Abstract
Several guidelines endorsed the indefinite use warfarin or heparin-containing products for acute venous thromboembolism (VTE) treatment and secondary prevention and discouraged the use of direct oral anticoagulants (DOAC) for patients diagnosed with antiphospholipid syndrome (APS). However, adequate anticoagulation despite warfarin therapy remains a challenge in APS patients. Using DOACs in APS patients is seen in clinical practice, despite the lack of evidence to support their use in this population. In this case series, we aim to evaluate the safety and effectiveness of apixaban use in nine patients with primary or secondary APS at King Abdulaziz Medical City (Riyadh, Saudi Arabia). All patients presented with APS and received apixaban with or without concomitant antiplatelet. Three patients had double positivity, and two patients had triple positivity of antiphospholipid antibodies (aPL). Some patients tolerated apixaban during the follow-up period, but recurrent VTE and stroke were reported in some of them. Bleeding complications were evident in some cases as well. In conclusion, warfarin remains the best choice to prevent VTE recurrence in patients with APS. On the other side, apixaban use in patients with APS may have some safety and effectiveness concerns evidenced by VTE recurrence and bleeding complications. The safety and effectiveness of utilizing apixaban in APS patients need to be assessed in well-controlled randomized trials.
Introduction
Thrombotic antiphospholipid syndrome (TAPS) is a manifestation associated with antiphospholipid syndrome (APS) with an increased incidence of venous, arterial, or microvascular thrombosis in the setting of persistent production of autoantibodies against various phospholipids.1-3 Antiphospholipid antibodies (aPL) that play a role in pathogenesis and determine the thrombotic risk include lupus anticoagulant (LAC), anticardiolipin antibodies (ACA), and anti-beta 2 glycoprotein-I antibodies (B2GPI).1-3
The interaction between the aPL and coagulation factors leads to the suppression of tissue factor pathway inhibitor and inhibition of protein C resulting in thrombin generation. 3 Antiphospholipid antibodies bind to platelets and endothelium, activating platelets, and the complement system. 3 As a result, the risk of myocardial infarction, venous thromboembolism (VTE), ischemic stroke, and peripheral arterial disease increases by 16-fold in patients with APS. 4 After the first thrombotic event, the recurrence rate increases sharply by 10% to 67%, and patients with triple-positive aPL are at higher risk for recurrence. 3
Warfarin is the cornerstone in treating VTE in patients with APS.5,6 However, over time, it became evident that warfarin perils in APS patients include warfarin resistance, in which patients require higher weekly doses to achieve therapeutic international normalized ratio (INR). 7 In addition, the estimated recurrence rate of thrombosis despite warfarin therapy is 5% to 20%, and increasing the INR intensity goal from (2-3) to (3-4) remains controversial. Some historical studies had suggested a higher intensity of vitamin K antagonist (VKA) therapy to prevent recurrences.3,8 Thromboplastins are variously responsive to aPL, and such interferences might perturb INR monitoring in some APS patients suggesting a falsely INR value.3,8
Direct oral anticoagulants (DOACs) provide similar efficacy to warfarin in treating and preventing atrial fibrillation and VTE with a better safety profile, and recommended over VKA.9-11 However, APS was underrepresented in the previous randomized controlled trials (RCTs).12-15 In the AMPLIFY trial, Apixaban 5 mg twice daily was compared to warfarin (INR target of 2.5) for VTE treatment; the baseline risk factors for recurrent VTE, including thrombophilia, were 2.85% and 2.2% in the apixaban and warfarin arm, respectively. 15 There was no subgroup analysis performed on the safety and efficacy in this specific patient population. 15 The DOACs have multiple advantages over warfarin, including fixed dosing, INR regular monitoring is unnecessary, and a more predictable pharmacokinetic and pharmacodynamic profile. 16 Thus, DOACs may become an appealing option for patients with APS, yet the evidence about DOAC effectiveness and safety in this patient population is limited.
Several guidelines recommended the indefinite standard-intensity VKA or heparin-containing products for either acute VTE treatment or secondary prevention, and discouraged DOAC use in the context of APS.5,6 Nonetheless, some of these guidelines limit the use of DOACs to patients who are nonadherent to warfarin due to limited evidence supporting their use. 5 The majority of the conducted RCTs, case reports, and case series investigated the utility of rivaroxaban on multiple DOAC agents in patients with APS.12,17-19 Among the DOACs, rivaroxaban seems to be the most extensively investigated compared with dabigatran and apixaban in patients with APS. 17 At the same time, little data exist on apixaban safety and effectiveness to prevent secondary thrombotic events in APS. 20 Therefore, this case series aimed to evaluate the safety and effectiveness of apixaban in APS-positive patients.
