disorders, and hypothyroidism. Infectious causes have only been rarely described in cases reports with etiologic agents including Epstein-Barr virus. 11 There are several proposed mechanisms for AVWS. One mechanism is immune-mediated in which nonspecific antibodies bind to VWF. This antibody-antigen complex is then cleared from the bloodstream by autoantibodies targeting VWF. Various non-immune-mediated mechanisms include destruction of VWF multimers due to high shear stress conditions (aortic stenosis, mechanical, or prosthetic heart valves), decreased synthesis (hypothyroidism), or increased proteolysis of VWF associated with ciprofloxacin consumption. 12 The mechanism behind infectious etiologies has not been well studied given the rarity of infectious causes of AVWS. However, our patient likely had an immune-mediated disease process given the significant response to IVIG treatment. Of note, our patient did not receive factor VIII or VWF replacement therapy which further corroborates an immune-mediated process given that IVIG administration alone was sufficient for improvement. SARS-CoV-2 has been shown to incite de novo production of autoantibodies against various antigens that lead to the development of numerous autoimmune conditions such as myocarditis, myositis, and thyroiditis. 13 It is reasonable to suggest that our patient may have developed autoantibodies against VWF or factor VIII in the setting of his COVID-19 infection. In addition, the patient was prescribed ciprofloxacin, which is a medication that is associated with AVWS. However, his bleeding symptoms had already started about 1 to 2 weeks prior, making it less likely that ciprofloxacin was the inciting factor for the development of AVWS.
A unique aspect of this case involves the VWF multimer analysis (Table 1). As depicted, there was an increased amount of low-molecular-weight multimers along with normal ranges of high-molecular-weight (HMW) and intermediate-molecular-weight multimers. In typical cases of AVWS, there is a selectively reduced level of HMW multimers. 14 Thus, our case exhibits an atypical multimer pattern of AVWS which could be representative of COVID-19–associated cases of the syndrome.
In this patient, both transverse myelitis and AVWS were attributed to prior COVID-19 infection given that symptoms for both conditions arose after recent infection. This case demonstrates the importance of keeping a broad differential in patients with any history of COVID-19 infection. Clinicians should be particularly vigilant for syndromes that follow even after symptoms of COVID-19 have already resolved. The scale of the global COVID-19 pandemic has unmasked a myriad of syndromes related to SARS-CoV-2, many which are still being discovered as the body of evidence continues to grow. Thus, there is an imperative need for additional research and documented clinical observations to better understand the subsequent neurological and hematological sequelae of active, recent, and remote infection with SARS-CoV-2.
Supplemental Material
sj-docx-1-hic-10.1177_23247096221109205 – Supplemental material for A COVID Constellation: A Case of Transverse Myelitis and Acquired von Willebrand Syndrome
Supplemental material, sj-docx-1-hic-10.1177_23247096221109205 for A COVID Constellation: A Case of Transverse Myelitis and Acquired von Willebrand Syndrome by Collins Mbonu, Nneka Molokwu and Alexander Matelski in Journal of Investigative Medicine High Impact Case Reports
Footnotes
Acknowledgements
We would like to acknowledge Mary Ann Kirkconnell Hall, MPH, Senior Medical Writer for the Division of Hospital Medicine at Emory University School of Medicine, for her assistance with the formatting and editing of this case report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
