Abstract
Splenic rupture due to any cause is a life-threatening complication and commonly attributed to trauma. Atraumatic splenic rupture is very rarely reported, and the incidence is currently unknown. Anticoagulants and dual anti-platelet medication can increase the chances of a splenic rupture. Surgical removal of the spleen may be warranted to prevent a life-threatening bleeding. Early identification and intervention are required for most patients as only a few qualify for medical management.
Introduction
Splenic ruptures are potentially fatal, mostly attributed to trauma and less commonly to atraumatic etiology. In the absence of a defining abdominal pain or signs of peritonitis the diagnosis can be easily missed. Atraumatic splenic rupture (ASR) is reportedly associated with apixaban 1 and dual antiplatelet therapy 2 but neither was concurrently associated in the same patient. Atrial fibrillation is independently reported 3 to be associated with spontaneous rupture of the spleen. An appropriate clinical examination and computed tomography (CT) of the abdomen are vital for diagnosis and management. 4 Early risk stratification of patients to determine those eligible for active surgical and vascular interventions is warranted to minimize morbidity and mortality. ASRs are managed similarly to other splenic ruptures, whereas most of the lower grade ruptures can be medically managed. Recently, nonsurgical management is preferred for select patients to protect immunologic function following splenectomy. 5
Case Presentation
An 81-year-old male with a past medical history of hypertension, diabetes mellitus type 2, coronary artery disease, atrial fibrillation, and myasthenia gravis presented to the hospital with complaints of left-sided abdominal pain for few hours. He denied any trauma or fall. The abdominal pain had an intensity of 6 out of 10 on the pain scale, with no aggravating or relieving factors. It was associated with dry cough without any fever. He underwent left main coronary artery stenting 60 days prior to the presentation and was taking apixaban, clopidogrel, and aspirin for the management of the atrial fibrillation and coronary artery disease. He had minimal left upper quadrant tenderness on palpation without any guarding or rigidity. A CT of the abdomen (Figure 1) revealed splenic laceration, hemoperitoneum, and grade 2 perisplenic hematoma according to the American Association for the Surgery of Trauma splenic injury scale. Serum hemoglobin ranged from 8.1 to 9.3 g/dL throughout the hospital stay. He was monitored in the critical care unit for hemodynamic instability and surgical intervention was deferred due to stable hematoma. The patient’s apixaban, aspirin, and clopidogrel were held during the hospital stay. Repeat CT of the abdomen showed interval resolution of the perisplenic hematoma. At discharge, only clopidogrel was resumed.
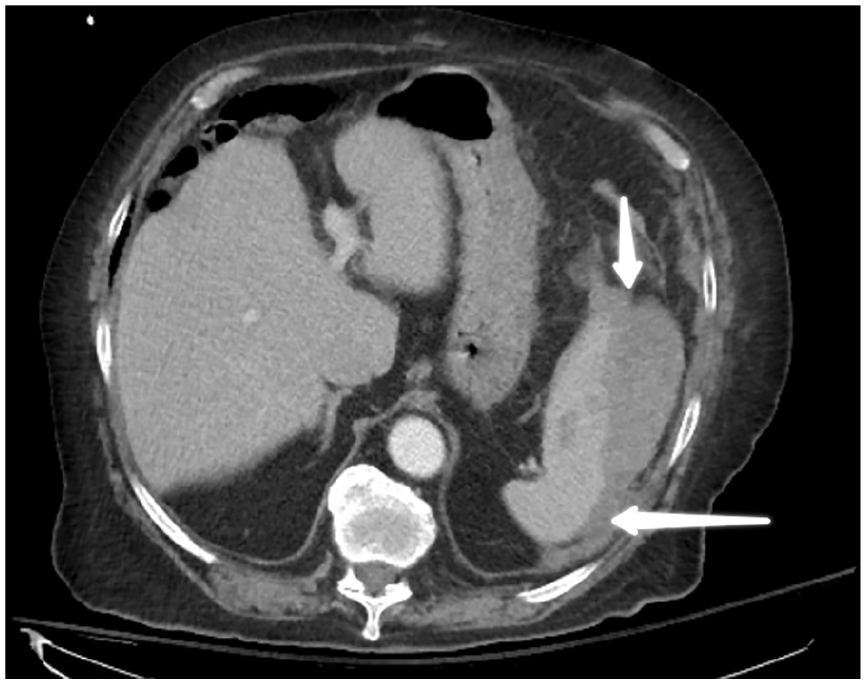
Spleen shown with white arrows showing splenic injury.
Discussion
We report the first case of ASR in a patient on apixaban and dual antiplatelet therapy in the absence of any other inciting risk factors. Splenic rupture is a well-known life-threatening condition, with a mortality risk ranging from 12% to 20%.6,7 A vast majority of splenic injuries are traumatic in origin, with a small proportion of patients presenting with ASR. Other major causes of nontraumatic rupture of spleen reported are neoplasm, infections, inflammatory diseases, medications, and medical treatment, mechanical causes such as direct abdominal trauma and idiopathic.8,9 Depending on the severity of the splenic rupture, clinical symptoms range from mild to very severe abdominal pain often associated with clinical signs suggestive of hemorrhagic shock. Ultrasound of the abdomen (57.1%) is less sensitive when compared with a contrast-enhanced CT of the abdomen (85.7%) in diagnosing ASR. 10 Hemodynamic and hemoglobin monitoring is critical to assess for ongoing blood loss. Etiologic factors, splenic injury grading, and clinical progression determines the need for surgery.6,11 The etiology of splenic rupture has been reported to be recognized in around 38% of cases before surgery and 50% during or after surgery. Total splenectomy is the primary treatment of choice for patients with hemodynamic instability or high-grade rupture or those with a pathologically altered spleen. Temporary measures including splenic artery embolization are reserved for candidates who are bleeding and unable to tolerate surgery. Very few patients qualify for nonsurgical treatment since conservative management is reported to have high failure rates. 7 Even though enoxaparin is reported to accumulate in the spleen preferentially,12,13 the data on apixaban or its mechanism is currently unavailable. Multiple systemic diseases like polycythemia vera, chronic pancreatitis, pancreatic cancer, or splenic flexure colon tumor can affect and damage the architecture of the spleen making it fragile and vulnerable to minor trauma. 11 Some older patients with baseline spleen damage may not recognize the inciting trauma. 14 Surgical management is also recommended in patients with unclear etiology of splenic injury so the spleen can be sent for pathologic evaluation to rule out systemic diseases, especially malignancy. 7 Although the emergent nature of splenectomy does not allow adequate time for vaccine prophylaxis, some nonsurgical candidates may benefit from vaccination in anticipation of a nonfunctional spleen and a possible surgery later.
Conclusion
Patients on apixaban and dual antiplatelet therapy are at high risk for ASR and the management is based on the grade of splenic rupture. For ASR, appropriate treatment stratification is necessary as some patients qualify for early surgical management. The literature on restarting anticoagulation or antiplatelet medication after splenic rupture is currently very limited.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Verbal informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