Methods
Study Design
This case series included nine adult patients with confirmed APS diagnosis who were using apixaban at King Abdulaziz Medical City (KAMC), Riyadh, Saudi Arabia, from January 1, 2016, until December 31, 2019. This case series aimed to describe the prevalence of thrombosis and bleeding in those populations. The study was approved by King Abdullah International Medical Research Center (Ref.# NRC21R/549/12).
Study Participants
This case series included all adult patients (age ≥18 years) with objectively diagnosed APS using apixaban between January 1, 2016, and December 31, 2019. Patients who started on apixaban were actively managed and followed up by King Abdulaziz Medical City providers.
Study Setting
This study was conducted in King Abdulaziz Medical City (Riyadh, Saudi Arabia). This is a large tertiary-care academic referral hospital with 1501 beds that provides all types of care to all National Guard soldiers and their families, starting from primary health care to specialized tertiary care.
Data Collection
We collected patients’ demographic data, comorbidities and laboratory tests, CHA2DS2-VASc, and HAS-BLED scores at the time of initiation. Moreover, laboratory tests such as coagulation profile, liver profile, renal profile, and complete blood count (Hgb and platelet counts in particular) were collected. Furthermore, thrombosis (i.e., stroke, pulmonary embolism [PE], deep vein thrombosis [DVT]), major bleeding, and minor bleeding confirmed by documentation were recorded for all patients.
Results
Nine patients had confirmed APS and were treated with apixaban. The mean age of included patients was 42.2 years, and females represented most of the cases. Six patients received aspirin concomitantly, and two patients used clopidogrel as a concomitant antiplatelet, as shown in Table 1. Most patients had primary APS except for one patient with a secondary APS. Three patients had double positivity, and two patients had triple positivity of aPL. In addition to APS, some patients had other comorbidities as presented in each individual case presentations.
Participants’ Characteristics Upon Initiation of Apixaban.
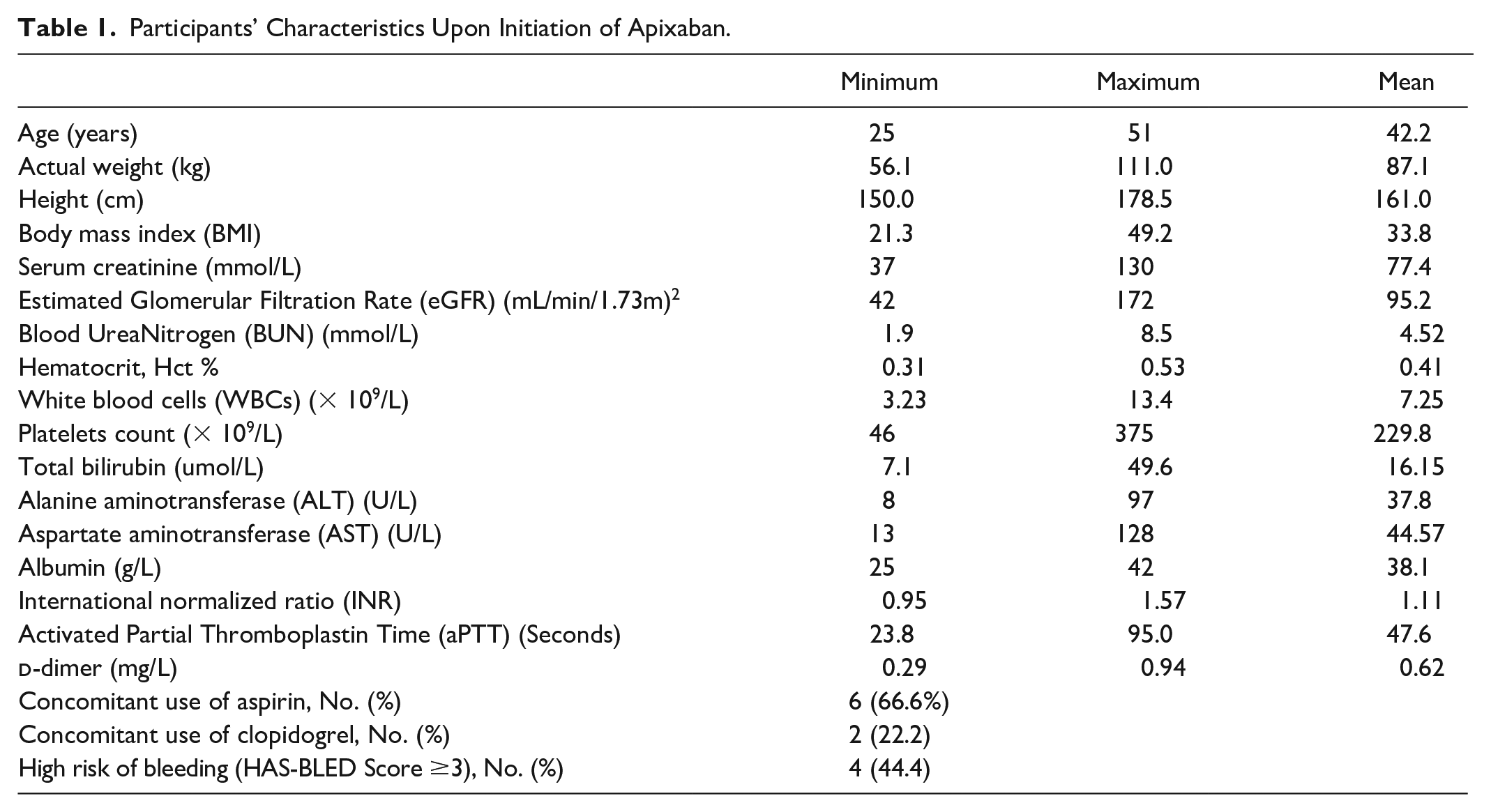
Upon patient follow-up, three patients developed VTE after apixaban initiation, and one patient had a stroke within 12 months of apixaban initiation. In terms of bleeding outcomes, two patients have developed major bleeding in the form of intracranial bleeding, and onepatient developed minor bleeding withno evidence of other types of bleeding in the remaining cases as represented in Table 2.
Clinical Manifestations of APS, Thrombosis, and Bleeding.
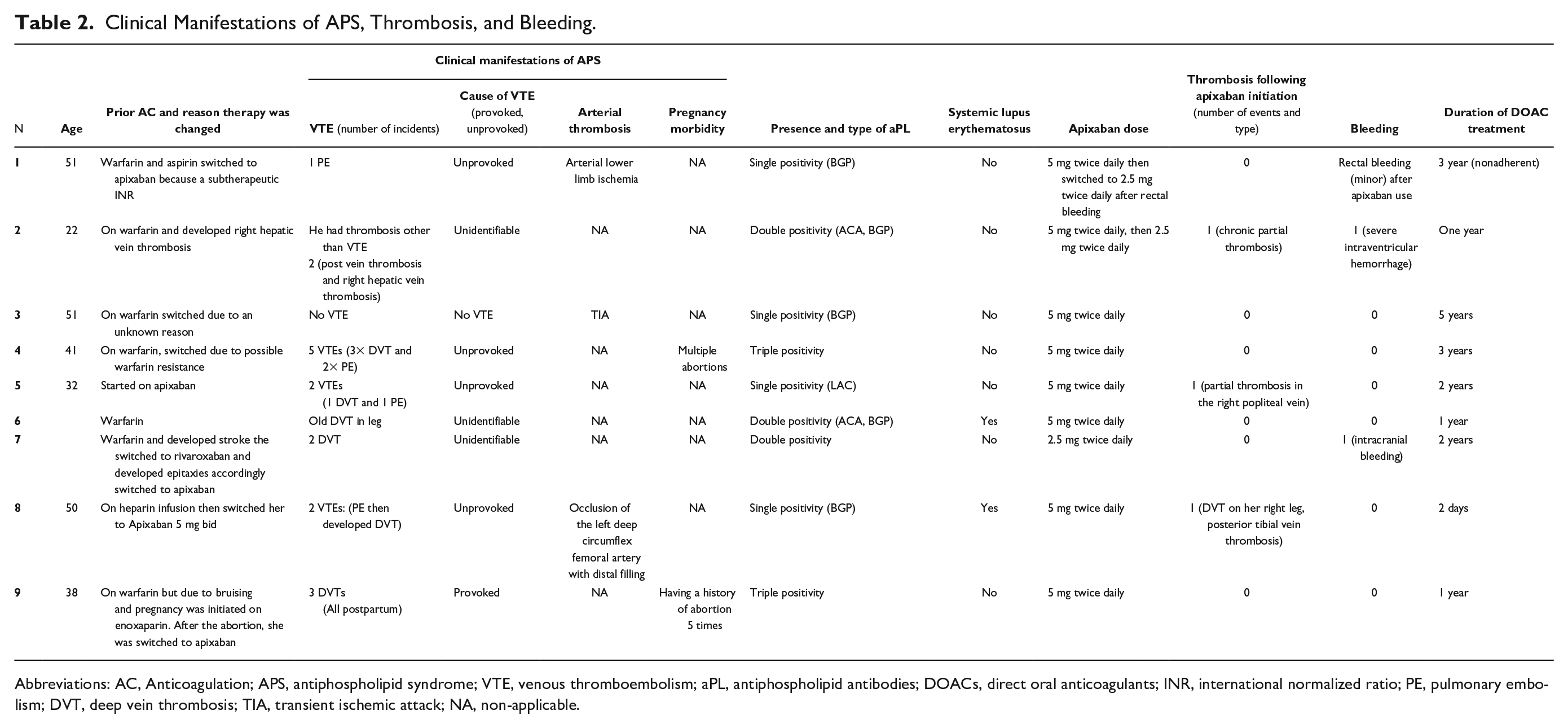
Abbreviations: AC, Anticoagulation; APS, antiphospholipid syndrome; VTE, venous thromboembolism; aPL, antiphospholipid antibodies; DOACs, direct oral anticoagulants; INR, international normalized ratio; PE, pulmonary embolism; DVT, deep vein thrombosis; TIA, transient ischemic attack; NA, non-applicable.
Patient Cases
Case 1
A 51-year-old male with a body mass index (BMI) ~43 kg/m2 and a history of unprovoked PE and DVT while on warfarin and aspirin. The patient reported being nonadherent to warfarin therapy. He started to follow-up in our center in 2018; at that time, his baseline INR was subtherapeutic at 0.9. Upon hematology consultation in mid-2018 and following a thrombophilia workup that shows positive B2 glycoprotein I (BGP) IgG (24.22) with negative anti-cardiolipin antibodies (ACA) (also LAC was indeterminate). He was diagnosed with primary APS (single positivity) requiring lifelong anticoagulant on apixaban 5 mg twice daily. One year later, the patient was admitted due to ST-elevation myocardial infarction (STEMI) requiring percutaneous coronary intervention (PCI) and discharged on clopidogrel 75 mg once daily, metoprolol tartrate 50 mg twice daily, and atorvastatin 80 mg. From the start of apixaban in mid-2018, until his last follow-up in September 2021, there were no recurrent venous thrombotic events except for a minor bleeding (per rectum) event that occurred a year after apixaban initiation. Currently, the patient is on 2.5 mg twice daily apixaban after the minor bleeding.
Case 2
A 22-year-old male with a past medical history of chronic kidney diseases, vein thrombosis, Budd-Chiari syndrome diagnosed in 2013, heart failure, post-transjugular intrahepatic portosystemic stent shunt. Also, the patient had a history of esophageal bleeding, acute pancreatitis complicated by acute respiratory distress syndrome, and decompensated liver disease (Child B) with hepato-pulmonary syndrome. He was diagnosed with primary APS (double positivity) and started on warfarin in 2016. One year after warfarin therapy, the patient developed right hepatic vein thrombosis so the hepatology team switched him to apixaban 5 mg twice daily at that time. In early 2018, the apixaban dose was reduced to 2.5 mg twice daily (no reason was documented for the dose reduction). Following apixaban initiation, he was admitted to the hospital twice within 6 months as a case of chronic partial thrombosis of the distal right superficial femoral vein, and another admission in later 2018 with brain computerized tomography (CT) revealed suspected developmental venous anomaly or vascular malformation in the right periventricular location and right thalamus. The patient died in the following admission due to severe intraventricular hemorrhage while on apixaban 2.5 mg twice daily.
Case 3
A 51-year-old female with a past medical history of transient ischemic attack (TIA), dyslipidemia, and diabetes mellitus (DM). A thrombophilia workup shows very high levels of B2 glycoprotein I IgA (18.42-37.14); 4 readings were elevated with negative anti-cardiolipin antibodies (LAC was not available). She was diagnosed with primary APS (single positivity) requiring anticoagulation. The patient had been receiving warfarin since 2014 but complained of headaches due to warfarin therapy without radiological findings. In 2016, she switched to apixaban 5 mg twice daily with no recurrent thrombotic events and bleeding events until mid-2021.
Case 4
A 41-year-old female with a past medical history of right-sided heart failure, chronic thromboembolic pulmonary hypertension (HTN), chronic kidney disease G2-A2, and cholelithiasis. The patient has a history of APS and was receiving warfarin 3 mg once daily. She developed several recurrent VTE episodes (3× DVT and 2× PEs) while on warfarin therapy. In mid-2018, she developed nonocclusive partial thrombosis of the right external iliac vein and common femoral vein. Patient INR was 3.5, and factor II level was 0.32, which was suppressed but was not correlated with INR if >3; the mixing study is corrected. Upon consulting the hematology team, they concluded that there was no evidence of lupus antibodies affecting the INR, but she might have had warfarin resistance. In addition, the patient has positive Lupus anticoagulant (LAC) , and the patient’s last anti-beta2-glycoprotein I (BGP) IgA was 6.5, anticardiolipin antibody (ACA) IgM level was 19.06. Later, she was switched to apixaban 5 mg twice daily. From the start of apixaban on February 14, 2018, until her last follow-up on December 19, 2021, there were no reported recurrent thrombotic or bleeding events.
Case 5
A 32-year-old female with a history of benign intracranial HTN, DVT, and PE due to contraceptive use, LAC, BGP antiphospholipid, and thyroidectomy due to papillary carcinoma. She was originally on apixaban 5 mg twice daily due to VTE; in early 2020, the patient was found to have APS with positive LAC; patient BGP IgA, IgG, IgM were 8.15, 42.13, 4.36, respectively. From the initiation of apixaban in 2019 until her last follow-up, there was only one recurrent thrombotic event as partial thrombosis in the right popliteal vein and no bleeding events.
Case 6
A 51-year-old female with a known case of DM, old leg DVT, and a positive family history of systemic lupus erythmatosu (SLE). She was taking warfarin and complained of nausea/vomiting and photophobia with a severe headache. Early in 2018, she was evaluated by magnetic resonance imaging (MRI)/magnetic resonance angiography (MRA)/magnetic resonance venography (MRV), which revealed multiple variable-sized acute infarctions in the deep white matter of the left periventricular region, parietotemporal lobe, and subcortical left occipital lobe. And she was diagnosed with right sigmoid and internal jugular vein thrombosis. Warfarin was on hold, and the autoimmune serology was obtained, which showed LAC with positive anti-cardiolipin (IgM) 45.69 and anti-cardiolipin (IgG) 63.07 +ve and beta glycoprotein (IgM) 2 beta glycoprotein (IgG) 120.61 +ve and beta glycoprotein (IgA) 22.17 and INR was 5.84. Months later, the neurology team recommended starting her on apixaban 5 mg twice daily for life. After 6 months of apixaban initiation, the repeated test for antiphospholipid antibody syndrome (APLS) showed BGP IgA 27.17 and IgG 40.29 and IgM 3.41, antinuclear antibody (ANA) 60.94; the patient developed thrombocytopenia, and apixaban was replaced by enoxaparin and then by warfarin.
Case 7
A 42-year-old female with a known case of APLS, HTN, bipolar depression, epilepsy with recurrent VTE, and multiple strokes. The patient was taking aspirin 81 mg for secondary prevention. The patient developed a stroke in 2015 while receiving warfarin, then switched to rivaroxaban and developed epitaxies. A serology test was conducted which revealed BGP IgA 6.78, IgG 100, IgM 3.31, ACA IgM 0.60, ACA IgA 24.09, ACA IgG 72.12, antinuclear antibodies (ANA) 13.73. A diagnosis with double-positivity APLS. The hematology team recommended starting her on apixaban 2.5 mg twice daily in mid-2016 and continuing aspirin (81 mg every 24 hours) as secondary prevention. The patient remained almost 2 years free from any thrombotic or bleeding events. However, in 2018, an MRI imaging was performed and showed an acute infarction, likely embolic in nature. During this event, apixaban therapy was held until the patient stabilized and then reinitiated. The latest records showed that the patient died in 2020 with a probability of stroke as a cause.
Case 8
A 50-year-old female is known to have APS (single positivity), otherwise medically and surgically free. She was admitted in 2018, complaining of sudden onset bilateral foot pain that occurred even at rest and blue discoloration in the tip of toes in both feet but resolved upon admission. The patient had a prior similar episode, and she sought medical advice. Upon investigation with CT, the patient was found to have a major PE in the left pulmonary artery and occlusion of multiple arteries in the lower limbs (subacute bilateral lower limb ischemia). Laboratory findings showed positive B2 glycoprotein and IgM was 57.94. She was started on heparin infusion, atorvastatin 40 mg daily, and alteplase infusion. One week after the heparin infusion, she was switched to apixaban 5 mg twice daily for 2 days, then developed DVT on her right leg, and posterior tibial vein thrombosis. After she that, was switched to enoxaparin 80 mg subcutaneous every 12 hours for a month and continued with warfarin until her most recent follow-up (June 10, 2020) with no thrombotic events detected.
Case 9
A 38-year-old female was diagnosed with primary APS (triple positivity). During an outpatient clinic visit in 2017 due to a history of postpartum recurrent DVT (3 times in the last 8 years) and a history of multiple consecutive pregnancy losses (2 in first trimester, and 1 in second trimester). Laboratory findings are as follows: in January 2017, ACA IgG was 111.08 but ACA IgG was 96.6 in April. In January 2017, B2 glycoprotein IgG was 27.55 but was 21.19 (strong positive). In February 2018, she had a positive LAC. She was on lifelong anticoagulation with warfarin, but she stopped 2 weeks before the clinic visit due to bruising. At that time, the patient was pregnant and started on enoxaparin 70 mg subcutaneous twice daily for 120 days. Two months later, she returned to the clinic after another abortion (fifth time). So, she was switched from enoxaparin to apixaban 5 mg twice daily. A year later, her treating team stopped apixaban due to insufficient evidence to use in APS and put her warfarin with a target INR of 2-3. She was continued with warfarin for life, taking into consideration to switch to enoxaparin if pregnant.
Discussion
The current case series provides additional knowledge on the usefulness and safety of apixaban for secondary prevention in patients with APS ranging from single- to triple-positive aPL. To our knowledge, this is the largest case series primarily focusing on apixaban use in patients with APS. Thrombotic events after apixaban initiation were observed in 5 patients (55.5%). This rate was higher than previously reported thrombotic recurrence in the previous case series from 0% to 6%. This discrepancy in the rate of recurrent may be driven by other DOAC agents used in the prior studies and the variation in the follow-up period.17,21 In this case series, the reported bleeding rate was 22%, in which one patient developed a major bleeding event (intracranial bleeding and lower gastrointestinal [GI] bleeding). The bleeding rate is similar to the previous case series where one out of three patients (33%) with APS who received apixaban developed nonfatal GI bleeding. 17 The current role of apixaban in treating APS remains unclear given the limited data available. Thus, a detailed treatment discussion addressing benefits and thrombotic recurrence risk with apixaban in patients with a high levels of warfarin aversion (eg, labile INR, multiple recurrent thrombotic episodes) remains the key factor for apixaban initiation in this patient population.
The potential use of apixaban in APS patients requires consideration similar to those non-APS patients who were underrepresented in clinical trials such as patients who are obese, with renal and hepatic failure, dialysis, older adults, or in those who are receiving interacting drugs. Several systematic reviews and meta-analyses compared DOAC with warfarin in patients with APS and showed that DOACs are not as effective as warfarin or even showed a significantly higher risk of thrombotic events with DOAC.22,23 Moreover, patients with APS who were treated with DOACs had an increased risk of recurrent thromboembolic events and recurrent VTE alone compared with those on VKAs with no differences between rivaroxaban and apixaban (risk ratio = 0.82, 95% confidence interval: 0.12-3.54). 24 The ASTRO-APS, an ongoing RCT compared apixaban (2.5 or 5 mg) with warfarin targeting an INR 2-3, was modified to exclude patients with prior arterial thrombosis due to a possibility of higher rates of stroke incidence, especially in patients with prior history of stroke.25,26 In the current case series, two patients had a history of either prior stroke or TIA. One of those experienced two recurrent stroke events while on a suboptimal dose of apixaban at 2.5 mg twice daily. On the contrary, the second patient was event-free during the treatment period with apixaban 5 mg twice daily, both patients were on antiplatelet therapy. Our findings align with Pengo et al. in terms of the addition of aspirin to the treatment regimen for patients with prior arterial thrombosis remains unknown but may be beneficial. 19
Second, the data safety monitoring modified the dose of apixaban to only include the 5 mg twice daily. In the AMPLIFY-EXT, patients who required long-term VKA such as aPL were excluded. 27 It is important to highlight that phase 3 trials of DOACs compared with warfarin in the general population demonstrated the efficacy of DOACs compared with standard-intensity warfarin targeting an INR of 2-3.12-15 Thus, the doses of DOACs used in these studies may not provide sufficient protection against thrombosis in patients who require high-intensity anticoagulation, such as patients with multiple recurrences, triple-positive aPL, warfarin resistance, patients with combined arterial and venous thrombosis, or patients with a history of stroke. 11 A systematic review and meta-analysis of patients with APS using DOAC reported that the rate of thrombosis recurrent in patients treated with standard-dose apixaban was approximately 1%. 28 Currently, there is the RISAPS trial (RIvaroxaban for Stroke Patients With AntiPhospholipid Syndrome) which is an ongoing trial anticipated to be completed by December 2023. 29 The study aims to assess the efficacy of rivaroxaban 15 mg twice daily versus high-intensity warfarin targeting INR 3.5 in patients who have APS with a history of stroke or other ischemic brain manifestations. 29 Until then, the optimal DOAC dosing to prevent recurrence remains unknown. It is important to note that the recurrence occurred in patients several months after they were initially event free. The time to recurrence remains a great concern due to its unpredictability. For example, in the Ordi-Ros study, arterial events in the rivaroxaban group occurred up to 31 months after randomization. 30 Similarly, the Trial of Rivaroxaban in Antiphospholipid syndrome (TRAPS) trials reported that 57% of the arterial events occurred within 8 to 23 months after randomization. The current case series adds to that evidence since we did observe the incidence of recurrence up to 48 to 60 months albeit doing well on apixaban. 19
This case series proposed that the potential role of apixaban in APS carries a high risk of recurrence and a high failure rate with unpredictable timing. However, the subtherapeutic INR (2-3) in patients with APS treated with warfarin and the underdosing of patients on apixaban (2.5 mg twice daily) after switching from warfarin may influence the thrombosis and bleeding risk of patients included in this study. Our case series has several limitations. First, the inherent nature of the case series and the small sample size. Second, the study’s retrospective nature limits the retrieval of essential patient information such as VTE risk factors, accurate dosing regimen during the follow-up period, and adherence to warfarin. Therefore, warfarin failure or resistance may be influenced by those confounding factors. Third, our case series took into account the following: (1) the clinical heterogenicity of APS, (2) heterogeneous aPL phenotype as triple-positive aPL inclusion deemed to be at high risk for recurrence at baseline. However, this could potentially shift the focus of future studies to assess DOAC in patients with APS and lower thrombotic APS risk phenotype. In addition, to further evaluate the suggestion by Sato et al that anti-Xa agents might have a high failure rate and considering empiric dabigatran in these patients requires further studies to validate its use. 31
Conclusion
Our case series raises a concern about the safety and effectiveness of apixaban in patients with APS, evidenced by recurrent VTE and bleeding complications. Warfarin remains the best option to prevent VTE recurrence in patients with APS. As patients with APS are usually prescribed warfarin for life long, the issue arises when patients’ preference is DOAC, or developed VTE recurrence while adherent to warfarin. Therefore, apixaban may be considered in patients who refuse warfarin therapy, are intolerant to warfarin, or have poor anticoagulant control despite adherence.. However, the results of the upcoming RCT and other RCTs investigating apixaban the safety and efficacy of apixaban use in patients with APS compared with warfarin are essential.
Footnotes
Acknowledgements
We would like to acknowledge the Saudi critical care pharmacy research (SCAPE) investigators who participated in this project. In addition, we like to thank Ali Al Zahrani for the data collection support from electronic sources.
Author Contributions
All authors contributed to data collections, analysis, drafted, revised, and approved the final version of the manuscript.
Availability of Data and Materials
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The study was approved, by King Abdullah International Medical Research Center, Institutional Review Board, Riyadh, Saudi Arabia (Ref. #: NRC21R/549/12). Participants’ confidentiality was strictly observed throughout the study by using anonymous unique serial numbers for each subject and restricting data only to the investigators.
Informed Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
